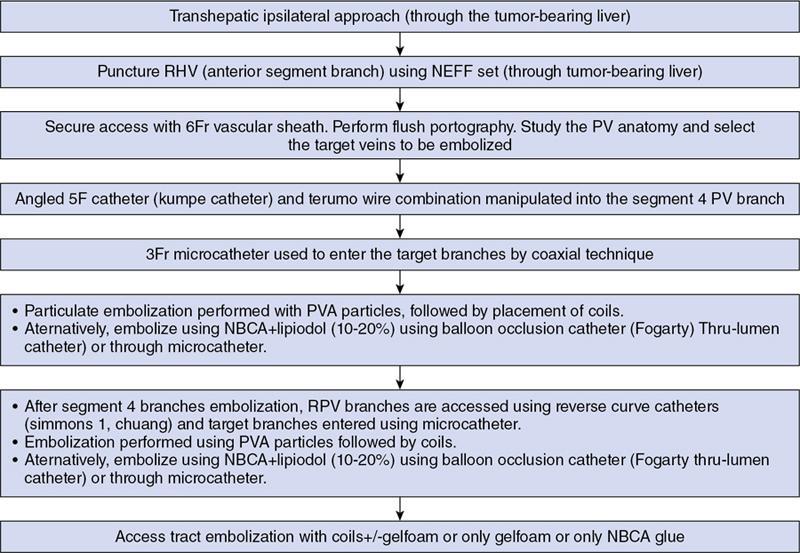
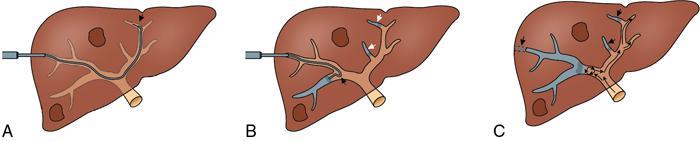
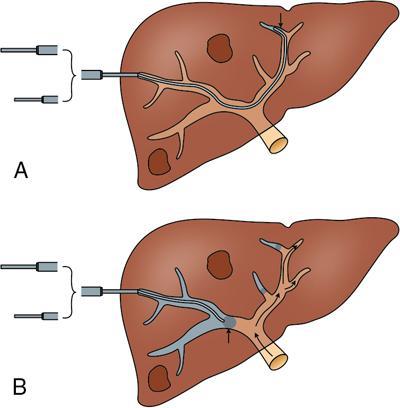
Srikanth Moorthy Liver resection is the mainstay of curative treatment for various primary and secondary hepatic malignancies. Advances in hepatobiliary surgical techniques have led to an increase in the major hepatic resections for various indications. Postoperative liver insufficiency can sometimes be life-threatening. The reported mortality after major liver resection ranges from 3.2% to 7% for patients with noncirrhotic liver and around 32% for patients with cirrhosis. The most common cause of mortality is postoperative liver insufficiency. Though postoperative liver insufficiency is multifactorial, a future liver remnant (FLR), that is, the percentage of the liver that remains after surgery, has shown to be a strong, independent predictor of postsurgical hepatic insufficiency related complications, which is related to size and function of the remnant liver. Preoperative portal vein embolization (PVE) can decrease the postoperative morbidity by increasing the size of the FLR and can also increase the number of patients eligible for curative liver resections. In this chapter, we discuss the indications, principle, technical considerations, current status and future directions of PVE. PVE is a technique used to selectively occlude the blood supply to one of the lobes of liver, diverting portal blood flow to the other lobe, the FLR. PVE was developed based on the fact that the liver can regenerate. Liver regeneration is the liver’s response to the injury. Liver regeneration happens by an increase in both the number and size of the liver cells. The regenerative response depends on the severity of injury with smaller injuries leading to focal mitotic response. Injuries where more than 10% of the liver is involved, a more diffuse response is noted by the rest of the normal liver with around 95% of cells undergoing mitosis. Animal studies have shown that postresection regeneration depends on the growth factors released by the liver and also on the extrahepatic growth factors transported by the portal vein. Studies have demonstrated that the degree and timing of liver regeneration correlate with the transient increase in portal flow following PVE. Based on the animal models studies, proposed mechanisms of hypertrophy are multifactorial that include the release of intrahepatic growth factors, nitrous oxide, gut-derived growth factors, and cytokines such as transforming growth factor-α and tumour necrosis factor (TNF)-α, carried by the portal vein. Hepatocyte growth factor (HGF) released by the remaining liver cells plays a crucial role in liver regeneration. Along with its direct effects on hepatocyte replication, HGF also stimulates the release of cytokines such as interleukin-6 and TNF-α, which also promote regeneration. Insulin acts synergistically with HGF and other growth factors and plays an important role in liver regeneration. These mechanisms explain why that cirrhotic patients with the diminished portal flow and diabetic patients with insulin resistance usually have diminished or delayed liver degeneration. Studies have shown that regeneration after PVE occurs slower than after hepatic resection. Noncirrhotic livers regenerate at the following rate after PVE, 12–21 cm3/day at 15 days, around 11 cm3/day at 30 days, and 6 cm3/day at 35 days. In patients with cirrhosis and diabetes liver regeneration is slower, approximately at 9 cm3/day at 15 days. Though the first PVE was performed in 1984, its use for inducing hypertrophy before hepatic resection was reported by Makuuchi et al. in 1990. In patients undergoing major hepatic resection, there is a sudden elevation in portal pressure at resection that can result in hepatocellular damage in the residual liver. This change in the portal pressure and manipulation during surgery may result in postresection liver dysfunction. PVE prevents perioperative liver dysfunction by mitigating the sudden increase in the portal pressure during surgery and also by increasing the FLR mass. This was further validated by the studies which showed that an increase in FLR volume is associated with improved biliary excretion, albumin uptake and postoperative liver function test values. Patients with primary or metastatic liver tumours eligible for resection and whose estimated FLR volume is not large enough for adequate function in the perioperative period. Points to consider when planning for PVE: FLR measurement is a very critical steps in planning for PVE and also in determining its outcome. Overestimation of FLR may lead to risk of postresection liver failure and the underestimation of FLR may lead to patients undergoing unnecessary PVE and delay the surgery. It is extremely important to consider the methodology used to estimate the FLR and liver function, and the other clinical factors that influence the FLR volume to minimize the risk of postresection liver failure. Absolute volume alone is not sufficient to predict the benefit of the PVE. There is a linear correlation between the liver volume and the body size and larger patients require larger liver mass to support essential function. Hence FLR should be standardized relative to the TELV. Standardized FLR (sFLR), expressed as a percentage of TELV is used clinically in decision-making in PVE. Computed tomography (CT) volumetry is a common and established method used for measuring volumes and has an error rate of <5%. Assessment requires delineation of the FLR volume, tumour volume and total liver volume, which is time-consuming. Also, in patients with large tumours and tumours with indistinct borders, this method can be tedious and imprecise. Vauthey et al. developed the alternate method to calculate TELV based upon the patient’s body surface area (BSA): TELV = –794.41 + 1,267.28 × BSA. There is conflicting literature regarding the superiority of one method over the other. Ribero et al. observed that CT volumetry underestimated the risk of hepatic dysfunction in 11% of patients. In contrast, Leung et al. observed that measured volumes correlated better with outcomes than estimated volumes. Martel et al. in a recent study demonstrated poor concordance between the two methods, with a difference in FLR of more than 5% in one-third of the patients. Some groups have tried to develop formulas to estimate total liver volume without using body weight–related variables, as body weight is subjected to effects of ascites and edema. Although new formulas predicted total liver volume more accurately in certain patient groups, they lack multicentre validation. Instead of relying on liver volumetrics, some groups studied the utility of quantifying hepatic function, through the indocyanine green (ICG) clearance test. ICG is a nontoxic dye that binds to albumin and distributes in the blood pool shortly after injection. ICG is cleared exclusively by biliary excretion. Around 97% of the dye is cleared within 20 minutes of injection. ICG-R15 test is used for assessment before PVE. The test value describes the percentage of ICG remaining in the blood at 15 minutes after injection. Higher ICG-R15 values indicate lesser hepatic functional reserve and hence the need for larger FLR. A value <10% is indicative of a good hepatic function. For patients with a normal liver, PVE is indicated if sFLR is <20%. Several studies have shown that hepatosteatosis, hepatotoxic chemotherapy drugs and diabetes increase postoperative complications. These groups of patients demonstrate reduced and delayed FLR hypertrophy. In these subsets of patients PVE is indicated if sFLR <30%. In cirrhotic patients who are fit for surgery, PVE is indicated if sFLR <40% (ICCG 15 is between 10% and 20%) and <50% (ICCG 15 is 20%). The complexity of planned surgery should be considered before planning PVE. For surgeries requiring a larger extent of the resection (e.g. hepatectomy with pancreaticoduodenectomy), a larger hepatic reserve may be required to reduce postoperative complications. The main aim of PVE is to achieve complete portal blood flow occlusion of targeted segments. Embolizing all portal branches in the diseased segments is important to avoid porto-portal shunts, maximise hypertrophy of FLR and prevent hypertrophy of segments planned for resection. A majority of the procedures are right lobe PVE right lobe portal vein embolization (RPVE) before right hepatectomy and right lobe plus segment 4 portal vein (PV) branch embolization before extended right hepatectomy. Left lobe PVE is rarely done because the right lobe is larger and is sufficient to support the function post left hepatectomy. Volumetric studies of the liver have shown that after extended left hepatectomy (segment 2, 3, 4 with extension to caudate lobe), sFLR is around 33% and after extended right hepatectomy, sFLR is <20%. PVE can be done by following approaches: Transileocolic PVE is a surgical approach done under general anesthesia. Through right lower quadrant incision, the ileocolic vein is exposed and a vascular sheath is secured into the vein. Then the catheter is advanced under fluoroscopic guidance into portal vein branches and subsequently, embolization is performed. The main advantage of this approach is that it avoids direct puncture of the liver which is helpful in the patients with large tumour burden. However, this is an invasive procedure requiring general anaesthesia. For RPVE, percutaneous transhepatic access to RPV branches can be via an ipsilateral approach (through the tumour-bearing liver) or contralateral approach (through the FLR). Initially, the peripheral portal vein branch is accessed under ultrasound guidance and the vascular sheath is secured. Flush portography is performed through the sheath to study PV anatomy. Then the target PV branches are accessed using various catheter and wire combinations and embolization performed using an appropriate embolizing agent.
9.28: Portal vein embolization: Principle, technique and current status
Introduction
Definition
Principle
Indications
Contraindications
Absolute contraindications
Relative contraindications
Planning for PVE
FLR measurement and threshold determinants
Technical considerations
Technique
Transileocolic approach
Transhepatic approach
Ipsilateral approach (Figs. 9.28.1, 9.28.2 and Fig. 9.28.3)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








