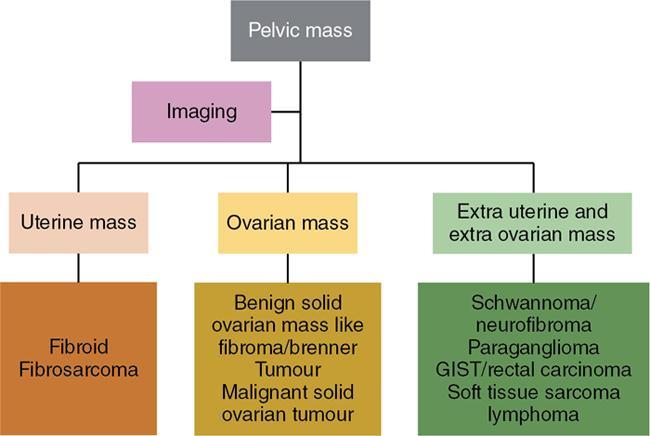
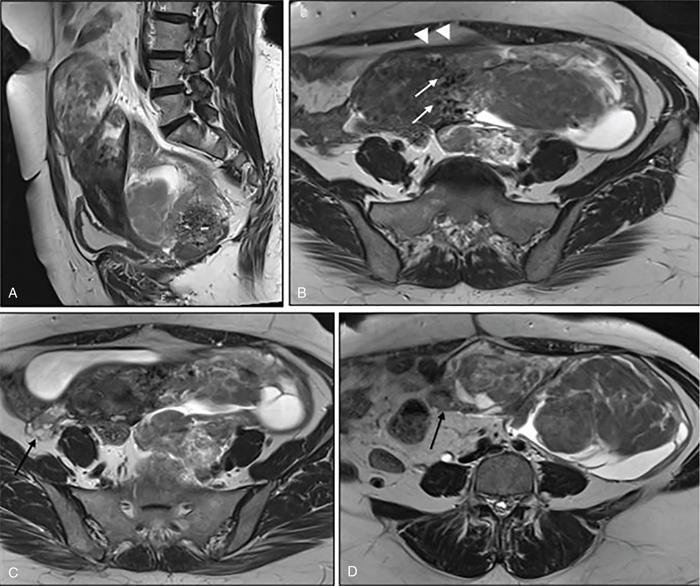
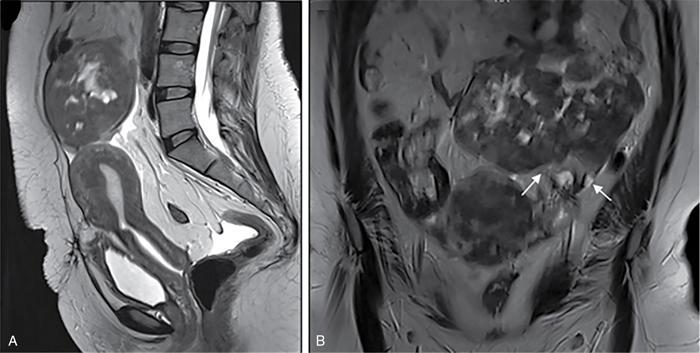
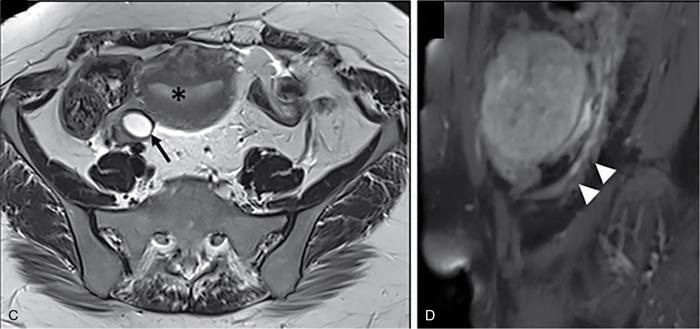
Suriyaprakash Nagarajan, Rupa Renganathan, Suhasini Balasubramaniam, Anupama Chandrasekharan, Kumarsampath Sumeena OVARIAN CYST VERSUS BLADDER Cystic lesions are common in female pelvis and mostly they originate from ovary. Ovarian cysts can arise from epithelial, stromal or germ cell components and can be benign or malignant cystic lesions. Pelvic Ultrasonography (USG) is the preferred imaging modality for evaluation of adnexal cystic lesions. Large simple ovarian cyst appears unilocular, anechoic with thin smooth walls. Such large simple cysts are confused with distended bladder. Distended bladder is a condition in which the urinary bladder is full and the patient is unable to void completely causing abdominal discomfort and pain. Distended bladder can be due to obstruction to passage of urine, neurological disorder or spastic sphincter. Differential diagnosis of cystic lesions in pelvis can be intraperitoneal, extraperitoneal and both intraperitoneal and extraperitoneal. Cystic lesions of intraperitoneal origin includes peritoneal inclusion cyst, paraovarian cyst, mucocele of appendix and hydrosalpinx. It is important to differentiate large simple ovarian cyst and distended bladder when patient presents with lower abdomen pain and the patient’s clinical history helps to make an accurate diagnosis (Table 11.4.1.1). UTERINE VERSUS EXTRAUTERINE MASS Ultrasound is usually the initial imaging modality for the pelvic mass. When USG findings are indeterminate to ascertain the organ of origin and to characterize, the next imaging modality is MRI due to its superior soft tissue resolution and multiplanar imaging capability (Figs. 11.4.2.1 and 11.4.2.2). Signs are demonstrated in Figs. 11.4.2.3 to 11.4.2.6. The sequences used in MRI pelvis are conventional T2WI in all three planes and T1WI in single plane. The problem-solving sequences are oblique coronal and oblique axial T2 along and perpendicular to the long axis of uterus in sagittal localizer. It can identify the organ of origin. Other sequences like T1 fat sat, diffusion-weighted imaging (DWI) and dynamic contrast T1WI with fat sat and subtracted images help to characterize the lesion further. BLADDER MASS VERSUS PROSTATE MASS Irregular mass lesions in the bladder neck are termed as ‘Bladder occupying lesions’. It is often difficult to determine whether the origin of these lesions is the bladder or the prostate gland. Transabdominal Ultrasound with colour Doppler studies and CT imaging do not usually delineate the origin of the lesions. The following features may aid in the differentiation (Table 11.4.3.1): History Painless haematuria Usually asymptomatic Investigations TAS, TRUS, MRI, Cystoscopy-guided biopsy PSA, DRE, TRUS, MRI, TRUS-guided biopsy At what point is the cross over between the two lesions T4 bladder cancer invading the prostate Aggressive prostate cancer protruding into the bladder Epicenter of the lesion Within the bladder Within the prostate gland Central necrosis in the mass Not commonly seen Seen in sarcomas IVU/CT urography Additional lesions in the urinary tract suggest that the bladder occupying mass in the bladder neck is of bladder-origin Prostate masses do not commonly cause masses in the urinary tract except for the very rare metastasis Virtual cystoscopy Additional lesions in the bladder wall and dome suggest that the bladder occupying mass in the bladder neck is of bladder-origin MRI T2 hypointensity Bladder masses are usually T2 hyperintense Seen in adenocarcinomas MRI T2 hyperintensity Bladder masses are usually T2 hyperintense Urothelial tumours infiltrating the prostate, neuroendocrine masses MRI T1 and T2 hyperintensity Bladder masses are usually T1 hypointense and T2 hyperintense Mucinous adenocarcinoma MRS Elevated choline values are seen in highly cellular masses Elevated choline:citrate ratio in prostate malignancies No significant finding in mucinous adenocarcinoma DWI Diffusion restriction is seen in malignant bladder masses No restriction in mucinous adenocarcinoma BPH versus bladder mass Exophytic BPH has signal intensities and appearance similar to and is contiguous with BPH within the gland OVARIAN MASS VERSUS PARAOVARIAN MASS Ultrasonography (USG) is the primary imaging modality in patients presenting with pelvic symptoms. Transvaginal, transabdominal or both should be performed in evaluation of such patients to differentiate ovarian and nonovarian origin of the lesions. Ovarian lesions can be a simple ovarian cyst, complex cyst with septations and solid components or a solid mass. Paraovarian lesions are remnants of the Wolffian duct in the mesosalpinx along fallopian tube or the ovaries and do not arise from the ovary. Paraovarian cysts are classified based on their site of origin into paratubal mesosalpingeal cysts, hydatid cysts of Morgagni, paraovarian cystadenoma and subserosal cysts. Differentiation of ovarian and paraovarian lesion poses significant diagnostic challenge. Both Computed tomography (CT) and Magnetic resonance imaging (MRI) are essential problem-solving tool in determining the site of origin of a pelvic mass. The first step in pelvic mass evaluation is to find out if it is ovarian or nonovarian in origin. Characterization of paraovarian or ovarian lesions is of utmost importance in order to optimize therapeutic procedures and it influences patients management (Figs. 11.4.4.1 and 11.4.4.2). Findings such as ovarian vascular pedicle sign, claw sign, bird beak sign and visualization of normal ovary helps to differentiate between ovarian and paraovarian lesions. Subsequently ovarian lesions should be categorized into benign, indeterminate and malignant masses (Table 11.4.4.1).
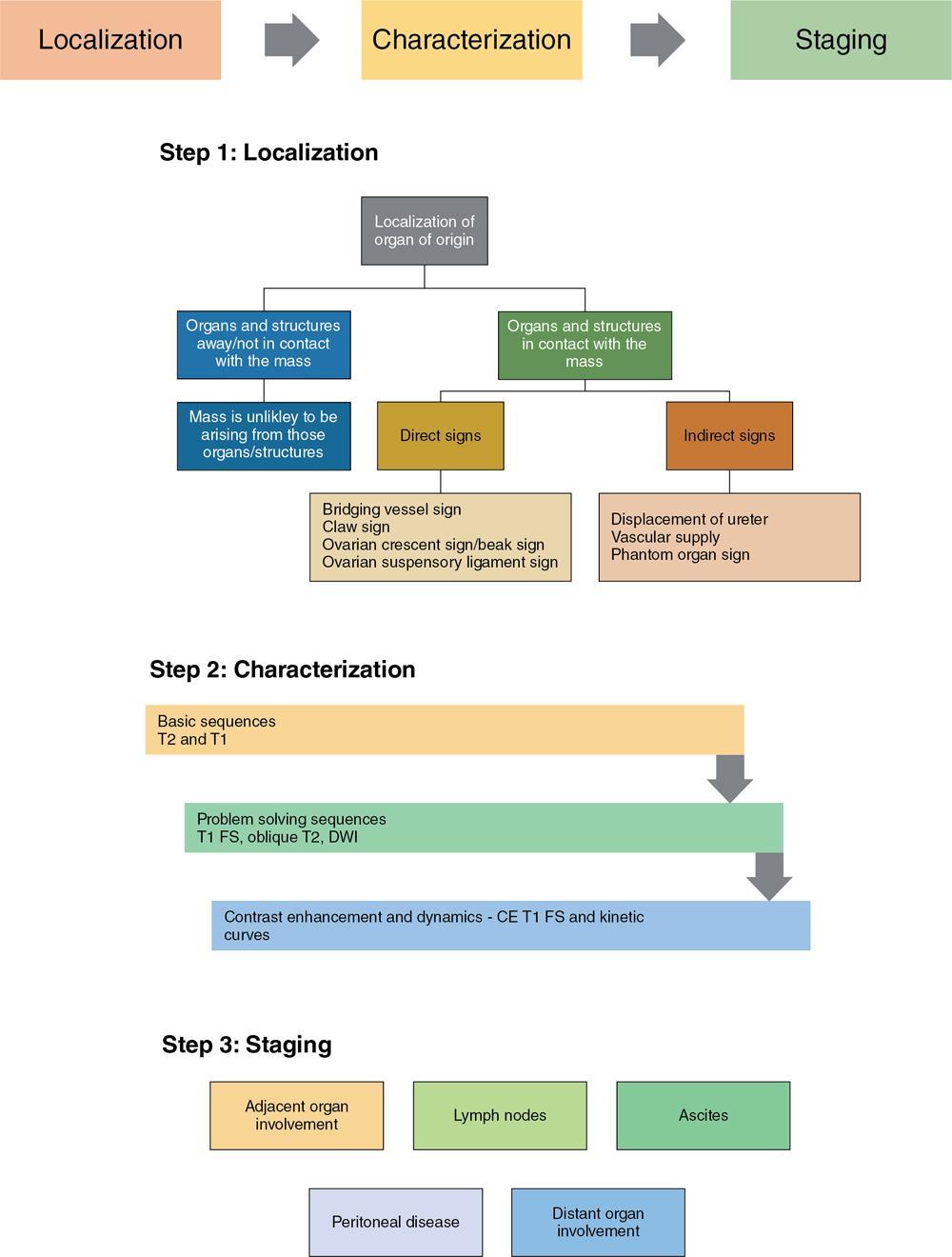
11.4: Radiologic approach to lesions (a systematic approach to clinical scenarios/radiological abnormality)
Ovarian Cyst
Distended Bladder
Ovarian cyst in plain radiograph is noted on one side of the pelvis. Areas of calcification can be demonstrated.
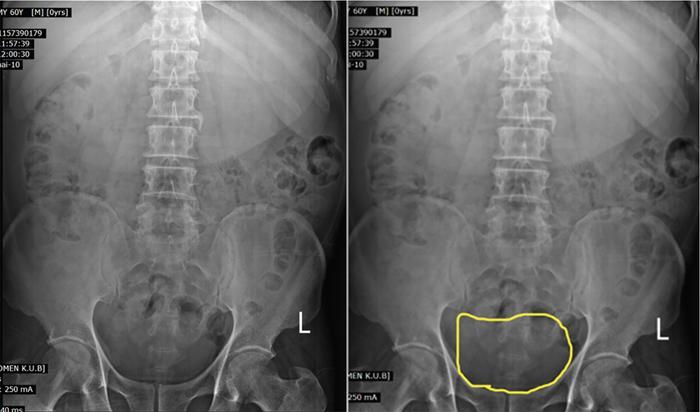
Plain radiograph (Fig. 11.4.1.1) shows a midline opacity arising from the pelvis extending above the pubic symphysis which gets relieved on catherization.
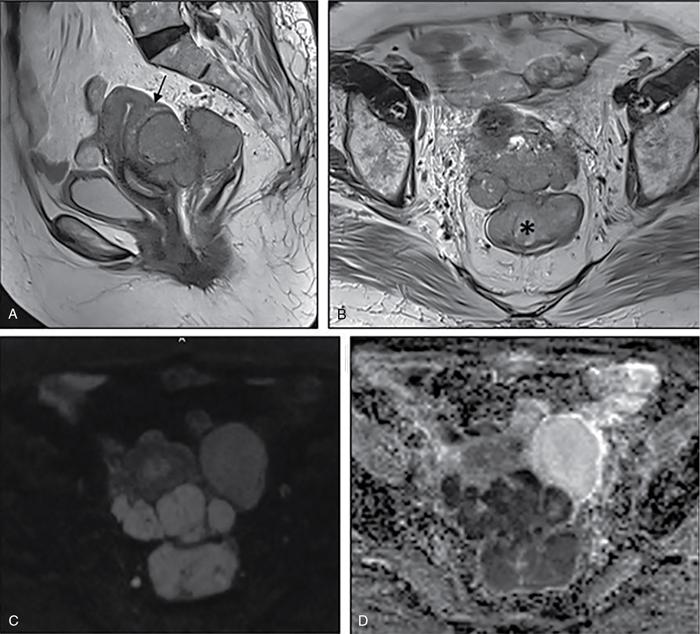
Ipsilateral ovary not seen separately from the lesion (Phantom sign) or the cyst is seen to arise from within the ovary (embedded organ sign).
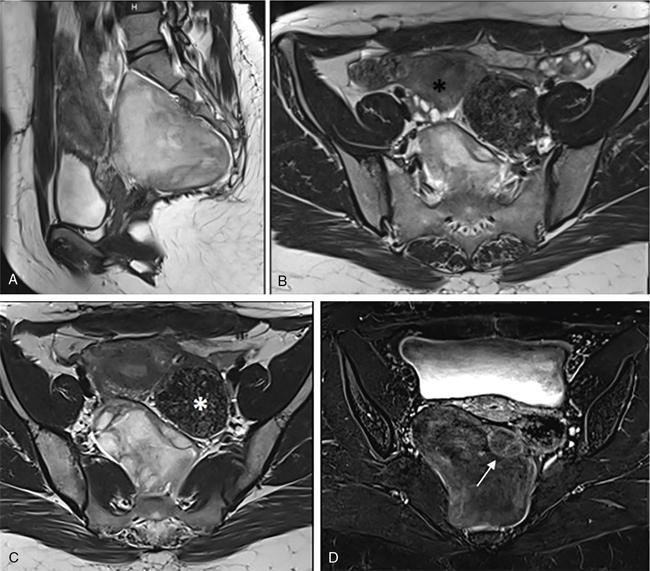
Ovaries can be demonstrated separate from the distended bladder.
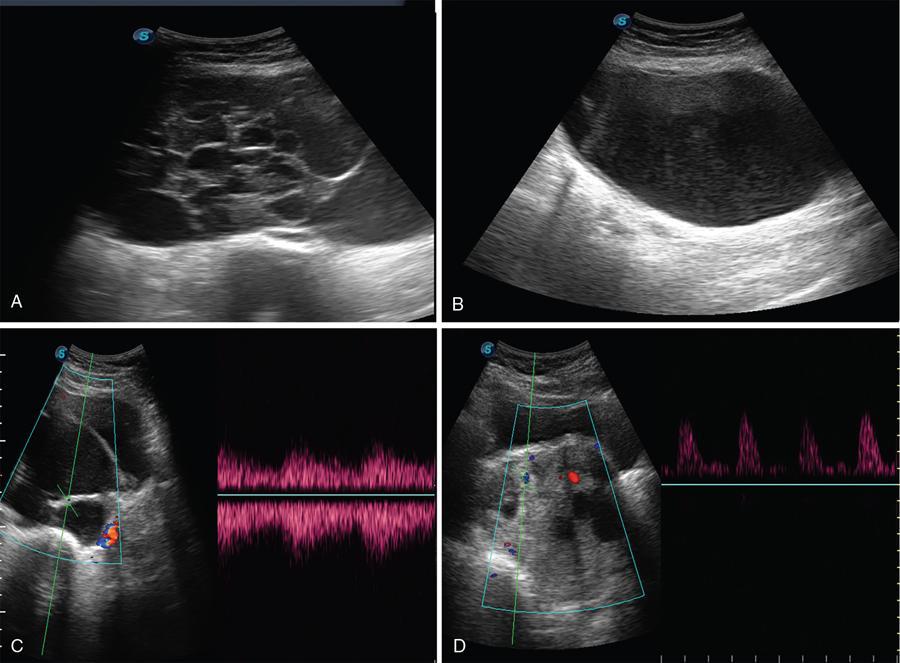
Complete or incomplete septae often seen (Fig. 11.4.1.2A).
Septations are not seen in distended bladder. In long-standing bladder distension there is significant amount of debris, moving with the movement of the patient.
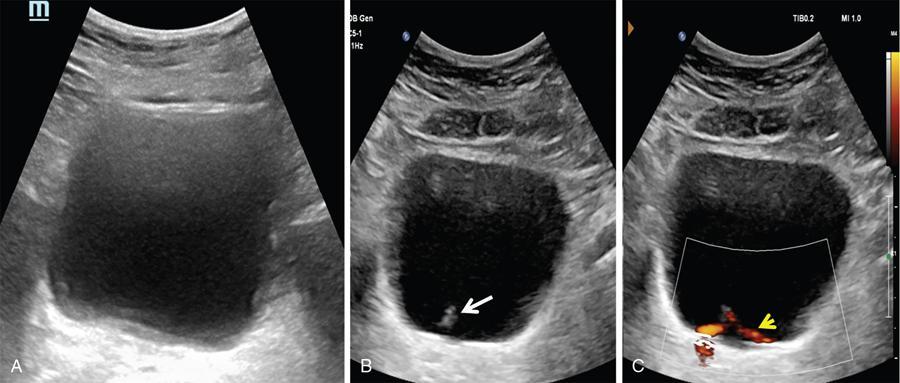
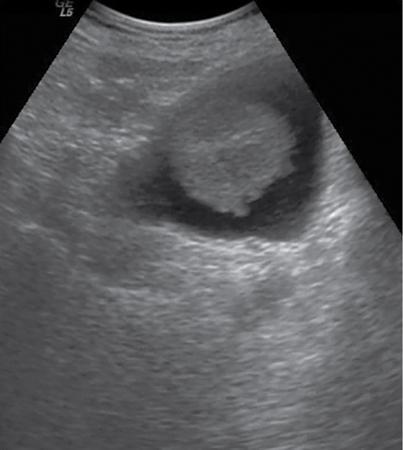
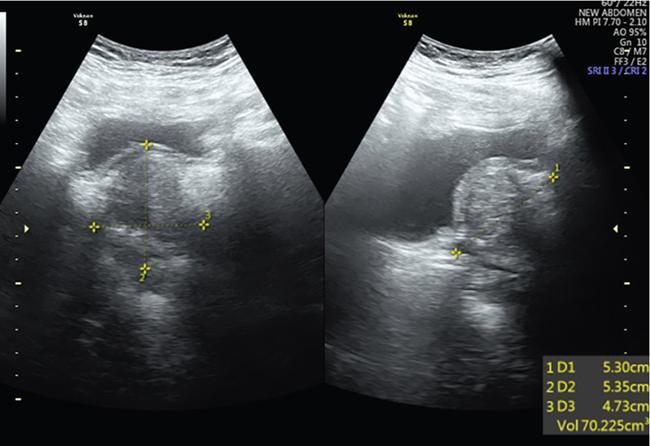
Ovarian cysts are anechoic lesions noted on one side of pelvis (Fig. 11.4.1.2B). Normal ovarian stroma can be seen around the cysts in case of small cysts.
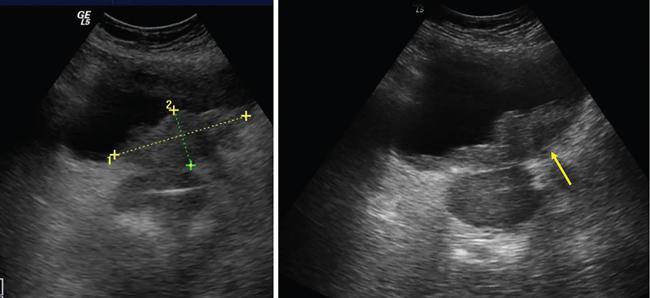
Bladder is visualized as a round or oval shaped anechoic fluid filled structure in the midline of pelvis (Fig. 11.4.1.3A).
Colour Doppler shows vascularity in the septum (Fig. 11.4.1.2C).
Ultrasonography shows distal ureteral jet. Doppler imaging shows the stream of urine entering the bladder through the ureteral orifice (Fig. 11.4.1.3B).
No evidence of trabeculations in cyst wall.
Distension of bladder due to neurological conditions shows trabeculation in bladder wall.
Solid components more commonly seen (Fig. 11.4.1.2D).
Solid components not usually seen in distended bladder.
Imaging approach
S. No.
Feature
Bladder Neck Mass
Prostate Mass
1
2
2
3
4
5
7
8
9
10
11
12
13
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








