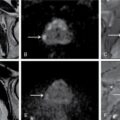
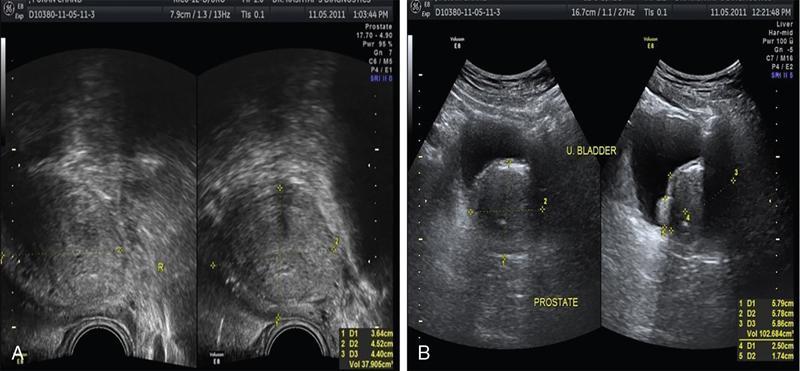
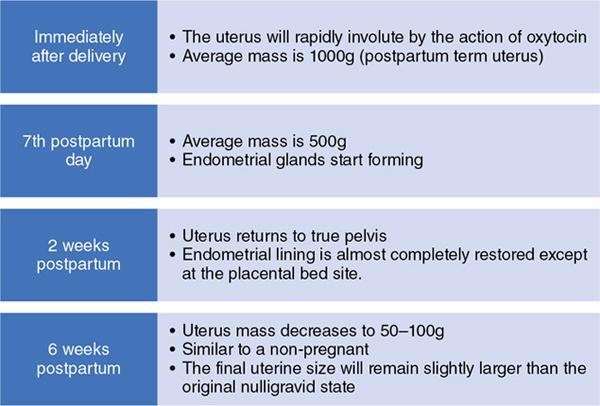
ULTRASOUND MALE PELVIS Vivek Kashyap Pelvic ultrasound is a noninvasive technique for imaging major organs in the pelvis including the bladder, prostate gland, blood vessels and bowel. It is usually the first line of investigation to evaluate the prostate and seminal vesicles. Ultrasound is a safe and reliable imaging resource to assess the pathology and guide towards appropriate treatment plan. To start pelvic ultrasound with evaluation of the shape and size of the bladder. The bladder is in the pelvis, posterior to the pubic bones. The peritoneum is seen superior to the urinary bladder and reflecting over the anterior surface. The trigone is a triangular area bounded by the two ureteric and urethral orifices. The urinary bladder has an outer loose connective tissue layer, known as subserosa which bounds the three muscular layers. The innermost is the mucosal layer. These layers should be smooth and uniform and the thickness will depend on the bladder distension. Prostate can be seen posterior to the urinary bladder when examined in transverse views. Prostate is seen at the inferior part of the urinary bladder in sagittal view (Fig. 11.1.1.1A). It should be examined in both the views. Urinary bladder neoplasms are usually seen arising from the walls of the urinary bladder, though these can be of varying shapes, usually are polypoidal and can present as solitary or multiple lesions, often seen on opposing walls of the urinary bladder, also known as ‘kissing lesions’ (Fig. 11.1.1.1B). For evaluating the bladder, it should be moderately filled. An overfilled bladder will cause patient discomfort and can compress structures posterior to it. The bladder should be scanned in the transverse and sagittal planes and prevoid volume is calculated. After micturition, the length, width and height (cm) of the bladder are recorded in sagittal and horizontal planes. The postvoid residual volume is calculated using the formula: Postvoid residual volume (PVR) = Length × Width × Height × 0.5 Normally there is no residual urine. PVR less than 50 is usually considered as insignificant. PVR volume more than 100 mL is considered significant in adults. Ureteric jets are the normal visualization of the physiological periodic efflux of urine from the distal ureter into the bladder. Ureteric jet can be appreciated by colour Doppler examination as a sudden burst of colour in the bladder that lasts for a few seconds. Bladder wall thickening is considered significant if >3 mm in a distended bladder and >5 mm in a collapsed bladder; however, it’s a good practice to fill the bladder completely before doing the investigation (Fig. 11.1.1.2). The common causes of bladder wall thickening include: Urinary bladder diverticula are examined very well on ultrasound (Fig. 11.1.1.2). Congenital diverticula, also known as Hutch diverticula are generally seen near the ureteral orifices. Acquired diverticula are generally due to bladder outlet obstruction. Bladder mucosa herniates through weak portions of the bladder walls, this may create diverticula with a narrow neck leading to urinary stasis, which may in turn cause stones, infection tumours or ureteric obstruction. These diverticula appear as an outpouching sac from the bladder and urine may be seen flowing into or out of the diverticulum. Intraluminal pathologies such as vesicle calculus are seen within the bladder, calcifications are seen in the bladder wall or in the mass lesions. Faeces, inflammatory or neoplastic bowel lesions, enlarged lymph node, masses from nerves, vessels and bones, ectopic or transplant kidney, accessory spleen can also be diagnosed from pelvic ultrasound. Ultrasound use for the male pelvis even though limited, has great importance to detect free abdominal fluid, undescended testes in the groin and hernias. Dilated urethra in posterior urethral valves, and occasionally a valve itself can be seen in real-time sonography, aided by transperineal scanning. Prevoid and postvoid volume Wall thickness Ureteric jets Size and volume Morphology of central and peripheral zone Morphology of seminal vesicles ULTRASONOGRAPHY INGUINOSCROTAL REGION V. Sivakumar Ultrasonography is the ideal noninvasive imaging modality for evaluation of scrotal abnormalities. It is capable of differentiating the most important aetiologies of acute scrotal pain and swelling, including epididymitis and testicular torsion, and is the imaging modality of choice in acute scrotal trauma. In patients presenting with palpable abnormality or scrotal swelling, ultrasonography can detect, locate and characterize both intratesticular and extratesticular masses and other abnormalities. A 12–17 MHz high-frequency linear array transducer provides excellent anatomic detail of the testicles and surrounding structures. In addition, vascular perfusion can be easily assessed using colour and spectral Doppler analysis. In most cases of scrotal disease, the combination of clinical history, physical examination and information obtained with ultrasonography is sufficient for diagnostic decision making. This review covers the normal scrotal anatomy as well as various testicular and scrotal lesions. Ultrasound is the widely used modality of choice for evaluation of scrotal abnormalities. Scrotal ultrasound is very helpful differentiating the most common causes of acute scrotal pain and swelling, including epididymoorchitis and torsion testis, and is the choice of investigation for acute scrotal trauma. Detection, localization and characterization of intratesticular and extratesticular pathologies are made easy using scrotal ultrasound. A 12–17 MHz high-frequency linear probe or transducer provides valuable anatomical information of testes and adjacent structures. Colour and spectral Doppler analysis can be used in assessing the vascular perfusion status of scrotal structures in real time. In most cases of scrotal disease, the combination of course, clinical history, clinical examination and imaging finding on ultrasound and Doppler is very essential in the diagnosis of scrotal diseases. Clinical examination sometimes leads to nonspecific signs and symptoms such as vague scrotal pain, suspicious scrotal mass or swelling. In such situations, ultrasound is useful in diagnosing and characterizing intratesticular and extratesticular pathologies as well as distinguishing benign or malignant lesions as well as identifying conditions requiring emergency surgical attention. Scrotum is a fibromuscular sac like structure divided into two hemiscrotum, right and left by a midline septum or raphe. Both the testes are located within the scrotum. The normal testicle appears like a ovoid structure with a homogeneous intermediate echogenicity and measuring approximately ~ 5 × 3 × 2 cm (craniocaudal, anteroposterior, width) (Fig. 11.1.2.1) Epididymis is elongated comma-shaped structure abutting posterior margin of testes and divided into three parts, head, body and tail. Epididymal head is seen in close relation to superior pole of testis and appears iso- to hyperechoic to the adjacent testicular parenchyma (Fig. 11.1.2.2). Epididymal body and tail can be visualized posterior to and along inferior pole of the testes and appears isoechoic to testicular parenchyma. A separate fibrous capsule called the tunica albuginea in each of the hemiscrotum encloses the testes and epididymis. This fibrous capsule can be noticed as a thin echogenic band like structure surrounding the testis on ultrasound. Tunica albuginea is covered by the tunica vaginalis, which is a remnant of processus vaginalis. It represents a closed sac of peritoneum and has two layers. The visceral layer of tunica vaginalis is seen lining the tunica albuginea of testis and is separated from the parietal layer that lines the inner aspect of the scrotal wall by minimal fluid-containing interspace. The tunica albuginea extends into the posteromedial aspect of testis to form the mediastinum testis (Fig. 11.1.2.3). It contains ducts, nerves and blood vessels. On ultrasound mediastinum is noticed as a thin echogenic band like structure. There are numerous fibrous septa extending from mediastinum testis dividing the testicle into multiple small lobules. The lobules are made of seminiferous tubules, they converge to form the tubuli recti, which are the testicular ducts that open into the rete testis. In around 20% of population, the rete testis can be visualized as a hypoechoic area adjoining the mediastinum testis. Efferent ductules coming out of rete testis can be seen draining into the epididymal head. From epididymal head, the ducts converge into a single duct in the epididymal body and exit the epididymal tail as the vas deferens. The vas deferens runs along the contents of spermatic cord and courses superiorly within the inguinal canal. The spermatic cord contains nerves, testicular artery, deferential artery, cremasteric artery, lymphatic ducts and the pampiniform venous plexus. On ultrasound the spermatic cord appears as an echogenic band like structure within the inguinal canal. The major vascular supply to the testes is by the paired testicular arteries which arise from the abdominal aorta. The testicular artery after entering into the tunica albuginea branches into capsular arteries and recurrent rami branches which then courses centrifugally into the mediastinum. A collateral circulation comprising of deferential artery and the cremasteric artery is also seen. Arterial supply to the remaining parts of the scrotum is from the pudendal arteries, which arise from the internal iliac artery. Anatomic relationships of the vascular structures within the spermatic cord are important and alteration in this normal relationship is associated with few pathologies. High insertion of tunica vaginalis to the spermatic cord, the bell-and-clapper deformity leaves the testes to rotate freely within the tunica and can lead to intravaginal testicular torsion. The pampiniform venous plexus continues as the testicular vein in the deep inguinal canal. The right testicular vein drains directly into the inferior vena cava (IVC) whereas the left testicular vein drains into the IVC via the left renal vein. Epididymal and testicular appendages usually represent embryologic remnants. They are better visualized in ultrasound in the presence of a hydrocele. The appendix of testis (hydatid of Morgagni) can be visualized as an isoechoic, oval structure between the epididymis and testis (Fig. 11.1.2.4). An appendix of epididymis is a pedunculated structure arising from the epididymal head, which is also isoechoic to testicular tissue. Ultrasound of the scrotum includes both greyscale and colour Doppler evaluation of testicles, epididymis on both sides and the scrotum. Spectral Doppler evaluation can be used to demonstrate normal waveforms in the testicular artery and vein. For scanning a high frequency, broad bandwidth 17–5 MHz or 12–5 MHz linear array transducer is used. With the help of a towel placed under the scrotum the testicles and scrotum are elevated. With another towel the patient retracts and covers his penis. Initially with a side-to-side large-field imaging both testes are demonstrated using both greyscale and Doppler techniques. Echogenicity and perfusion of each testis is examined. Each testis is imaged longitudinally in medial, mid, lateral portions and transversely in upper, mid and lower pole regions. Three-dimensional (3D) measurement of each testicle is taken (length, width and height). Epididymis evaluation includes longitudinal and transverse imaging including head, body and tail. Colour Doppler study of testes and epididymis is done on both sides and status of capsular and centripetal arteries assessed and documented. Spectral waveform of both arterial and venous flow is documented on each side. Optimization of colour and spectral Doppler analysis is done to clearly depict low velocity venous flow. Colour Doppler imaging in various planes and comparison with contralateral asymptomatic testis is done in specific evaluation for testicular torsion. Scrotal survey should be done for extratesticular findings such as hydrocele, varicocele or calcification/scrotolith. The pampiniform plexus of veins should be evaluated for the diameter and its augmentation on standing position or Valsalva manoeuvre. Scrotal wall should be assessed for thickening and oedema. Extratesticular and intratesticular pathologies, either focal or diffuse should be identified and characterized using grey scale and Doppler imaging techniques appropriately. Focal lesions should also be measured three dimensionally. Presence of solid, cystic, calcific, complex cystic areas in the lesion should be characterized. Labelling of the lesion/images in respect to location (upper, middle or lower), orientation (longitudinal and transverse) and position of the patient (supine, decubitus and standing) is done. Labelling of the lesion/images during specific manoeuvres such as Valsalva should also be done. For scanning a high frequency, broad bandwidth 17–5 MHz or 12–5 MHz linear array transducer is used. Initially with a side-to-side large-field imaging both testes are demonstrated using both greyscale and Doppler techniques. Echogenicity and perfusion of each testis and epididymis should be examined. Each testis is imaged longitudinally in medial, mid, lateral portions and transversely in upper, mid and lower pole regions. Epididymis evaluation includes longitudinal and transverse imaging including head, body and tail. Spectral waveform of both arterial and venous flow is documented on each side. Ultrasound and colour Doppler are the first-line imaging modality for acute or chronic scrotal pathologies. Ultrasound is a safe and reliable imaging modality for demonstration of scrotal anatomy, localization of testicular pathologies and for assessing the vascularity. Since most of the testicular pathologies have characteristic ultrasound appearance, ultrasonography is able to clearly guide patient management and prevent unnecessary surgery. TRANSRECTAL ULTRASOUND OF PROSTATE Vivek Kashyap Carcinoma prostate is the second most common cancer in the world and the sixth leading cause of cancer death in men. The worldwide burden of prostatic cancer is estimated to touch 1.7 million new cases and 499,000 deaths by 2030. The prostate is an unpaired glandular organ. The average dimensions in a healthy young adult is 3.75–4.00 × 2.5–3.00 × 3.1–3.8 cm (w × h × l) and the volume ranges from 20–25 cm3. It produces the prostate-specific antigen (PSA) a glycoprotein which is an ingredient of the semen and is responsible for liquefying semen. The prostate has endocrine properties as it produces prostaglandins E, F and A, spermidine and spermine. It is also the place of the conversion of testosterone into dihydrotestosterone under the influence of the 5-α-reductase enzyme. COMMON INDICATIONS FOR A TRANSRECTAL ULTRASOUND (TRUS) OF PROSTATE Assessment of prostate size Abnormal growth within the prostate Male infertility Elevated PSA levels Detecting disorders within the prostate Micturating difficulties Because ultrasound provides real-time images, it can be used to guide biopsies which may be necessary, FNAC or tissue biopsy as well as brachytherapy seed placements. Transrectal US is a widely used clinical imaging method. For confirming the diagnosis of prostatic cancer, a systematic needle biopsy with US guidance is the diagnostic test of choice (Fig. 11.1.3.1). To understand the images produced by transrectal US, it is important to understand the zonal anatomy of the prostate gland. The outer zone is the peripheral zone, inner to it the transitional zone, then central zone and periurethral zone. The peripheral zone is the largest, containing almost 70% of the prostatic tissue and is the site of most prostatic cancers. It is separated from inner zones by a surgical capsule which may be seen as hyperechogenic line The transitional zone is much smaller (about 5%) and is seen as two small glandular areas such as saddle bags adjacent to proximal urethral segment. The central zone constitutes almost 25%–30% of the total gland size. The ducts of vas deferens and seminal vesicles pass through this zone. This zone shows only about 5% incidence of cancers and appears relatively resistant to diseases. The periurethral zone is only about 1% and encircles the urethra. The commonest features of benign prostatic hyperplasia are hypoechoic enlargement of inner zone. Other features could be calcifications along with diffuse or well-circumscribed rounded hypoechoic or hyperechoic nodules along with degenerative or retention cysts (Fig. 11.1.3.2A,B). Well-circumscribed hypoechoic nodules arising in the transitional zone may become so large that they distort the gland and appear to be in the peripheral zone and these are invariably benign. In cases of hyperplastic nodules occurring in the peripheral zone, they will appear as firm to hard nodules and should undergo a biopsy. We must remember that prostatic enlargement may not correlate with urinary symptoms in all cases, therefore any urinary dysfunction calls for evaluation of the prostate, bladder, urethra, spine and CNS. Transvesicle ultrasound is done to evaluate prostate size, median lobe, bladder trabeculations, diverticula, tumours or calculi. Patients who have had TURP done before will invariably show a defect in the gland. However, the size of the defect will not always correlate with the amount of tissue removed during the procedure, nor does it correlate well with the success of the procedure. This is a condition of inflammation of the prostate and seminal vesicles. The prostate may appear normal in prostatitis or there may be a focal mass, ejaculatory duct calcification, irregularity or thickening of the capsule, periurethral glandular irregularity, dilated periprostatic veins and distended seminal vesicles. Chronic granulomatous prostatitis can present as diffuse large or small hypoechoic zones or a solitary hypoechoic lesion. The examination is generally painful and colour Doppler may show a very vascular focus (Fig. 11.1.3.3A). If we see a hypoechoic mass within the prostate with or without internal echoes may be an abscess. Common ones are degenerative or retention cysts in hyperplastic nodules. These are generally symptomless, unless they become large (Fig. 11.1.3.3B). Prostatic utricle cysts are in the midline and are caused by dilatation of prostatic utricle. These can be associated with unilateral renal agenesis and rarely contain spermatozoa. Other type of cysts seen are Mullerian duct cyst which are a little lateral and are also tear drop and do not contain spermatozoa. Ejaculatory duct cysts are small and could be a small diverticulum of the ejaculatory duct or an obstruction. These can be fusiform in shape with pointed ends and contain spermatozoa. These are associated with infertility. Seminal vesicle cysts are another type, which could be large and solitary, associated with ipsilateral renal agenesis sometimes. In cases of male infertility, transrectal sonography has an important role in assessment of causes of azoospermia for ejaculatory duct obstruction or anomalies (about 1%–2%) Seminal vesicles, dilated ejaculatory ducts and midline cysts can suggest obstruction. There is a strong association of absence of vas with cystic fibrosis. The results of transrectal scanning in infertile men with azoospermia can be normal appearance (25%), bilateral absence of vas deferens (34%), bilateral occlusion of the vas, seminal vesicles or ejaculatory ducts by calcification or fibrosis (16%), unilateral absence of vas (11%), obstructing cysts (9%), ductal obstruction due to calculi (4%). Some of these causes, more so in distal two-thirds could be surgically correctable and therefore it is essential that careful evaluation is done. Haematospermia is blood in the semen. This could be acute (red in colour) or dark brown (old blood). Nonspecific inflammation of the prostate or seminal structures is the most common cause, resolving on its own after some time. We should exclude tumours or infection of prostate or bladder. Transrectal scanning can detect calculi or cysts in the seminal vesicles or ejaculatory ducts (Fig. 11.1.3.4). Other causes includes vascular malformation or sometimes following a prostatic biopsy. Ultrasound elastography is an imaging technique for mapping tissue stiffness of the prostate gland. Two elastography techniques that have developed are strain technique, and second, shear wave technique. The tissue stiffness information provided by US elastography can improve the detection rate of prostate malignancy and provide guidance for biopsy. Prostate elastography has high sensitivity for detecting prostate malignancy and shows high negative predictive values, ensuring that few malignant lesions will be missed. Transrectal US has been used for local staging of prostate cancer in some studies but is generally considered insufficient. Extracapsular extension (ECE) can be demonstrated by the bulging or irregularity of the capsule adjacent to a hypoechoic lesion. Seminal vesicle invasion (SVI) is diagnosed by demonstrating the extension of a lesion at the base of the prostate into a seminal vesicle. Solid hypoechoic lesions within the seminal vesicles or asymmetry of the seminal vesicles are indirect indicators of disease extension. Additional transrectal US-guided biopsies of the seminal vesicles can be performed when extra prostatic extension into the seminal vesicles is suspected. Studies performed in the 1980s showed when cancers are larger (stage T3) and more readily palpated, sensitivity for detecting ECE and SVI by transrectal US was more than 80%. With advent of colour duplex and power Doppler US, there has been renewed interest in the use of transrectal US to identify cancer in hyper vascular areas, with only modest improvements in sensitivity and specificity. Transrectal US continues to play an important role in therapy for directing brachytherapy seeds into the accurate location within the prostate. Cryotherapy and focal ablation of the prostate cancer also requires transrectal US guidance. Future developments in transrectal US may include the utilization of microbubble contrast agents and targeted imaging. Microbubbles coated with surface ligands that preferentially target tumour neovascularity can be used. Because of the large size of these bubbles (1 m), these agents can provide information about large-vessel microvascularity which can be useful in detecting cancer; however, if in future smaller-size microbubbles are available, they can further improve detection. Seminal vesicles have symmetric appearance and lie cephalic to the prostate and posterior to the bladder. They have a typical ‘bow-tie’ appearance in transverse scans, and a club or tennis-racket shape in longitudinal scan. They show homogenously fine echoes but less echogenic than prostate. Volume of seminal vesicles tends to shrink after the fifth decade. TRUS provides excellent anatomical details of seminal vesical and ejaculatory ducts. TRUS helps to diagnose calculi in seminal vesicle or ejaculatory duct. Also TRUS provides high-resolution imaging of the prostate, seminal vesicles and vas deferens and is the investigation modality of choice in diagnosing congenital and acquired abnormalities implicated in the cause of obstructive azoospermia. USG FEMALE PELVIS M. Alamelu Ultrasound is a noninvasive, easily available and informative investigation. A skilfully done and well-interpreted ultrasonogram image usually obviates the need to proceed to additional expensive and composite cross-sectional imaging tests. Ultrasound is high-frequency sound waves (20 kHz), inaudible to human that can be safely transmitted into human body to scan the tissues. Images produced in ultrasound depend on the mechanical properties of tissues and their distance from the source of the sound as it will differentially affect the energy of the sound waves (Table 11.1.4.1). As the frequency of the transducer scan-head increases, the resolution increases and penetration into the body tissue decreases. The piezoelectric crystals convert electric energy into mechanical energy and sound waves are produced. Pelvic pain Pelvic masses Dysmenorrhea (painful menses) Amenorrhea Abnormal uterine bleeding Follow-up of previously detected abnormality Infertility Pelvic infection Evaluation after pelvic surgery, delivery or abortion To localize intrauterine device (IUD) Screening for genital tract malignancy in high-risk patients Pelvic organ prolapse The transducer is placed longitudinally over the adequately distended bladder, the gain settings to be adjusted for optimal visualization (Fig. 11.1.4.1). Scanning is to begin longitudinally from midline between umbilicus and pubic symphysis, angulated to the sides, moved laterally to both right and left side (Fig. 11.1.4.2). Next, scan transversely beginning from pubic symphysis down to umbilicus upwards (Fig. 11.1.4.3). If needed, patient can be asked to turn obliquely (30–40 degrees) for evaluation of ovaries and adnexa (Fig. 11.1.4.4). The following should be assessed: The overall length of uterus is evaluated in the sagittal view from fundus to cervix (to the external os, if seen). The length can either be measured as a straight line from fundus to cervix by using the outer-to-outer technique or can be measured from fundus of uterus along the endometrial lining and endocervical canal using the outer-to-outer technique (Fig. 11.1.4.5). The anteroposterior measurement of the uterus is made in the sagittal view from its anterior to posterior uterine walls. The maximum width can be measured in the transverse or coronal plain. The cervical component should be excluded from uterine length measurement for volumetric assessment of uterine corpus. The neonatal uterus, with exposure of maternal hormones is well depicted with US. The uterus is more narrower and shorter, with a mean length of 3 cm in infants of 4–12 months. Cervix attains a tubular shape and is proportionate to the uterine body. After 1 year of age, uterine length gradually increases and rapid growth occurs as the girls approach puberty. The mean uterine length 3 cm or less for young girls, 3–4.5 cm for prepubertal girls and 5–8 cm for pubertal girls. During puberty, the uterine body becomes more thicker and rounded, pear shaped, with a ratio of uterine body-to-cervix – 1.5:1, an appearance similar to that in women of reproductive age (Table 11.1.4.2). Source: J.E. Langer, E.R. Oliver, A.S. Lev-Toaff, B.G. Coleman, Imaging of the Female Pelvis through the life cycle, Radiographics 32 (2012) 1575–1597. H.J. Paltiel, A. Phelps, US of the pediatric female pelvis, Radiology 270 (2014) 644–657. Depending on the extent of the urinary bladder distension the normal uterus assumes a number of positions. Depending on the relationship of the angle of the long axis of the uterine body to the long axis of the cervix (flexion) and the long axis of the uterus to the long axis of the vagina (version) various positions are described. The most common position for uterus is in anteversion and anteflexion (Fig. 11.1.4.6). When the uterus is positioned in retroversion or retroflexion, it will be difficult to image transabdominally in most cases and is imaged well with an endovaginal transducer. Following every delivery, the uterus of parous women becomes more thick and round. After caesarean delivery, the uterus typically appears more elongated with variable deformity or thinning at the site of the lower uterine incision. The postmenopausal uterus is atrophied and small. The reduction in uterine size is most rapid during the first decade after menopause. The range can vary between 3.5 and 6.5 cm in length and 1.2 and 1.8 cm in anteroposterior dimensions. The endometrium should be assessed for the thickness, presence of focal abnormalities, its echogenicity, and the characteristics of fluid or masses in the cavity. The endometrium has a variable echogenicity, depending on its water content and the cellular density, that varies with the hormonal status of the patient. Neonatal uterus – The endometrium appears as echogenic line and endometrial fluid is often seen, due to residual effect of maternal hormones. The endometrium becomes less obvious, a thin echogenic line seen centrally in only 50% of girls of prepubertal age. The postpubertal endometrium is easily visible with transabdominal US and varies in thickness with the phases of the menstrual cycle. The thickness of endometrium correlates with the histologic changes to the estrogenic stimulation. The endometrium has a trilaminar appearance in preovulatory phase of menstrual cycle, then thickness becomes homogeneous after ovulation. The thin central hyperechoic line is not an endometrial layer it represents the interface between the anterior and posterior endometrial aspects. It should demonstrate as a continuous line on imaging parallel to the long axis of the uterus. Disruption of the central echogenic line, or any heterogeneity of the endometrium, indicates an underlying intracavitary lesion such as polyp, myoma or adhesion (Fig. 11.1.4.7). Progressively increasing echogenicity of the functional zone (compactum and spongiosum) seen with completion of the preovulatory phase and during the secretory phase. The homogeneously echogenic endometrium of the secretory phase poses difficulty in visualization of the central interface. For this reason, assessment of the focal abnormalities in endometrium is best done in the early phase of the menstrual cycle. The thickest part of the endometrium to be measured perpendicular to its longitudinal plane in the anteroposterior diameter, using the outer-to-outer technique, from echogenic to echogenic borders. The endometrial thickness is correctly measured from the anterior endometrial–myometrial interface to the posterior endometrial–myometrial interface. The adjacent myometrium (hypoechoic) and fluid in the cavity should be excluded in the measurements. The junctional zone thickness can be up to 12 mm (Fig. 11.1.4.8). In the women taking oral contraceptive pills or using other hormonal contraceptives, such as progesterone-releasing intrauterine devices, the endometrium is generally thin throughout the entire menstrual cycle. If the patient has an IUD, its location should be documented. Sonohysterography is an effective adjunct to further clarify an abnormally thickened or an incompletely visualized endometrium. The normal myometrium has three layers. The innermost layer is thin, relatively hypovascular and hypoechoic and is referred as the subendometrial halo. The next is the intermediate layer. It is the thickest layer and has uniform echotexture. The outer layer is also thin and slightly hypoechoic. The outer and the intermediate layer is separated by the arcuate vessels. These arteries again branch into radial arteries which penetrate through the intermediate layer and reach the inner layer. In some women the arcuate vessels (the veins) can be prominent and mimic cystic changes. During the reproductive ages, the normal uterine myometrium has homogeneous echogenicity with smooth margins. The nongravid uterus can demonstrates myometrial contractions, directed from the cervix to the fundus in the periovulatory phase, presumably to promote sperm transport, have been shown to have higher rates of successful fertilization compared to women with uncoordinated or absent wave activity. Postpartum uterus – the arcuate arteries undergo calcification, which can be seen earlier in diabetic women. This is a part of normal aging process. At times, small hyperechogenic foci are seen in inner myometrium, a few millimetres in size and nonshadowng in appearance, which represent dystrophic calcifications and have no clinical significance. The myometrium and cervix should be analysed for contour changes, echogenicity, masses, cysts and the symmetry between its anterior and posterior segments. The size and location of any pathological lesions should be measured in at least two dimensions. The normal postpartum uterus: The appearance of cervix varies with age of female. The cervix can be visualized as cylindrical-shaped structure on sagittal views and round/oval on coronal view. An echogenic central line denotes the interface between the two mucosal layers. Endocervical canal is 2–4 mm in single-layer thickness and in the centre it has a small amount of anechoic fluid or mucus. The plicae palmatae appear as serrations in the mucosa due to compact folds. The cervical stroma usually has similar echogenicity to myometrium and may demonstrate a narrow hypoechoic submucosal zone. Colour Doppler US shows little or no vascularity in the cervix. US beam is swept through the entire length of the cervix, from the internal os through the external os and upper vagina and laterally through the cervix including the bilateral parametrial and other adjacent pelvic tissues. This provides better, delineation of the cervical borders and the extent of any lesions. In case of any abnormality in the cervix identified at US, use of transducer pressure is extremely beneficial to determine if the lesion is located in the endocervical canal is arising from the stroma or from the cavity, particularly in case of a pedunculated intracavitary myoma or pedunculated polyp. On application of transducer pressure over the cervix, the intracavitary lesion may show mobility if a lesion is from the cavity mucosa, whereas lesions arising from the cervical stroma will be fixed in position despite the transducer pressure. The neonatal cervix is disproportionately large and bulbous on imaging. With increasing ages, the cervix becomes proportionate to the uterine body, producing a tubular shape. The branches of the uterine arteries can be identified on both sides of the cervix at colour Doppler flow imaging. The vagina seen as a collapsed tubular structure, distal to the uterine cervix and inferior to the urinary bladder. Caution: overdistension of the urinary bladder may cause urine accumulation in the vagina, misinterpreted for vaginal cysts. The presence of tampons or menstrual blood should also be looked. The adnexa include the ovaries, fallopian tubes, supporting ligaments and peritoneal folds of the lateral pelvis and the blood vessels. The adnexal mass lesions to be analysed for its position, relationship to the ovaries and uterus, size, shape, sonographic characteristics. The ovaries are demonstrated by scanning in transverse or longitudinal planes. They are seen lateral to the uterine corpus. The position of the ovary varies with the length of the infundibulopelvic ligament, presence of adhesions and any other anatomic abnormalities that displaces the ovaries. In neonates, the ovaries generally are easily imaged transabdominally as they are located above the true pelvis. As the child grows, the ovaries are embedded deeper in the pelvis, adjacent to the uterus. Ovarian size is to be measured in three dimensions (longitudinal, transverse and anteroposterior diameters) on views obtained in two orthogonal planes for the calculation of ovarian volume. Normal ovarian volume: simplified formula for volume calculation is 0.5 × length × width × thickness (Table 11.1.4.3). Follicles in ovaries in women of reproductive age can be demonstrated as echo-sparse, well-circumscribed areas within the ovarian stroma with varying sizes between 5 and 20 mm. Follicles are simple cysts with size of 9 mm or less. Ovarian mimics: The addition of 3D ultrasound is used to differentiate ovarian multiseptated cysts from hydosalpinges. Fallopian tubes are tubular structures extending from posterosuperior aspect of the uterine fundus. They open into the peritoneal cavity. Bilateral tubes are located in the superior edge of the broad ligament. They are 10–12 cm in length and 1–4 mm in diameter. A fallopian tube has four anatomic segments. The interstitial or intramural segment is located at the uterine cornua, within the myometrium. The isthmic segment is the narrowest portion of the fallopian tube. The ampullary segment is closer to the ovary, and constitutes around one-half of its total length. The infundibulum is funnel-shaped lateral most segment. The free edge of fallopian tube is composed of around 25 finger-like projections, called fimbriae. The fimbriae are suspended over the ovary and capture the ovum after its release. The fallopian tube is difficult to visualize in its normal state in USG. Fallopian tubes are frequently visualized in pathological conditions such as hydrosalpinx or neoplasm, the tube may be more clearly defined. Transvaginal ultrasonography is a better imaging modality for tubal visualization. The urinary bladder usually is a landmark for anatomic orientation in transvaginal and transabdominal scanning. Overdistension of the bladder will displace the uterus too posteriorly to hinder adequate imaging and also the patient will experience gratuitous discomfort. Conversely, in case of insufficient bladder filling, the posterior uterine wall or fundal lesions can be missed. Caution: a full bladder mimics a unilocular, anechoic pelvis cyst. In doubt cases, a postvoid scan is to be suggested for definitive evaluation. The bilateral urethrovesical junction and the distal urethra can be visualized in an adequately filled bladder. The cul-de-sac is posterior to the uterus and is to be evaluated for the presence of free fluid or loculated fluid and any mass lesions. Minimal free fluid (usually less than 1 cm in thickness) in the cul-de-sac is considered an unremarkable finding in girls of prepubertal ages and women of reproductive age. Artefacts are echoes that are falsely imaged without a corresponding anatomical structure (Fig. 11.1.4.9).
11.1: Radiological techniques in reproductive imaging
Male pelvic ultrasound
Clinical indications
Delay in starting to pass urine
Difficulty in completely emptying the bladder
Symptoms of urgency
Identifying an enlarged prostate
Haematuria
Lymphadenopathy,
Presence of fluid/blood

Technique

FOCAL
DIFFUSE
Checklist for Male USG Pelvis
Bladder
Prostate
Introduction
Normal scrotal anatomy on ultrasound

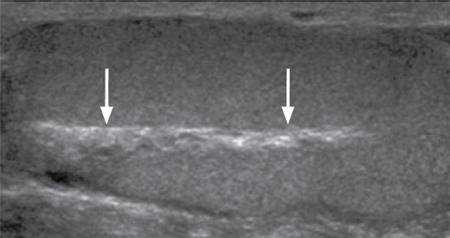
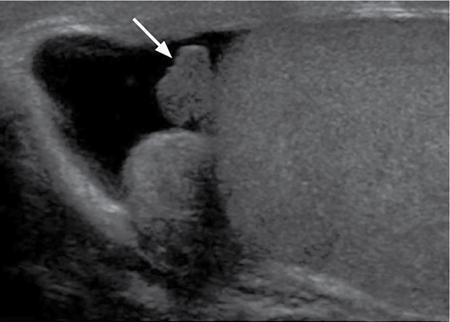
Technique and scanning protocol
Summary
Introduction

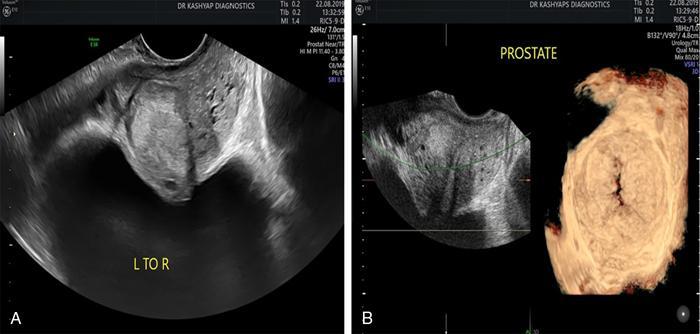
Benign prostatic hyperplasia
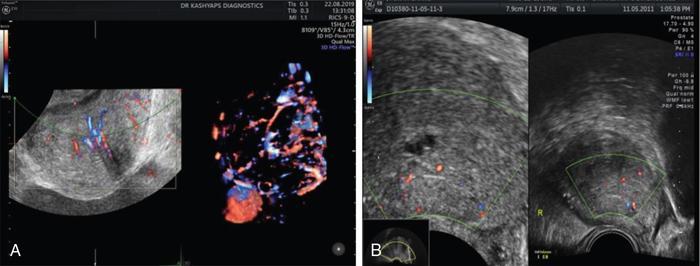
Prostatitis
Prostatic cysts
Prostate in infertility
Haematospermia
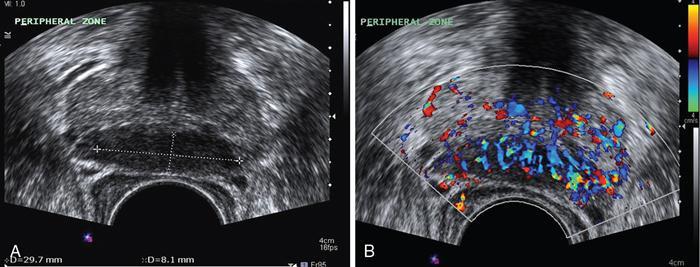
Prostate cancer

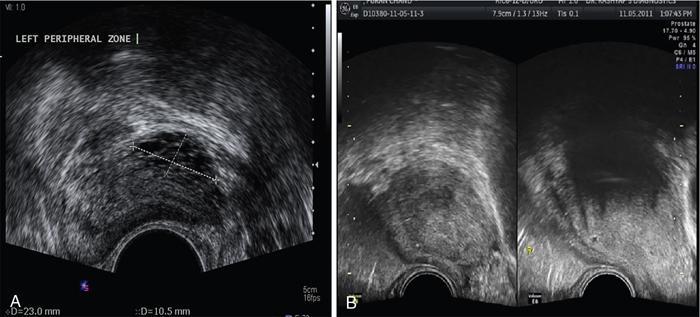

Role of elastography
TRUS for staging of prostate cancer
Introduction
Dense structures (Bone)
Strong reflectors
Appear as white
Hyperechoic
Soft tissue
Weak reflector
Grey
Isoechoic
Fluid
No reflector
Black
Hypoechoic/Anechoic
Indications of pelvic USG
Technique
Patient preparation
Scanning method
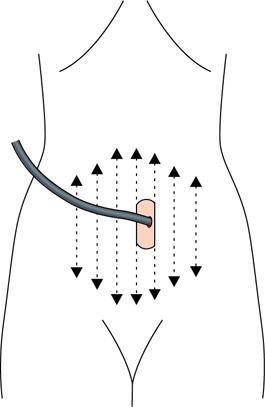

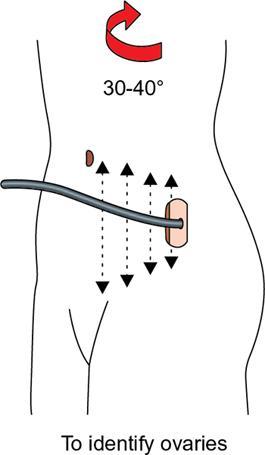

Uterus
Uterine size, shape and orientation

Stage
Uterine Length (cm)
Uterine Body to Cervix Ratio
Neonatal
3.5
2:1
Paediatric
1–3
1:1
Prepubertal
3–4.5
1–1.5:1
Pubertal
5–8
1.5–2:1
Reproductive
8–9
2:1
Postmenopausal
3.5–7.5
1–1.5:1

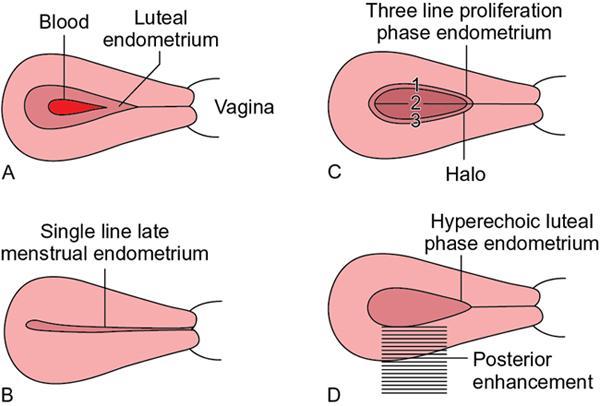
Endometrium

Myometrium
Cervix
Vagina
Adnexa
Ovary
Fallopian tube
Urinary bladder
Cul-de-sac
Artefacts
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree