Drushi Patel CONTRAST-ENHANCED ULTRASOUND IN ADULT GENITOURINARY IMAGING Mridula Muthe Ultrasound is the initial screening examination for the evaluation of the renal morphology and suspected renal lesions due to its multiple advantages such as easy accessibility, bedside assessment, affordability and lack of radiation. Vascularity of the kidney and renal lesions can be studied with the use of colour Doppler ultrasound however, it provides information only about the macrovasculature. With the use of microbubble ultrasound contrast agents (UCAs) macro as well as the microvasculature can be studied. Contrast-enhanced ultrasonography (CEUS) is also more sensitive than Doppler ultrasound as it is not affected by blood velocity and angle of insonation of the ultrasound beam. In urology, the established applications of CEUS in adults include characterization of cystic lesions and differentiation between pseudotumours and solid lesions. Box 10.23.1.1 provides a list of European Federation of Societies for Ultrasound in Medicine and Biology (EFSUMB) recommended indications of CEUS in renal evaluation. EUROPEAN FEDERATION OF SOCIETIES FOR ULTRASOUND IN MEDICINE AND BIOLOGY (EFSUMB) RECOMMENDED INDICATIONS FOR USE OF UCA IN RENAL EVALUATION Renal ischaemia. Evaluation of solid renal lesions. Differentiation between solid renal lesions and pseudotumours. Characterization of complex cystic masses. Characterisation of indeterminate renal masses. Renal infections. Follow-up of nonsurgical complex masses. To improve lesion visualization in patients undergoing renal tumour ablation under US guidance and to detect residual tumour either immediately or after ablation. Isolated blunt moderate-energy renal trauma. Follow-up of trauma patients managed conservatively. Characterization of thrombus in renal vein and inferior vena cava. In patients with contraindications for CT- and MRI-based contrast media. Evaluation of lesions in transplant kidneys. Evaluation of ischaemia and vascular complications in transplant kidneys. Tumour response assessment to biologic therapy. Intracavitary CEUS for guiding percutaneous nephrostomy. Source: P.S. Sidhu, V. Cantisani, C.F. Dietrich, O.H. Gilja, A. Saftoiu, E. Bartels, et al., The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in non-hepatic applications: update 2017 (long version). Ultraschall in der Medizin-Eur. J. Ultrasound 39 (02) (2018) e2–e44. CEUS involves the intravenous injection of UCAs consisting of gas microbubbles. These UCAs remain purely in the intravascular compartment as they are small enough to avoid filtration by the lungs and too large to enter the interstitial compartment. UCAs cause marked amplification of signals from the flowing blood, thus providing information about the microvasculature and parenchymal perfusion. Sonovue is a second-generation UCA and is the only UCA available in India. It consists of sulphur hexafluoride gas which is exhaled by the lungs and is surrounded by phospholipid monolayer which is metabolised by the liver, making it safe for patients with renal insufficiency. For renal evaluation, a dose of 1–1.5 mL of Sonovue is administered as a bolus dose followed by a 10 mL saline flush. CEUS is performed on machines with contrast-specific software’s using a low mechanical index (MI) technique. The use of a dual image display is beneficial for the evaluation of small lesions. A conventional B-mode ultrasound is first performed to acquire measurements, assess morphology and to detect focal lesions along with Doppler ultrasound to assess vasculature. However, due to limitations in the assessment of focal lesions, microvasculature, complex cysts, poor contrast between the lesion and cortex or medulla, it is preferable to perform a CEUS study for additional characterization. The enhancement pattern of the kidney is different than that of the liver due to the dual blood supply of the liver as opposed to afferent arterial supply and efferent venous drainage of the kidney. After an intravenous bolus of UCA, kidneys show intense enhancement. Contrast is first seen in the main renal artery, followed by its branches. CEUS has only two enhancement phases, that is cortical phase which lasts for 15–30 seconds and a parenchymal phase (enhancement of both cortex and medulla) for 25 seconds–4 minutes after UCA administration. The renal cortex enhances first, followed by the outer medulla which is followed by gradual enhancement of the pyramids (Fig. 10.23.1.1). During the washout phase first, there is a reduction in the medullary enhancement followed by a slower washout of the cortex. As kidneys don’t excrete UCAs, there is no opacification of the pelvicalyceal system. UCAs can act as a problem-solving tool in patients with renal failure as they are not nephrotoxic. Due to its excellent ability to depict renal vascularization, it can be used for detecting perfusion abnormalities. CEUS is useful for detecting vascular causes of renal function deterioration. Renal infarcts appear as wedge-shaped nonenhancing areas in all phases. Renal infarcts show a focal complete lack of parenchymal enhancement on CEUS whereas ischaemic areas show reduced enhancement. CEUS can confidently differentiate infarction from cortical necrosis by the demonstration of preserved hilar vascularity in the latter. Renal pseudotumours such as foetal lobulations, dromedary hump, parenchymal hypertrophy due to renal scarring and column of Bertini can be accurately differentiated from true tumours using CEUS. Pseudotumours show similar enhancement to the adjacent parenchyma and appear isoechoic on all phases while true tumours show different enhancement pattern on at least one phase (Fig. 10.23.1.2). True tumours will cause mass effect and distort the normal vascular architecture of the renal parenchyma whereas in pseudotumours the vascular architecture is preserved. In addition, the identification of medullary pyramids within the mass points towards it being a pseudotumour rather than a true mass. Cysts can be categorized according to the Bosniak classification using CEUS. Due to its superior spatial and temporal resolution, CEUS is considered superior to CT for detecting additional septations, wall or septal thickening and solid components (Figs 10.23.1.3–10.23.1.5). CEUS allows for real-time imaging of blood flow within the septae, wall or nodules within the cysts which may not be seen on CT or MR imaging. The sensitivity of CEUS is comparable to CT for the classification of renal cystic lesions as benign or malignant however, it is not as useful for staging. The follow-up of inoperable complex cystic lesions is better performed with CEUS rather than CT due to the absence of ionizing radiation. Complex cysts or masses with calcifications are not suitable for evaluation with CEUS. CEUS allows a detailed evaluation of the circulation in mass lesions as it demonstrates the macro as well as microvascular enhancement patterns. CEUS has more sensitivity than CT for detecting blood flow in hypovascular lesions. It can also be used to distinguish between complex cysts and solid lesions which remain equivocal on CT and ultrasound (B-mode and colour Doppler). CEUS is sensitive in identifying cystic areas, necrosis, debris and haemorrhage in small tumours that may be difficult to visualize on CECT and MR. Isoechoic lesions on conventional ultrasound are better characterized by CEUS (Fig. 10.23.1.6). Hyperdense renal lesions on noncontrast CT (NCCT) with HU values between 20 and 70 are better evaluated by CEUS which can classify these lesions as solid or cystic. CEUS can suggest the histological subtype of tumours. Clear cell carcinomas show heterogeneous hyperenhancement in the cortical phase and rapid washout along with a peripheral enhancing rim or pseudocapsule seen during the parenchymal phase. Papillary carcinoma shows progressive heterogeneous enhancement during the cortical phase and displays hypoenhancement as compared to the cortex on all phases. Malignant renal vein thrombus will show enhancement as compared to bland thrombus on CEUS. Tumours that are smaller than 3 cm commonly show homogeneous enhancement, regardless of their histologic subtype. Differentiation between angiomyolipoma, renal cell carcinoma and oncocytoma is not always possible. Although, CEUS has excellent sensitivity for the detection of malignancy, it has a higher false-positive rate as compared to CECT. Hence, combined use of CEUS for its high sensitivity and CECT for its high specificity may be performed for an accurate diagnosis. CEUS is effective in patients with complicated pyelonephritis, for identifying inflammatory parenchymal involvement, characterized by round or wedge-shaped hypovascular parenchymal areas, most conspicuous during the late parenchymal enhancement phase. An abscess is seen as a nonenhancing area, with or without peripheral or septal enhancement. CEUS can also be used for the follow-up of renal abscesses. All the aforementioned indications of CEUS in native kidneys also apply to renal transplants. Infarction, vascular complications such as arterial and venous thrombosis can be detected. Quantitative CEUS for assessment of graft dysfunction is a subject of active research. CEUS can be used as an alternative to CT especially in children with isolated blunt moderate energy injuries who are hemodynamically stable. It can also be used in the follow-up of conservatively managed trauma patients and in renal impairment. During Focused Assessment with Sonography for Trauma (FAST), the examination should begin with the kidneys as they have a fleeting enhancement. The kidneys are studied in the arterial phase with two separate doses administered for either kidney during FAST. On CEUS, lacerations, hematomas, and infarcts are seen as nonenhancing areas on late-phase images whereas contusions may show faint enhancement. Pseudoaneurysms and active bleeding are diagnosed on the arterial phase. Crucial complications such as devascularized parenchyma and acute cortical necrosis can also be diagnosed on CEUS. Pelvicalyceal system injury cannot be detected by CEUS as UCAs are not excreted by the kidneys. CEUS can be performed in conjunction with percutaneous ablation therapies. Preablation evaluation with CEUS is essential to assess lesion vascularity and to compare pre and postprocedure tumour viability. Also, the identification of a pseudocapsule predicts improved ablation efficacy. CEUS has demonstrated high sensitivity, specificity and accuracy for the early detection of residual unablated tumour. Dynamic contrast-enhanced ultrasound is a useful tool for early identification of responders and nonresponders, enabling tailoring of the treatment regimen. It is performed by two methods – Bolus injection of a UCA with time-intensity curve analysis (commonly used) and intravenous injection of UCA with disruption–replenishment analysis. In patients with chronic kidney disease, the contrast enhancement is less intense and fades earlier as compared to a normal kidney. CEUS aids in characterizing renal lesions in patients with renal failure. Characterization of lesions with indeterminate appearances in patients with renal dysfunction, on conventional US prevents unnecessary further evaluation with contrast-enhanced CT or MRI and further deterioration of the renal function as well as unwarranted investigations. CEUS nephrostomogram can be performed after percutaneous nephrostomography to confirm the correct positioning of its tip, detect the site of obstruction and to diagnose complications. UCAs can be safely administered in patients with minimal risk. They can be safely administered in patients with renal insufficiency as they are not excreted by the kidneys. Prior laboratory tests are not indicated as they are not nephrotoxic and don’t interact with thyroid function. Most adverse effects are mild such as nausea, headache, chest discomfort, chest pain and resolve spontaneously. The contraindications include known hypersensitivity, patients with right-to-left shunts, severe pulmonary arterial hypertension (pulmonary arterial pressure >90 mm Hg), uncontrolled systemic hypertension, and respiratory distress. The rate of anaphylactic reactions is significantly lower than iodinated contrast media and comparable to gadolinium-based contrast agents. Caution should be exercised in patients with severe acute coronary disease and in patients with unstable angina. CEUS is a rapidly evolving technique, can be conveniently performed as an extension of conventional ultrasound and acts as a valuable tool in the characterization of indeterminate renal lesions, especially when the lesion is small. The advantages of CEUS include quick assessment, real-time imaging, high contrast resolution, excellent safety profile, lack of ionizing radiation, great repeatability, and most importantly, ideal for contrast evaluation in patients with deranged renal function. DUAL-ENERGY AND SPECTRAL IMAGING IN GENITOURINARY SYSTEM Aditi Chaitanya Gujarathi-Saraf Dual-energy CT (DECT) also known as ‘spectral imaging’, was first conceptualized in the 1970s. However, the clinical applications of DECT have recently emerged as owing to improvements in postprocessing techniques. The development of a single-detector, single-source DECT (ssDECT) with the capability for rapid alternation between two peak voltage settings (‘fast switching’) and a dual-detector, dual-source DECT (dsDECT) system in last few years have made possible various applications in solving clinical problems. In DECT, the simultaneous use of two different X-ray beam energy settings (80 and 140 kVp) allows the differentiation of materials on the basis of their attenuation characteristics (material density). There are two major applications of DECT in genitourinary system. First being characterization of renal calculi to differentiate those containing uric acid (UA) from those which do not. The other application is to characterize small renal masses which include differentiating complex cysts from renal tumours, detection of solid growth in PCKD patients and detection of residual tumour after thermal ablation of a renal neoplasm. The principle of DECT imaging is based on the differential absorption of X-ray energy at variable kVp settings. In DECT, two energy levels (typically 80 and 140 kVp) are used to acquire images that can be processed to generate additional datasets. Thus, a tissue or any material in the body shows different degrees of attenuation when scanned at different X-ray energies. Conventional CT numbers (attenuation measurements in Hounsfield units) are related to the linear attenuation coefficient of a particular material. The unique linear attenuation coefficients obtained by imaging at two different energies can be used to discriminate between different materials (e.g. fat, calcium, iodine and water). The characterization of material on DECT depends on their CT number ratio (CTR) also called dual-energy ratio. DE RATIO = CT number of a given material in the low-energy image CT number of the same material in the high-energy image DECT and spectral CT can be performed by three techniques (Fig. 10.23.2.1): Two sets of material-specific images are created to detect the presence or absence of a material. The two basic materials selected should have significant difference in mass attenuation coefficients and atomic number. The two commonly selected pairs are: Iodine-specific images are most commonly used in the clinical practice. When iodine is paired with water, two separate image data sets are generated. On water density image, the voxels that show change in attenuation due to presence of iodine are removed. These are instead represented on iodine density images. Thus, virtual unenhanced images are generated which resemble the conventional unenhanced images. Renal calculus disease is a common disease affecting approximately 10%–14% of population in India commonly affecting 31–45-year-old patients. Accurate diagnosis of composition of renal calculi is crucial as the frequency of prevalence and recurrence rate of renal calculi is high in most states of India. The obstructing calculi can lead to obstructive uropathy, pyelonephritis, urinary sepsis with nearly 50% of the patients being affected by renal failure. There are multiple compositions of renal calculi, most common being calcium oxalate (70%), calcium phosphate (20%), uric acid (8%) and cystine (2%). In general, UA calculi have lower attenuation values than nonuric acid calculi (i.e. struvite, cystine and calcium) on conventional CT. However, it may be difficult to distinguish between the two due to overlap in attenuation values. Here DECT becomes a problem-solving tool as it uses the two-material differentiation approach. Thus, in addition to size and location, DECT provides information about the composition of calculi to the urologist. DECT has high sensitivity of 88%–100% and high accuracy of 93%–100% to differentiate UA calculi from nonuric acid UA calculi. This is of utmost clinical importance as UA calculi can be treated medically whereas non-UA calculi require invasive methods of treatment such as extracorporeal shockwave lithotripsy (ESWL) or percutaneous nephrolithotripsy (PCNL). In clinical practice, NCCT scan of KUB region is performed at 80 and 140 kVp to generate two image datasets and obtain the DE ratio. A cut-off value of DE ratio of 1.2 is used to differentiate UA calculi from non-UA calculi (Fig. 10.23.2.2). Following flow chart shows differentiation of renal calculi based on DE ratio.
10.23: Recent advances
Introduction
F. Piscaglia, C. Nolsøe, C.A. Dietrich, D.O. Cosgrove, O.H. Gilja, M.B. Nielsen, et al., The EFSUMB guidelines and recommendations on the clinical practice of Contrast Enhanced Ultrasound (CEUS): update 2011 on non-hepatic applications, Ultraschall in der Medizin-Eur. J. Ultrasound 33 (01) (2012) 33–59.
Renal CEUS
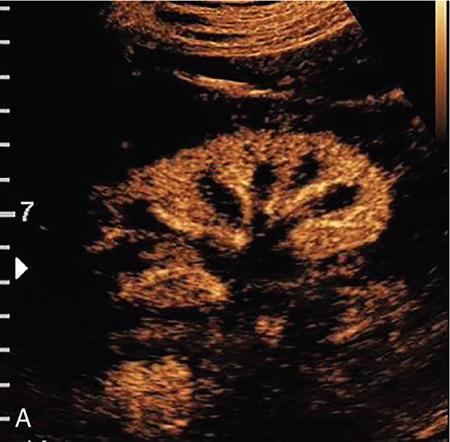
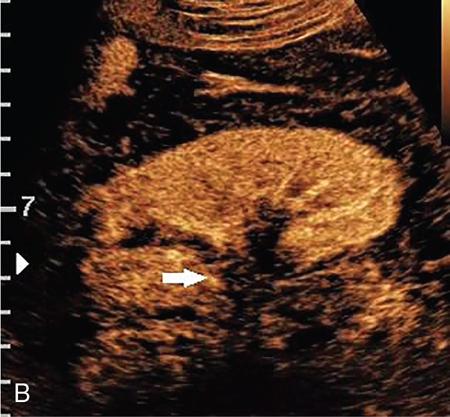
Evaluation of focal renal lesions
Renal ischaemia.
Renal pseudotumours.
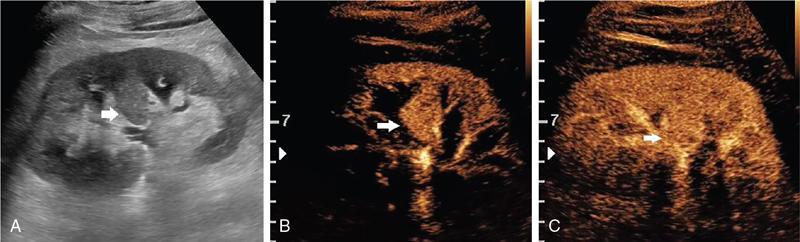
Cystic renal lesions.
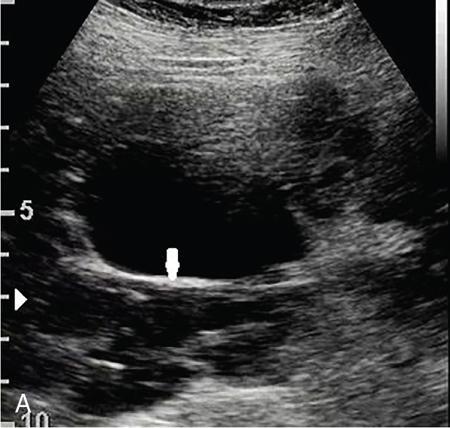
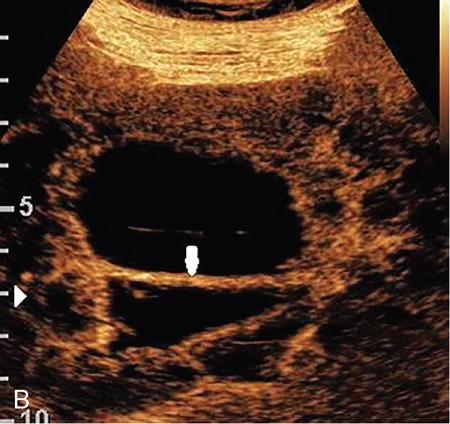
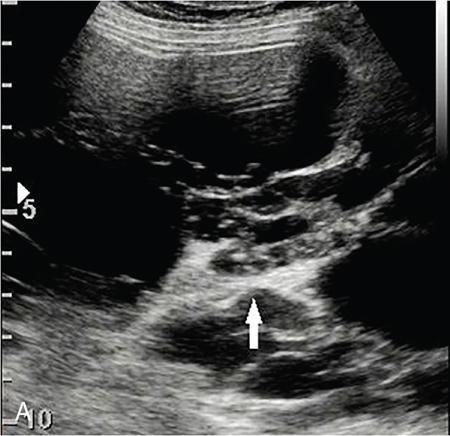
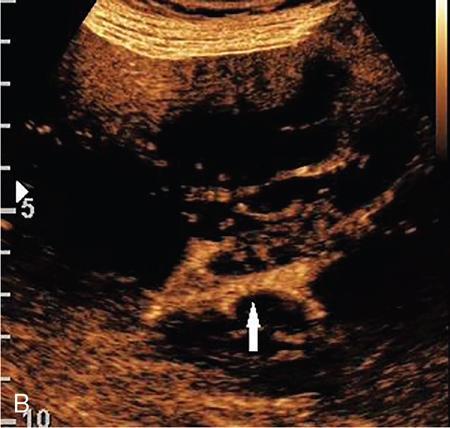
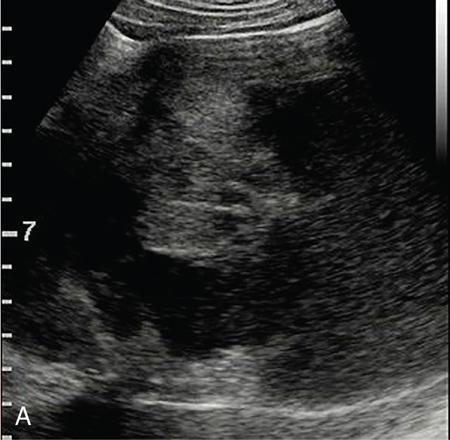
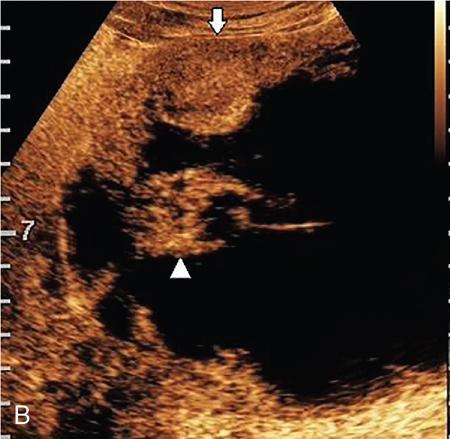
Solid renal lesions.
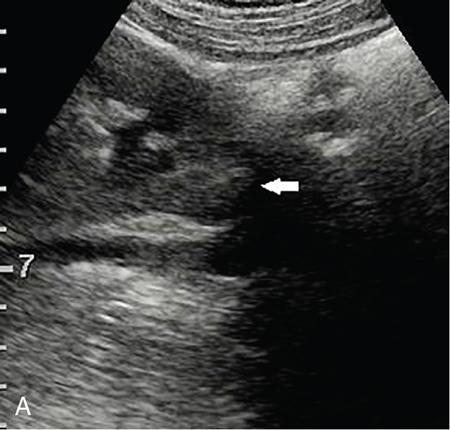
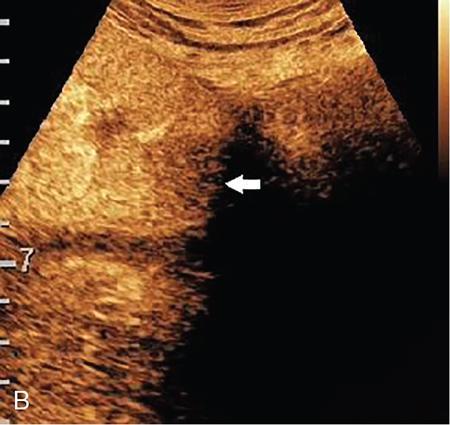
Renal infections.
Renal transplant evaluation
Trauma
Intervention
Tumour response assessment.
Evaluation of patients with renal insufficiency.
Intracavitatory CEUS for percutaneous nephrostomy.
Safety of UCAs
Conclusion
Introduction
Technical considerations
Principle
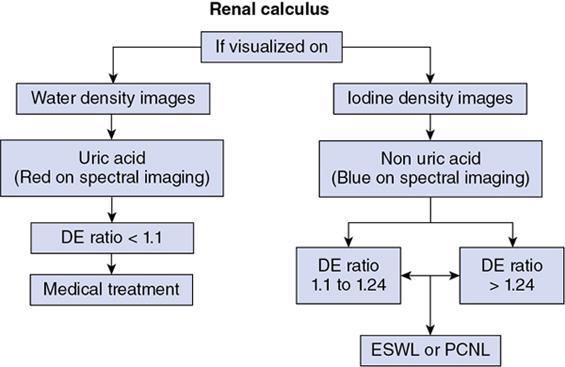
DE ratio

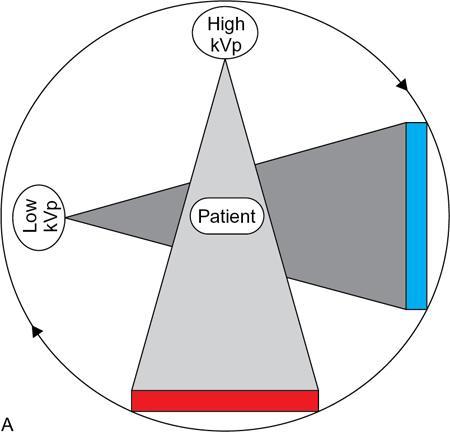
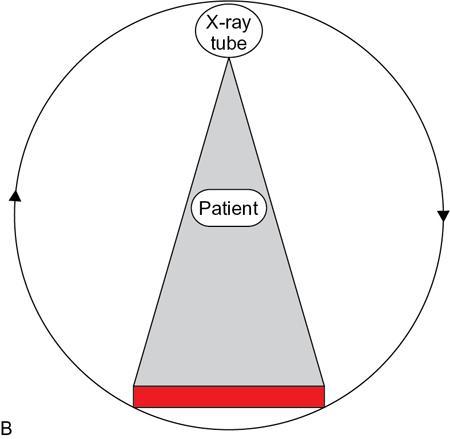
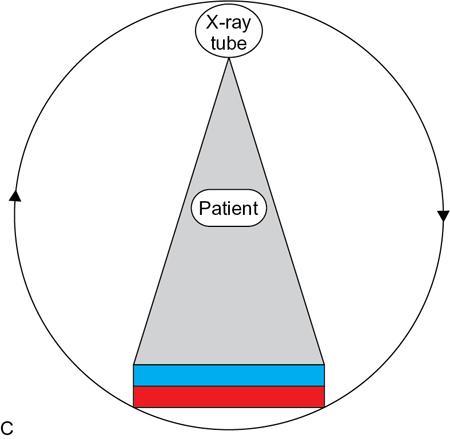
Spectral image reconstruction from projection datasets
Image display/material density display.
Applications
Renal calculus characterization.
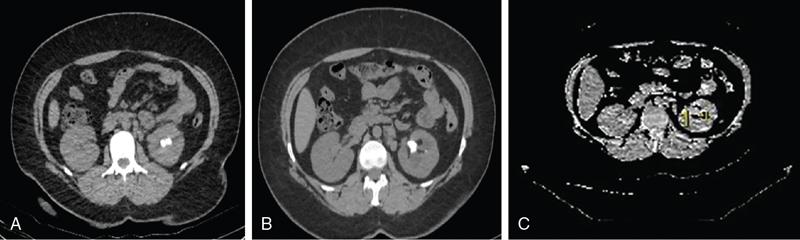
Characterization of renal lesions
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Recent advances
10.23.1
10.23.2






