Sumit Mukhopadhyay, Saugata Sen, Aditi Chandra, Argha Chatterjee, Priya Ghosh, Anisha Gehani Cloaca is the part of hindgut caudal to the attachment of the allantoic diverticulum. It divides into a broad anterior urogenital (vesicourethral and urogenital sinus) part and a narrow posterior anorectal compartment/primitive rectum, separated by the urorectal septum. This anorectal compartment gives rise to mid rectum and lower rectum and upper part of anal canal proximal to dentate line. The peritonealized upper part of rectum develops from hindgut proximal to cloaca. The distal most part of anal canal below the dentate line develops from invaginated ectoderm known as proctodeum (Fig. 8.5.1). Initially during development, the rectum is suspended from the posterior abdominal wall by a mesentery, along with the rest of the gut. After completion of rotation of the gut, rectal mesentery fuses with the posterior abdominal wall, majority of rectum becomes retroperitoneal and hindgut mesentery is seen as mesorectum. The artery of the hindgut is inferior mesenteric artery. Anatomically, classically taken as 6 inches (approximately 15 cm), or beginning at the level of S3 vertebra. Surgically defined as 15–18 cm from anal verge (AV), thus anal canal and rectum are considered together for the purpose of surgical and radiological anatomy, though management differs in cases of rectal adenocarcinoma and anal squamous cell carcinoma (SCC). Various trials on rectal cancer neoadjuvant therapy have taken different lengths of cut-off for rectal cancer starting from AV: 9 cm, 12 cm, 15 cm or 16 cm (Table 8.5.1). Rectum continues as sigmoid colon superiorly. The point of transition between rectum and sigmoid colon is controversial. Anatomically, this is the point where sigmoid mesocolon ends, appendices epiploicae are lost and taenia coli gradually converge and disappear. There is no single transition point between these features. These features are not visible to the radiologist/endoscopist as well. Several features defining rectosigmoid junction/transition are variously accepted by different radiologists: Rectum continues as anal canal, which opens to the exterior at AV (mucocutaneous junction). Anorectal junction (ARJ) is considered to be the point at which the angle of the long axis changes, and corresponds to the upper border of puborectalis muscle. The change in axis of anal canal and rectum is well visualized on sagittal MRI. Dentate line lies along the anal crypts (the openings of anal glands), and vertical folds called columns of Morgagni extend superiorly from the dentate line. Anatomists consider the dentate line to be the landmark for ARJ, but the dentate line lies distal to the puborectal sling, and this definition is not clinically relevant. Anal canal is directed posteroinferiorly. At ARJ, the long axis of rectum changes so that it is directed anteroinferiorly. Mid rectum and upper rectum follow the sacral concavity, and the long axis of rectum is again directed posteroinferiorly. Upper rectum is frequently mobile and may not be in midline. Three incomplete folds consisting of mucosa, submucosa and circular muscles, named as Valves of Houston, are seen within the lumen of rectum during endoscopy, but these are not visualized radiologically. The middle valve is roughly at the level of anterior peritoneal reflection, and the superior most valve is at the level of rectosigmoid junction. Surgically and radiologically, rectum is divided into thirds: lower third, middle third and upper third (Table 8.5.2). Most commonly, this division is according to distance or length from AV. Lower third is 0–5 cm, middle third is 5–10 cm, upper third is 10–15 cm from AV (Fig. 8.5.6). Some radiologists define low rectum as 6 cm or less from AV, mid rectum as 7–11 cm and upper rectum as 12–15 cm. The justification of defining low rectum as 6 cm or less from the AV is that, management of most of the tumours at this location suffers from surgical challenges due to tapering of mesorectum and proximity of other pelvic structures. These tumours have a higher rate of CRM positivity, abdomino-perineal resection (APR) and permanent stoma, higher rates of recurrence and mortality, poor function of stoma after sphincter preservation. Another way of defining low rectum, surgical management of which differs from the upper two-thirds, is the portion lying distal to the proximal origin of levators at pelvic side wall. This definition was provided by the English National Low Rectal Cancer Development Programme (LOREC) and is best visualized in the coronal plane by a line joining proximal origins of levators at pelvic side walls (Fig. 8.5.7). ARJ is an important landmark in the management of rectal cancer. Distance between lower border of the tumour and ARJ is important for treatment planning. ARJ is defined as the point at which puborectalis fuses with the levator muscles, and the long axis of rectum changes from anteroinferior to posteroinferior at this point. Its upper extent begins few centimetres above the dentate line, and its length varies from 2–5 cm from AV. Histologically the proximal end of the anal canal is the point at which the columnar epithelium of the rectum becomes the transitional epithelium. Anal canal mucosa has several longitudinal columns, known as anal columns, which end at the dentate line. Below the dentate line the mucosa is smooth and transitions into skin at AV. Approximately at the level of dentate line, the internal anal sphincter (IAS) ends, and the smooth mucosa leads to the intersphincteric plane, distal to which the external anal sphincter extends up to the AV (Fig. 8.5.8). Upper third of rectum is anteriorly covered by peritoneum. Peritoneal covering gradually widens superiorly till it is covered by peritoneum on all sides at the level of sigmoid colon. The exact level of peritoneal reflection varies between individuals, and is quite often seen in axial and sagittal MRI. In axial T2-weighted MRI, a thin hypointense layer with V-shaped attachment to anterior rectal wall is seen representing the anterior peritoneal reflection. This is known as the ‘sea-gull’ sign. Mesorectum and mesorectal fascia (Fig. 8.5.10): lower two-thirds are surrounded by extraperitoneal fat and connective tissue known as mesorectum, which contains lymph nodes, nerves and blood vessels (descending branches of superior rectal artery and corresponding veins). Mesorectal fat is contained within a thin layer of fascia, known as mesorectal fascia (MRF). MRF separates mesorectal fat from extra mesorectal tissues of the pelvis. A relatively avascular plane of areolar tissue lies between MRF and parietal pelvic fascia, facilitating surgery. Importance: Assessment of structures within mesorectum is important for staging and prognostication of rectal cancer. Mesorectal node involvement is N disease. Tumour extension within mesorectal vessels is known as extramural vascular invasion (EMVI) and is thought to be associated with higher chances of vascular dissemination and distant metastases. MRF forms the boundary of the surgical excision plane in total mesorectal excision (TME) – the standard surgery for rectal cancers at present. It is important to understand the various fascial reflections of the pelvis in order to understand pathways of disease spread. The plane between the MRF and the pelvic fascias is the surgical plane. Posteriorly, covering the sacrum, presacral venous plexus and hypogastric nerves, lies the presacral fascia. It fuses with MRF inferiorly at the level of levator ani muscle. The space between presacral fascia and MRF is known as the retrorectal/rectosacral/pelvirectal space, and that between the sacrum and presacral fascia is the presacral space. The presacral fascia continues laterally as the parietal pelvic fascia which covers the lateral pelvic wall. It has two lamellae which encase the pelvic visceral nerves as they course forward from the sacrum to the anterior pelvic organs. The rectosacral fascia/Waldeyer fascia, called as rectosacral ligament by anatomists, is a thickening arising from presacral fascia and running forward to meet the MRF. This divides the rectosacral space into a superior and an inferior compartment, communicating with each other. In males, anteriorly, between the rectum and prostate-seminal vesicles, lies the rectoprostatic fascia/Denonvillier fascia. It is difficult to distinguish from the closely related MRF, and carries the hypogastric nerve and vascular branches to the prostate and male genital organs. In females, anterior to the MRF, lies the rectovaginal septum. According to most views, this septum consists of two layers. The anterior layer corresponds to Denonvillier fascia, and extends from the pelvic floor to the posterior wall of vagina and uterus. The posterior layer is in close relation to the MRF, runs from the pelvic diaphragm and ascends to the peritoneal reflection before fusing with the rectal wall. The lateral rectal ligaments are controversial structures as they are not visualized by imaging and may not carry important structures such as nerves and vessels. However, in some cases, the middle rectal artery may run through them and the accompanying lymphatics may provide a pathway between mesorectal and extra mesorectal lymph nodes. In females, the pelvic floor may be considered to have three compartments, anterior compartment containing bladder and urethra, middle compartment containing the vagina and the posterior compartment containing the rectum. These compartments are supported by the endopelvic fascia and levator ani muscle. The levator ani has several compartments, of which the two most important ones are the iliococcygeus and puborectalis. The iliococcygeus starts as the same fibres as the EAS, and then fans out as a sheet to insert at the pelvic sidewall at the tendinous arch. Posteriorly these fibres fuse in the midline to form the levator plate/raphe. The pubococcygeus and puborectalis are considered together as pubovisceralis muscle. It inserts lateral to the symphysis pubis anteriorly, and forms a sling around the rectum, pulling it anteriorly. Components of the levator ani can be identified in T2-weighted MR images. In males, there are two compartments, anterior containing bladder, urethra, prostate and seminal vesicles and the posterior compartment containing the rectum. Pubococcygeus consists of pubourethralis and puborectalis in males. In sagittal images, the pubococcygeal line is an important reference line, drawn from the inferior border of symphysis pubis to the last joint of the coccyx, representing the level of pelvic floor. The H line is drawn from the inferior aspect of the symphysis pubis to the posterior wall of the rectum at the level of ARJ. This represents the anteroposterior width of the levator hiatus, and upper limit of normal is 5 cm. The M line is the vertical descent of the levator hiatus, drawn as a perpendicular line dropped from the pubococcygeal line to the posteriormost aspect of the H line, and should measure maximum 2 cm. The angle of the levator plate and the pubococcygeal line is also measured. In axial images, the entire normal levator is of uniform thickness and homogeneous low signal intensity. Coronal images show intact iliococcygeus muscle which is convex upwards. The upper half of the anal canal proximal to the pectinate line, the IAS, the conjoint longitudinal coat and the rectum drain lymph upwards into the mesorectal nodes (Fig. 8.5.11) and then lymphatics follow the superior rectal artery into the inferior mesenteric group of lymph nodes. The lymph from these nodes is carried by the intestinal lymph trunk(s). Anal canal below the dentate line along with the EAS drains to the superficial inguinal nodes. Lymphatic vessels also travel with the median sacral artery and drain the puborectalis muscle before finally joining the internal iliac lymph nodes. The internal iliac lymph nodes drain into pre-aortic and paraaortic (lumbar) lymph nodes and efferents from the nodes form the lumbar trunks. The intestinal trunks and the lumbar trunks enter the abdominal confluence of the lymph trunks at the level of the L1–L2 vertebrae, called the cisterna chyli, and then ascend as the thoracic duct. The pelvic lymph nodes lying outside the mesorectum are termed as ‘extra mesorectal lymph nodes’. Above the dentate line: blood supply is from the superior rectal artery, which originates from the inferior mesenteric artery, a branch of abdominal aorta. Superior rectal artery passes in the sigmoid mesocolon and divides into two branches behind the rectum at the level of S3 sacral segment. The superior rectal artery ends in the anal columns by forming a rich vascular plexus/anastomosis with the branches of inferior rectal artery. Blood returns via the superior rectal veins into the inferior mesenteric vein, which drains into the splenic vein and eventually into the portal venous system. Below the dentate line, the inferior anal canal obtains its blood supply from the inferior rectal artery, which is a branch of internal pudendal artery, originating from the anterior division of internal iliac artery. Blood returns via the inferior rectal vein, which drains into internal pudendal veins, internal iliac veins and ultimately into the inferior vena cava (systemic circulation). Thus anal canal is a site of portosystemic anastomosis. Due to the venous anastomoses that occur in the anal canal and the backup of blood flow into the rectal veins, haemorrhoids may be present in patients with portal hypertension. A small part of muscular wall of rectum and anal canal is also supplied by median sacral artery, a direct branch of abdominal aorta arising at the bifurcation of aorta. Nerve supply: The inferior hypogastric plexus lies laterally on the surface of MRF on both sides. It receives sympathetic nerve fibres from the superior hypogastric nerves and parasympathetic ‘nervi erigentes’ from the laterally situated sacral nerves (S2–S4). Laterally, inferior rectal branches of the pudendal nerves and internal pudendal arteries cross the ischio-anal fossa to supply the external sphincter and anal mucosa. T2-weighted small FOV images reveal the layers of rectal wall and details of perirectal soft tissue and pelvic floor most accurately. For adequate assessment of the integrity and involvement of different layers, axial images are needed, which should be obtained perpendicular to the axis of the rectum in that segment. Angle of acquisition should be altered according to the change in axis of rectum (Fig. 8.5.12). The layers of rectal wall from inside to outside are (Fig. 8.5.10): When examining the anorectum with a radial scanning echoendoscope, the internal and EASs can be seen as two distinct rings. The inner hypoechoic ring of tissue represents the IAS the outer hyperechoic tissue ring represents the EAS. The thickness of normal IAS is 2–3 mm, and for EAS: 7–9 mm. The IAS becomes thicker and more hyperechoic with age, while the EAS tends to become thinner with age. Endoscopically, rectum begins at the dentate line and extends to 15–20 cm from the AV. The normal five-layer appearance of rectal wall in EUS is as follows: Imaging in the rectum and anal canal region is mainly for staging of tumours. The most common neoplasm of this region is rectal cancer and preoperative imaging evaluation is required not only for staging early and advanced disease but also for assessing response to treatment and for surgical planning. The techniques described here will mainly pertain to rectal and anal neoplasms. Plain radiographs have a limited role in the evaluation of rectal neoplasms. Primarily, it may be used as a modality in the emergency setting when patients come with constricting colorectal strictures causing large bowel obstruction. Radiographs of the abdomen in supine and erect positions are performed to look for features of bowel obstruction, air-fluid levels and free air under the domes of diaphragm in case of perforation, respectively. Conventional luminal contrast study is now obsolete for diagnosis of tumours in the rectum and anal canal and have been replaced by cross-sectional imaging. Sinogram studies with diluted iodinated contrast have been performed to detect extent of rectal fistulae. Endorectal Ultrasound (ERUS) can differentiate between the layers of the rectal wall and helps detect and stage tumours within the different layers of the rectum. ERUS can view the rectal mucosa in 360 degrees. The layers visualized include rectal mucosa, muscularis mucosa, submucosa, muscularis propria and area between the muscularis propria and perirectal fat. The role of ERUS in rectum is primarily in staging and in posttreatment surveillance of rectal adenocarcinoma. Staging of early tumours requires an ERUS due to its superior diagnostic performance for differentiating T1 from T2 tumours in comparison to other cross-sectional imaging modalities like MRI. As far as surveillance is concerned, endoscopy is mandated as it can detect early asymptomatic recurrences which improves overall survival (OS). This is substantiated by multiple recent meta-analyses in literature. Hence, even for patients on ‘watchful waiting’ as a treatment option after chemoradiotherapy, it is imperative to perform a DRE, EUS, CEA and correlate the findings with restaging MRI. The various recommendations for postoperative surveillance for rectal cancer using endoscopy are enumerated in Table 8.5.3. ASCO: American society of clinical oncology; ESMO: European society for medical oncology; NCCN: National comprehensive cancer network; USMSTF: United States Multi-Society Task Force. Drawbacks include heterogeneity in operator skill and operator dependency as well as inability to pass stenosing lesions. Poor depth of penetration of the probe results in reduced visualization of the mesorectal fat, limited assessment of pelvic side walls and EMVI and reduced diagnostic accuracy for asserting involvement of CRM. Multidetector CT (MDCT) is the primary imaging modality used for rapid evaluation of not only bowel pathologies (luminal or mural) but also for evaluation of surrounding structures such as vessels, lymph nodes and mesentery. The ability to obtain high-quality clinical images through multiplanar reconstructions make it one of the most robust examinations for the initial evaluation of most bowel related pathologies. A routine abdominal CT scan would include acquiring a noncontrast axial image, followed by a portal phase image (at 70–90 seconds). However, for more detailed evaluation of solid organs, a general imaging dataset would include an unenhanced CT, followed by arterial phase (20–30 seconds), portal venous phase (70–90 seconds) and a delayed phase at 3 minutes. Many centres around the world omit the noncontrast scan to reduce radiation exposure. Lesions such as large tumours, pelvic nodes, collections and diverticulosis can be detected by contrast-enhanced CT. CT provides a more holistic evaluation and demonstrates possible complications of tumours such as obstruction, transition point and perforation that may not be clinically evident. However, accurate detection and staging of tumours in the rectum is better performed by MRI. Like TRUS, MRI can depict the layers of the rectal wall with high resolution, especially when performed at 3 Tesla and with an endorectal coil. Although use of endorectal coils may provide improved diagnostic accuracy for T stage as compared with phased-array coils alone, it is known that endorectal coils increase patient discomfort and may account for increasing motion artifacts. Insertion of such coils is also not possible in case of stricturous lesions. Performance of high-resolution imaging using phased-array MRI coils at 1.5 or 3 Tesla, as was used in multicentre trials was excellent. MRI technique and image quality play a critical role in evaluation of rectal cancers, and accuracy is dependent on obtaining high-resolution images that are perpendicular to the plane of the tumour. Particular note is to be made whether the tumour is mucinous or nonmucinous as the former have worse prognosis and higher tendency to metastasize. For technical aspects of MRI, it is recommended by ESGAR that bowels and bladder are emptied. Rectal distension is not indicated as it stretches out the CRM. Minimum slice thickness for such scans is 3 mm. DWI is essential for both primary staging and restaging. Microenemas may be considered to remove air from the rectum and reduce artifacts in DXI sequence. High-risk MRI features for distant metastases include EMVI, mesorectal tumour depth >5 mm, T4 stage, involved circumferential resection margin (CRM). In addition to initial staging prognostic features, MRI also helps in assessing response to neoadjuvant therapy which is also an indicator of survival and chances of recurrence. MRI is sensitive in detecting the presence of lymph nodes but remains nonspecific for differentiation malignant from benign nodes with high diagnostic certainty owing to the presence of micrometastasis even in small-sized nodes (up to 5 mm). Contrast administration is not recommended. Drawbacks of MRI include reduced diagnostic accuracy for identifying early rectal T1 and T2 tumours, and to sometimes differentiate T2 from early T3 lesions. Increased scan times in the elderly and cost may also be constraints in some settings. Virtual colonoscopy can help in identifying primary and synchronous colonic lesions. CT Colonography (CTC) is beneficial after incomplete colonoscopy (due to nonpassable stricture) to evaluate the remainder of the colon. CTC is advocated as a screening test for colonic polyps and colon cancer in vulnerable populations. It has a sensitivity of 93% and a specificity of 97% for detecting polyps >1 cm. MRI scores over all modalities in the local staging of rectal neoplasms. The more common application of PET-CT is in identifying nodal and distant metastases in rectal adenocarcinoma, melanoma and lymphoma. Limitations of PET include poor sensitivity in detecting small (<10 mm) colonic lesions and decreased fluorine-18-2-fluoro-2-deoxy-D-glucose uptake by mucinous tumours. For rectal melanoma, MRI may be performed along with a whole body PET-CT scan for regional and overall staging. Rectal lymphoma does not require local staging and a whole body PET-CT is generally acceptable as the imaging modality of choice. MRI is the imaging modality of choice due to better delineation of the extent and involvement of the anal sphincters. The inguinal nodes are regional nodes in anal cancer and this region is covered in the scan. The most common rectal tumour is rectal cancer. It is also one of the most common malignant neoplasms and the second most frequent cancer occurring in the large bowel. Majority of the patients are in the fifth to seventh decade. However, the incidence of rectal cancer has been on a rise in the younger population. Adenocarcinoma is the commonest histopathologic type of rectal cancer. Other than adenocarcinoma, several other neoplastic lesions also occur in the rectum, but are relatively uncommon (Table 8.5.4). Diagnosis of rectal cancer begins with physical examination/DRE followed by colonoscopy/sigmoidoscopy and biopsy from the mass (see figure below). If malignancy is confirmed on histopathological examination, imaging for staging is ordered (Fig. 8.5.14). MRI is the preferred method for locoregional staging. During staging of early tumours, ERUS also plays an important role. Majority of the guidelines (SAR and ESGAR) accept MRI as the modality of choice for locoregional staging and restaging after neoadjuvant treatment. CT thorax and abdomen is performed for metastatic evaluation. PET-CT is not routinely recommended in metastatic evaluation. ESMO guidelines outline the following indications for PET-CT: (1) If carcino-embryonic antigen (CEA) is high on presentation (2) Extensive EMVI. MRI of the liver may be recommended to evaluate any suspicious or equivocal lesions that are detected on CT scan. Therefore routinely pelvic MRI for locoregional evaluation and CT thorax and abdomen for metastatic evaluation is performed (Table 8.5.5). Based on the investigations, the rectal cancer is staged according to the latest AJCC classification (Table 8.5.6). T0 No evidence of primary tumour Tis Carcinoma in situ T1 Tumour invades submucosa T2 Tumour invades muscularis propria T3 a b c d Tumour invades subserosal tissue and perirectal tissue a <1 mm b 1–5 mm c >5–15 mm d >15 mm T4 a b Tumour invades peritoneum or other organs Tumour penetrates visceral peritoneum Tumour invades other adjacent organs or structures NX Regional lymph nodes cannot be assessed N0 No regional lymph node metastasis N1 a b c Regional lymph node metastasis (1–3 nodes) 1 lymph node 2–3 lymph nodes Tumour deposits in subserosa, mesentery/nonperitonealized perirectal tissues (cannot be differentiated from nodes on imaging) N2 a b Regional lymph node metastasis (>4 nodes) 4–6 node >7 nodes M0 No distant metastasis M1 a B c Distant metastasis Metastasis in one (1) organ Metastasis in more than one organ Metastasis to the peritoneum with/without other organ involvement Once the diagnosis is confirmed and staging investigations are completed, management is usually decided in multidisciplinary team meetings consisting of Surgeon, Radiation oncologist, Medical oncologist, Pathologist and Radiologist. The treatment protocols differ in the United States and Europe (Fig. 8.5.15). The different types of rectal surgeries are enumerated in the Table 8.5.7 and depicted in Fig. 8.5.16. Concept of use of neoadjuvant short course RT, long course RT with chemotherapy and chemotherapy only is a rapidly evolving field. Radiologist should be aware of the protocol used in their institution. A subset of patients (10%–23%) was found to have complete pathological response (pCR) in the postsurgical pathological specimen. There is significant evidence of prediction of pCR on presurgical MRI. Considering the ability of MRI in prediction of pCR, Prof. Habr-Gama and her group suggested the possibility of organ preservation in these patients. Hence ‘watch and wait’ policy came into vogue. In the subgroup where MRI predicts complete response, surgery can be avoided and patients may be followed up every 8–12 weeks using DRE, proctoscopy/sigmoidoscopy and MRI. Both T2W and DWI are used in MRI for prediction of complete response as well as for follow-up for prediction of recurrence. Close follow using the above-mentioned protocol ensures early detection of recurrence and hence treatment. Thus, MRI plays an important role in personalized treatment of rectal cancer. MRI plays an important role in rectal cancer management: During initial staging MRI helps in: In restaging after NACT, MRI helps in: Therefore good-quality high-resolution rectal MRI is required for accurate locoregional staging. Technique and protocol of MRI is summarized in the Tables 8.5.8 and 8.5.9. 1.5T-3.0T magnet strength High resolution T2w sequences Small FOV images Axial and coronal images – parallel and perpendicular to the rectal tumour For low rectal tumours coronal images –perpendicular to the anal canal
8.5: Rectum
Embryology
Overview
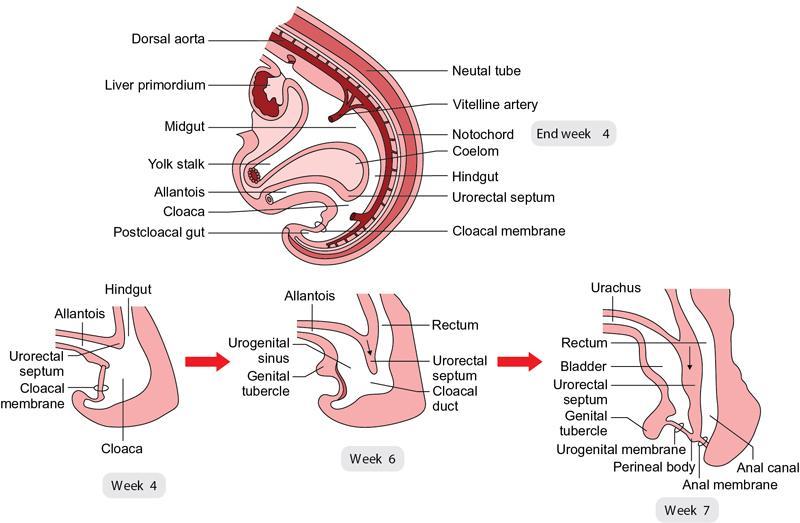
Radiological anatomy
General anatomy
Length
Definition by
Length
Anatomists: View 1
6 inches (15 cm)
Anatomists: View 2
Distal to S3 vertebra
Surgeons
15–18 cm from anal verge
Hwang MR et al. (2014) National Cancer Centre, Republic of Korea (3)
9 cm
Trans-Tasman Radiation Oncology Group trial 01.04 (2013)(4)
12 cm
NASABP R-03 trial (5)
15 cm
Bosset J-F et al. (2006), multicentre trial: EORTC Radiotherapy Group Trial no. 22921(6)
MRC CR07 and NCIC-CTG C016 trial (7)
Sauer R et al. (2004), multicentre trial (8)
16 cm
Hofheinz R-D et al. (2012), German multicentre trial (9)
Upper limit/rectosigmoid junction
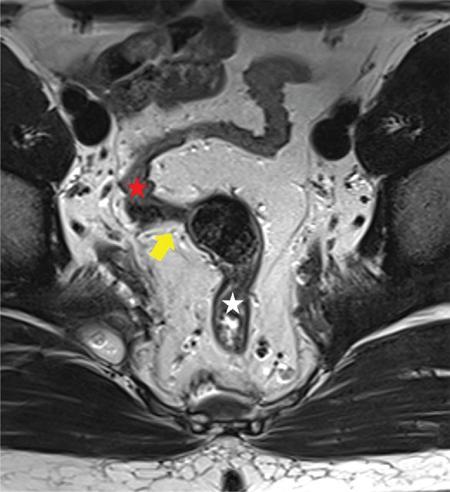
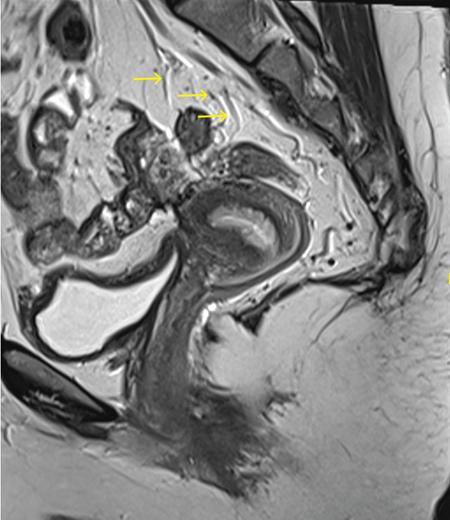
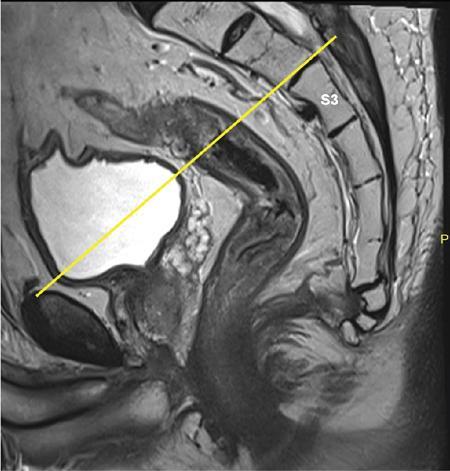
Lower limit (Fig. 8.5.5)
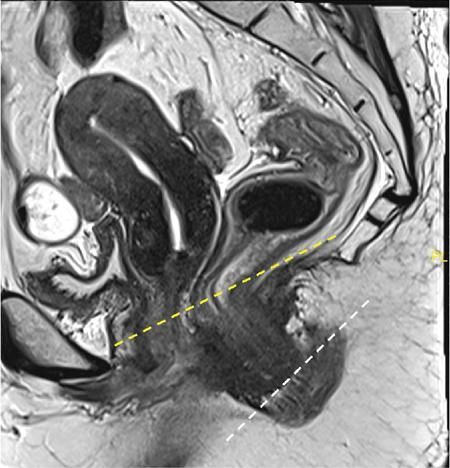
Course and features
Divisions
Common Definition Used by Radiologists
Alternate Definition
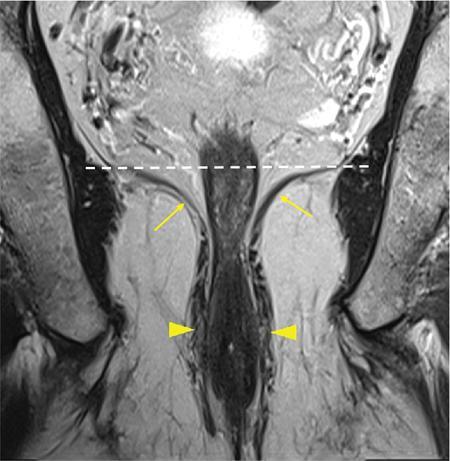
Definition by LOREC
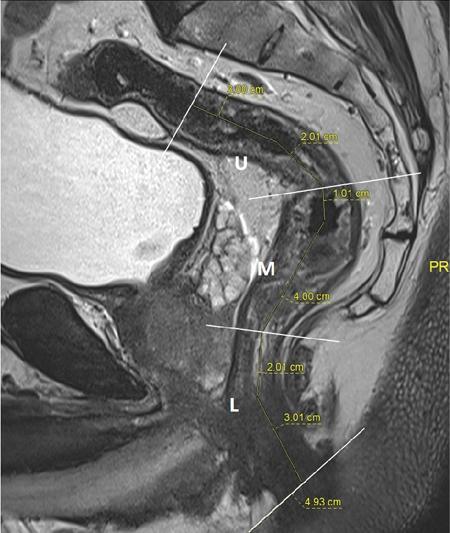
Low rectum
0–5 cm from anal verge
0–6 cm from anal verge
Part of rectum lying distal to the proximal origin of levators at pelvic sidewall
Midrectum
5–10 cm from anal verge
7–11 cm from anal verge
–
Upper rectum
10–15 cm from anal verge
12–15 cm from anal verge
–
Anal canal: When separately considered
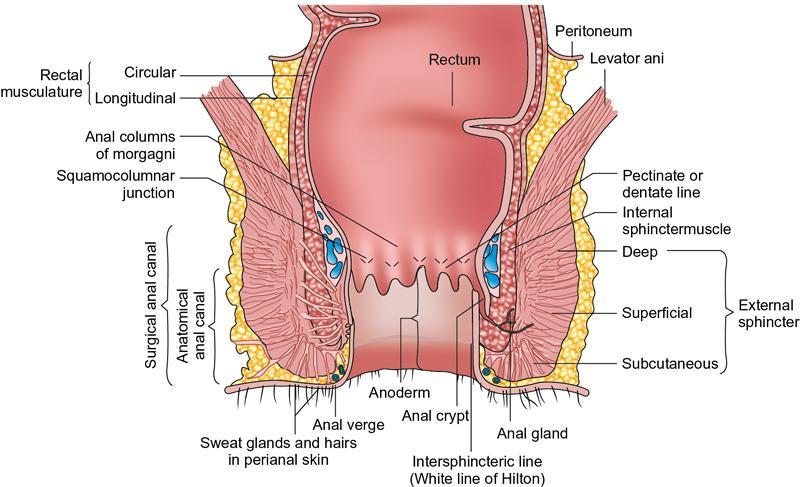
Layers/histology (Fig. 8.5.9)
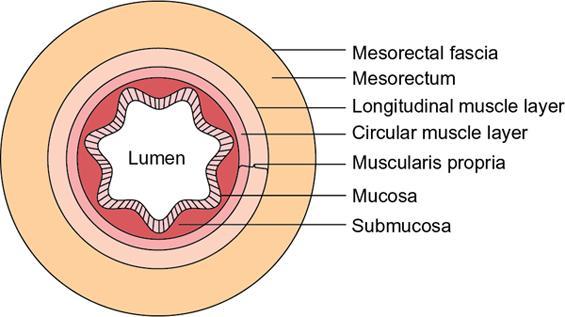
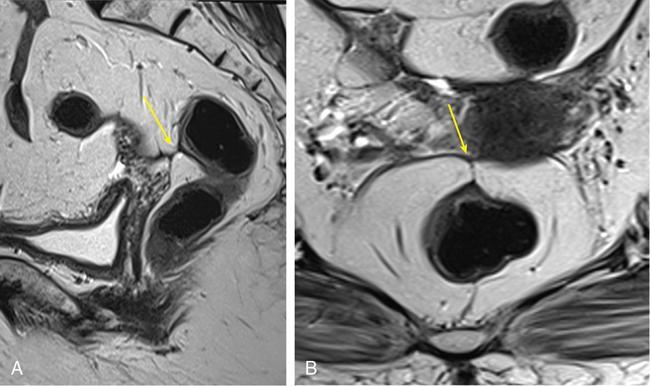
Peritoneal covering
Pelvic fascial planes
Sphincters
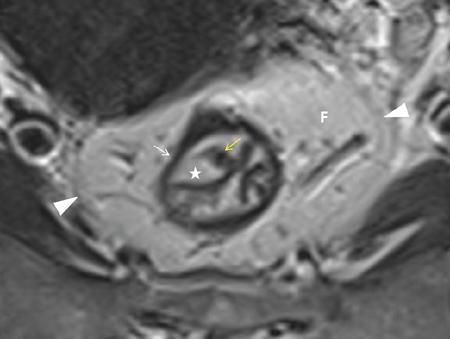
Levator/pelvic floor anatomy
Image interpretation, normal lines and landmarks in T2-weighted MRI
Nodal drainage pathway
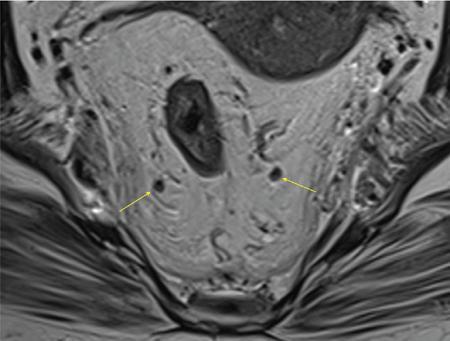
Vascular supply
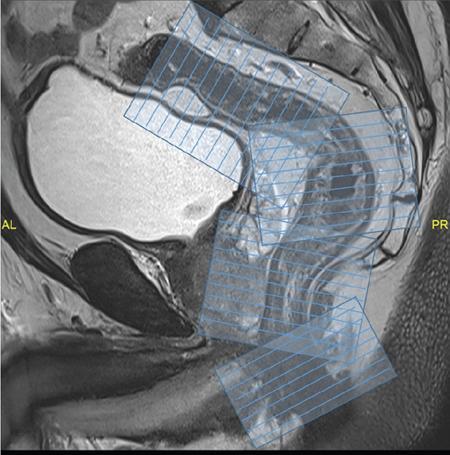
MRI anatomy
Endorectal ultrasonographic anatomy
Imaging techniques
Plain radiograph
Barium/gastrografin studies
Endorectal ultrasound
ASCO 2013 (42)
Colonoscopy at 1 year, followed by every 5 years
ESMO rectal 2013 (43)
Colonoscopy every 5 years up to age 75
NCCN 2018 (44)
Colonoscopy at 1 year, repeat at 3 years, then every 5 years subsequently. If adenoma is found, repeat annually
USMSTF 2016 (45)
Colonoscopy at 1 year, repeat at 3 years and then every 5 years.
Multidetector CT
MRI
CT colonography
PET-CT
Summary of recommendations
Locoregional staging of rectal cancer
Staging of other rectal neoplasm
Staging of anal cancer
Rectal pathologies
Rectal tumours
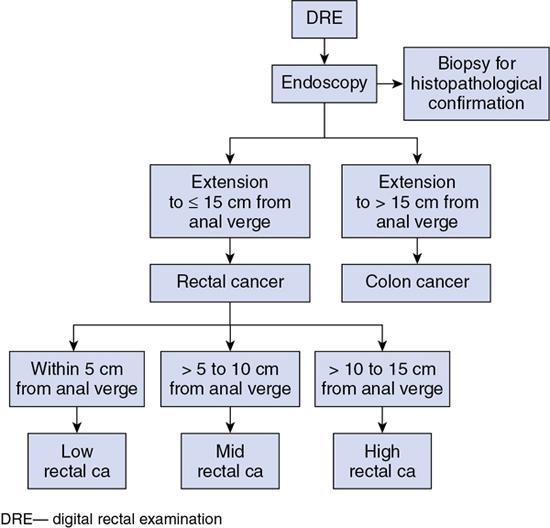
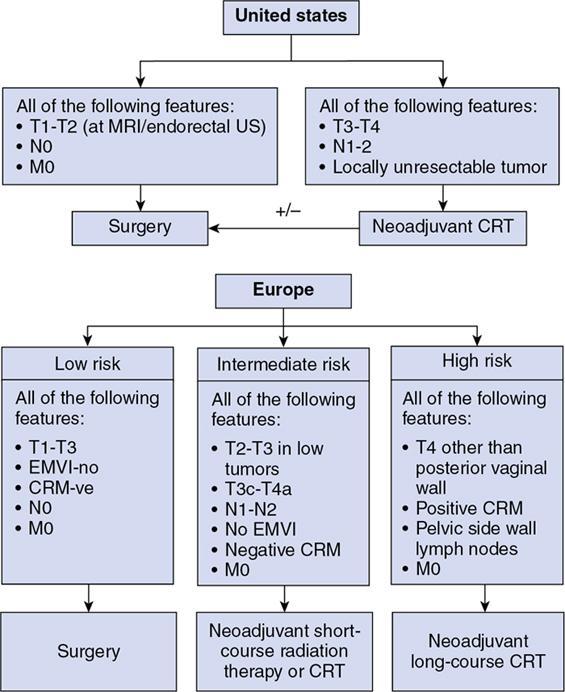
Diagnosis, staging and management of rectal cancer
Parameter
Investigation of Choice
Other Investigations
Tumour location
DRE/sigmoidoscopy
MRI
Tumour morphology
Histopathology
Tumour (T) Staging–early
ERUS
MRI
Tumour (T) Staging–advanced
MRI
ERUS
Nodal staging
MRI
ERUS/CT
Sphincter involvement
MRI
DRE/ERUS
Metastatic evaluation
CT
PET-CT if EMVI/raised CEA
MRI – liver evaluation
PRIMARY TUMOUR (T)
REGIONAL LYMPH NODES (N)
DISTANT METASTASES (M)
Types of Surgery
Indication
Transanal Endoscopic Microsurgery (TEM)
T1(sm1)
Total Mesorectal Excision (TME)
T1(sm2, sm3), T2, T3a, T3b
Post-CRT T3/T4
Low Anterior Resection (LAR)
Tumours above anorectal junction
Abdomino-Perineal Excision (APE)
Tumours below anorectal junction/levators not involved
Extra Levator Abdomino-Perineal Excision (ELAPE)
Tumours below anorectal junction/levators involved
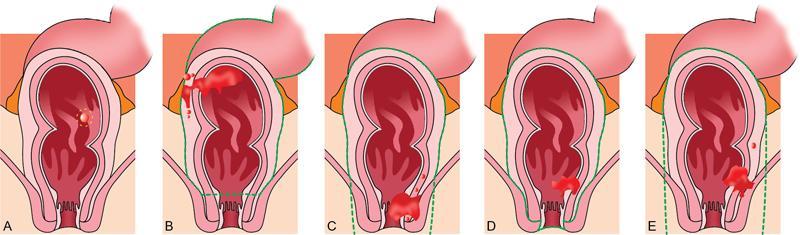
(A) Transanal endoscopic microsurgery (TEM) with resection of a tumour.
(B) Low anterior resection (LAR) with total mesorectal excision (TME) with sigmoid colectomy.
(C) Abdominoperineal resection (APR) and TME, with resection of the sphincter complex.
(D) Intersphincteric abdominoperineal resection (APR) and TME.
(E) Extralevator abdominoperineal resection (ELAPE) and TME.
MRI imaging
Recommended
Optional
Not recommended
Bowel preparation
Endorectal coil
Spasmolytic agents
Endorectal filling with gel
Microenema
IV Gadolinium contrast
MRI Protocol
LARGE FOV FOR PELVIS
SMALL FOV FOR RECTUM
T2 TSE Axial
DWI Axial
T2 TSE Sagittal
T2 TSE Coronal
T1 TSE Axial
RESOLVE DWI Axial
TR (ms)
4500–5000
8800–13500
3500–5500
3000–6000
500–900
4500–6500
TE (ms)
85–100
75
80–100
80–100
12–24
55–60
FOV (mm × mm)
380 × 330
420 × 315
190 × 190
180 × 180
180 × 180
240 × 240
Slice thickness (mm)
5
5
3
3
3
3
B values
NA
50,400,800
NA
NA
NA
50,400,800 ![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Radiology Key
Fastest Radiology Insight Engine






