Etiology
Rheumatoid arthritis (RA) is an autoimmune disorder of unknown cause with variable clinical manifestations. Genetic factors play a major role in disease susceptibility and expression.
The B lymphocyte is important in the pathogenesis of RA, and therapeutic B-cell depletion has been shown to be beneficial in active RA. The exact trigger for autoantibody formation is unknown, but in animal models, both B cells and non–B-cell antigen-presenting cells are required for the development of autoimmunity. Most connective tissue diseases (CTDs) have known human leukocyte antigen (HLA) associations, and the link with the HLA-DRB1 gene is best understood. In RA, CD4 + T cells have a role in the development of pulmonary parenchymal disease inasmuch as the interstitial pneumonias seen in patients with RA are characterized by increased CD4 + cells (T-helper/inducer cells) when compared with those with idiopathic interstitial pneumonia.
Prevalence and Epidemiology
RA is estimated to affect 1% of the adult population worldwide, but there are some marked demographic differences. RA has a higher incidence in North American Indians (5.3%) and is two to three times more common in women, but some manifestations of RA, such as pleural effusions, necrobiotic nodules, and pulmonary fibrosis, are more frequent in men. Patients with RA have a shortened life expectancy by 5 to 10 years.
Cigarette smoking has a synergistic effect on the pulmonary manifestations of RA, and smoking is the most consistent independent (and preventable) predictor of interstitial lung disease (ILD) in RA. The exact pathogenic role of cigarette smoke remains unclear, but the gradient of risk associated with the development of pulmonary involvement in RA increases with exposure to cigarette smoke. Smokers frequently have several abnormalities on high-resolution computed tomography (HRCT), and it can be difficult to tease out the contribution of pulmonary involvement in RA versus smoking-related disease.
A positive rheumatoid factor (RF) early in the course of the disease is a prognostic marker for premature mortality. The relative risk of dying has been estimated to be six times higher in seropositive patients.
More than a third of patients with RA die of infections; in half of such cases this is not recognized before death. The most common sites of infection are the respiratory and urinary tracts. A national study of RA patients in Finland demonstrated that the life span of subjects with RA was shortened by up to 20%, and the excess deaths were usually due to cardiovascular causes, infections, and amyloidosis. Patients with RA also have an increased risk of death from lung cancer, hematopoietic malignancies, chronic obstructive pulmonary disease, and renal failure. Disease-modifying drugs are associated with malignancy, and there are several case reports of methotrexate-induced lymphoma.
Individuals with rheumatoid arthritis–associated interstitial lung disease (RA-ILD) are often asymptomatic. Among patients with joint symptoms for less than 2 years (early rheumatoid disease), 58% had evidence of interstitial lung disease, but only 14% of those patients were clinically symptomatic. In an autopsy study of long-standing RA in 81 patients, only 35% had evidence of RA-ILD, but the cause of death was respiratory failure related to that ILD in 9%. Patients with a definite or probable usual interstitial pneumonia (UIP) pattern of lung fibrosis of RA-ILD have a shorter survival than those with nonspecific interstitial pneumonia (NSIP) pattern. However, regardless of HRCT pattern, a lower forced vital capacity or decline in forced vital capacity is independently associated with risk of death.
Clinical Presentation
RA is manifested insidiously as arthralgia, joint swelling, and stiffness. Pleural effusions and pleuritis are the most common pulmonary manifestations of RA. Approximately 20% of patients have symptomatic pleural disease, with the majority having “mild pleurisy.” Pleural effusions are more frequent in middle-aged men with RA and typically occur during episodes of active arthritis. In RA pleural effusions can resolve over a period of weeks to months, but they may persist for years. Although RF may be present in the exudative effusion, it is a nonspecific finding that can also be present in effusions caused by systemic lupus erythematosus, tuberculosis, and malignancy. The most frequent respiratory symptoms in RA are exertional dyspnea or cough; airway disease may be manifested as wheezing but can be asymptomatic.
Pathophysiology
Pathology
More than 20 pathologic processes have been described in the lung parenchyma of RA patients, and they can occur synchronously, with up to six different pathologic patterns reported in the same lung biopsy specimen (personal communication with T.V. Colby). Separating subclinical disease from symptomatic disease is an important clinical distinction because pulmonary complications are a common cause of death in patients with RA.
No large series have correlated the histopathologic and HRCT appearances in patients with CTDs, and in general, some extrapolation is needed from series that characterize the appearances of the individual idiopathic ILDs. Pulmonary involvement may precede the articular manifestations of disease.
In a study of surgical lung biopsy results in patients with various CTDs, the majority of those with RA had a histologic diagnosis of NSIP, and the rest had follicular bronchiolitis, UIP, or organizing pneumonia. There is a selection bias in patients who come to surgical biopsy, and UIP is likely underrepresented because patients with a confident HRCT diagnosis of UIP are not generally referred for biopsy. This perhaps explains why UIP is usually thought to be the predominant radiologic pattern in RA, whereas biopsy studies generally favor NSIP.
Although pulmonary rheumatoid (necrobiotic) nodules are frequently mentioned in the literature, they appear to be relatively rare in clinical practice, and in one autopsy study of nearly 1000 patients, no examples were documented. However, HRCT studies have reported pulmonary nodules in up to 28% of cases, but because these nodules do not have a pathologic correlate, the exact nature of the nodules is not known. Pulmonary rheumatoid nodules can vary in size from 0.5 to 7 cm and are pathologically identical to subcutaneous rheumatoid nodules ( Fig. 39.1 ). Rheumatoid nodules contain a necrotic center surrounded by palisaded histiocytes and leukocytes. The occurrence of pulmonary rheumatoid nodules can antedate the arthritis, and although nodules are generally asymptomatic, they can cavitate and result in catastrophic hemoptysis or pneumothorax. Pulmonary nodules are usually found in patients with subcutaneous nodules and positive RF, and they can occasionally regress and disappear spontaneously. Because patients with RA are frequently immunosuppressed, cavitating nodules can become infected. Caplan syndrome represents an association of pulmonary nodules and pneumoconiosis in patients with RA ; it was initially described in coal miners, but similar syndromes have been reported in foundry workers, roof tile makers, and individuals exposed to various mineral dusts.

Follicular bronchiolitis is characterized by lymphoid hyperplasia of bronchus-associated lymphoid tissue, with the follicles distributed along the bronchioles ( Fig. 39.2 ). In one series the majority of patients with biopsy-proven follicular bronchiolitis had RA, and it is important to remember there is considerable overlap between lymphocytic interstitial pneumonia (LIP) and follicular bronchiolitis. The two conditions are distinguished pathologically by expansion of the interstitium in LIP versus the degree to which mononuclear cells are concentrated around the airways in follicular bronchiolitis.

There have been a few reports of a triad of recurrent alveolar hemorrhage, crescentic immune complex–mediated glomerulonephritis, and RA. These patients have nail bed hemorrhage and necrotizing vasculitis. In an attempt to characterize the vasculopathy seen in some patients with RA, a survey of rheumatologists revealed that most practitioners see fewer than five patients with RA vasculitis annually. Pulmonary vascular involvement is infrequently recognized either clinically or pathologically.
Pulmonary amyloid deposition in patients with RA is rare, but there are isolated case reports, including a patient with diffuse septal pulmonary amyloidosis. Amyloidosis, follicular bronchiolitis, and smoking-related ILD are all potentially responsible for the small pulmonary nodules that are so frequently seen on HRCT.
Lung Function
Patients with RA frequently have abnormal pulmonary function test (PFT) findings as a result of the various pathologic processes. There are several studies that describe a range of PFT abnormalities, but these studies often contain cigarette smokers. A study of PFT abnormalities in lifelong nonsmokers who had significant RA (defined by treatment of the disease with an antirheumatic drug) but no clinical evidence of ILD found that more than a quarter of this cohort had abnormal PFT results but no pulmonary symptoms, and these abnormalities were both restrictive and obstructive. The mixture of an obstructive or restrictive picture is not surprising in view of the range of pulmonary diseases encountered in RA. There is good correlation between evidence of air-trapping on HRCT and functional indices of early airway obstruction in patients with RA. Subtle changes in PFT values can alert the clinician to occult disease, and when the patient has mild pulmonary hypertension, diffusing lung capacity for carbon monoxide (DLCO) can be reduced in the presence of normal HRCT findings. Lung function tests may have an important role in monitoring disease progression; however, joint disease can limit a patient’s ability to exercise, and therefore altered exercise tolerance cannot always be used as an indicator of clinical deterioration, and the debility caused by RA itself may limit patients’ ability to perform lung function testing.
Manifestations of the Disease
In most patients with RA the onset of disease is insidious, but in a fifth of patients the joint involvement has a more acute onset. Joint symptoms include pain, swelling, and stiffness, with the number of involved joints increasing over a period of weeks to months. Approximately 10% of patients have a fulminant onset, and a few start with episodic symptoms, which then progress to persistent disease. The arthropathy in RA typically involves the small joints of the hands and feet and then progresses to large joints in a symmetric distribution. There is abnormal proliferation of the synovium of joints and resultant pannus. In chronic disease deformities of the hands can be very severe. The cervical spine is often involved in patients with RA, and subluxation or impaction of C1 on C2 is a frequent complication.
The wide range of extraarticular manifestations in RA is summarized in Table 39.1 .
| Dermatologic | Palmar erythema, subcutaneous rheumatoid nodules, vasculitis |
| Ocular | Keratoconjunctivitis sicca, episcleritis, scleritis, choroiditis |
| Cardiac | Pericarditis, myocarditis, coronary vasculitis, valvular nodules |
| Neuromuscular | Entrapment neuropathy, peripheral neuropathy, mononeuritis multiplex |
| Hematologic | Felty syndrome (rheumatoid arthritis, splenomegaly, and leukopenia), large granular lymphocyte syndrome, lymphomas |
| General | Fever, lymphadenopathy, weight loss, fatigue |
| Other | Amyloidosis, secondary Sjögren syndrome |
Radiography
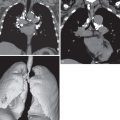
The radiographic manifestations of pulmonary disease in RA patients can be broadly divided into pleural and parenchymal involvement. Pleural disease reflects the “polyserositis,” and thus pericardial effusions are a frequent accompaniment. Pleural effusions tend to be asymptomatic and small to moderate in size. Radiographic evidence of a pleural effusion has been reported in 1% of all RA cases. The effusions are generally unilateral but can be bilateral, and on chest radiography, 20% of patients with RA demonstrate pleural thickening ( Fig. 39.3 ). Long-standing effusions may become loculated, simulating empyema despite sterility. Rheumatoid pleural involvement can result in thickening of both parietal and visceral pleura, resulting in pleural fibrosis and trapped lung. Core biopsy of this thickening typically shows nonspecific inflammatory changes rather than rheumatoid granulomas or malignancy.

A readily recognizable but rare form of lung involvement on chest radiography is the presence of cavitating necrobiotic nodules ( Fig. 39.4 ). The plethora of ILDs encountered in patients with RA are generally nonspecific; furthermore, some of these diseases may not be readily detected by chest radiography (e.g., follicular bronchiolitis). The relative insensitivity of chest radiography is especially evident in patients with constrictive bronchiolitis; even in individuals with functionally severe airflow limitation, the chest radiograph may be normal.

The classic radiologic pattern of pulmonary fibrosis in RA is a reticular or reticulonodular pattern ( Fig. 39.5 ). There may be evidence of honeycombing, and the disease is usually symmetric and basal in distribution. Diffuse reticulonodular fibrosis is seen in 5% of all RA patients, and pulmonary fibrosis is more frequently found in those with subcutaneous nodules, antinuclear antibodies, and RF titers.

Patchy consolidation may represent organizing pneumonia or infection. Organizing pneumonia typically shows changing, bilateral peripheral consolidation on chest radiography ; however, infection, drug-induced lung disease, and malignancy are differential diagnoses.
In a prospective study of patients with early RA (preceding chest symptoms), chest radiographs showed ILD in 6% of patients, whereas HRCT showed ILD in 33% of patients. Chest radiography has been reported to have a sensitivity of 23% for “fibrosing alveolitis” in RA, which implies that chest radiography is not good at detecting fibrotic lung disease in patients with RA.
Computed Tomography
HRCT is particularly helpful in the context of RA-ILD because it often shows more than one pulmonary disease, which may not be evident on chest radiography. Diagnostic precision in RA-ILD is challenging as a result of (1) the wide range of potential simultaneous pathologies, (2) the possibility of a concurrent CTD, and (3) the need to determine the significance of limited pulmonary disease. Further possible diagnostic confounders include the presence of smoking or drug-related disease and immunosuppression-related infection. Because the reported lifelong prevalence of significant lung disease has not been higher than 20% in any series of RA patients, it seems that HRCT may be detecting truly subclinical pulmonary disease. RA patients with pulmonary symptoms have bronchiectasis, pulmonary nodules, and areas of ground-glass opacity and honeycombing on HRCT. These same findings, with the exception of honeycombing, are seen to a lesser degree in RA patients without respiratory symptoms. See Table 39.2 for a summary of HRCT findings and patterns in connective tissue disease, including RA.
| Pathologic Diagnosis | High-Resolution Computed Tomography Appearance |
|---|---|
| Nonspecific interstitial pneumonia (fibrotic subtype) | Predominantly ground-glass opacity with underlying distortion and fine reticulation, which may or may not be subpleural and basal |
| Usual interstitial pneumonia | Reticulation with honeycombing in a peripheral and basal distribution. The disease tends to creep anteriorly and subpleurally in the upper lobes |
| Organizing pneumonia | Consolidation that is most commonly peripheral or peribronchovascular; often around the perimeter of the secondary pulmonary lobules (perilobular distribution) |
| Respiratory bronchiolitis–associated interstitial lung disease | Indistinct centrilobular nodules and patchy ground-glass opacities; minor lobular air trapping |
| Desquamative interstitial pneumonia | Usually random distribution of extensive ground-glass opacity; sometimes small cysts |
| Diffuse alveolar damage | Widespread ground-glass opacity and consolidation |
| Lymphoid interstitial pneumonia | Ground-glass opacity and/or nodules; cysts in approximately 50% of cases |
| Follicular bronchiolitis | Small centrilobular/peribronchial nodules and ground-glass opacity |
| Constrictive bronchiolitis | Mosaic attenuation, attenuated pulmonary vessels in areas of decreased lung density, and abnormalities of the large airways (bronchial dilation and/or wall thickening) |
| Lymphoproliferative disease | Consolidation, masses, nodules, or ground-glass opacity; thickened interlobular septa may be seen |
| Amyloid | Variable appearances; bronchovascular structures may be infiltrated. Reticulonodular/linear pattern in the lungs. Large nodules may calcify. |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree