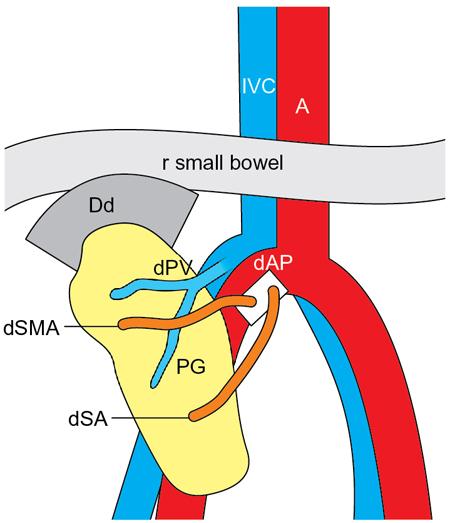
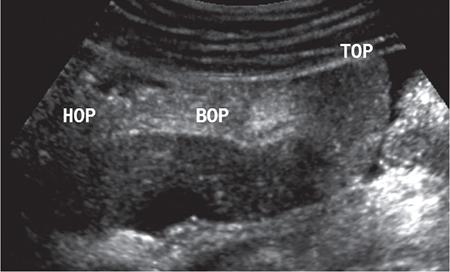
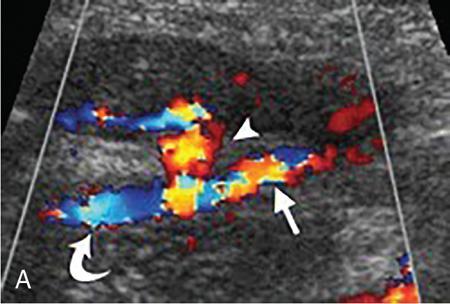
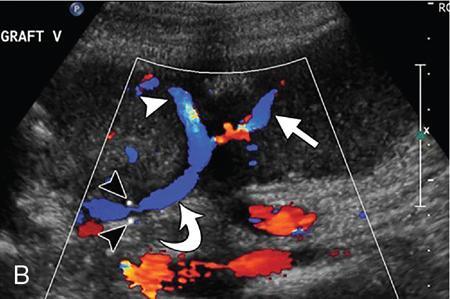
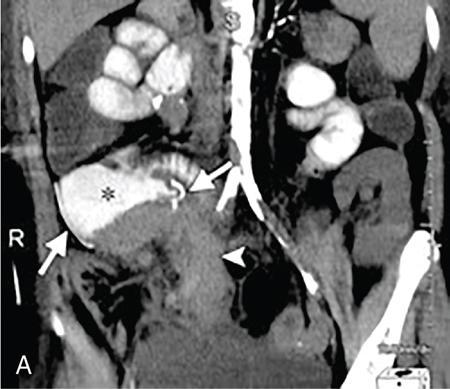
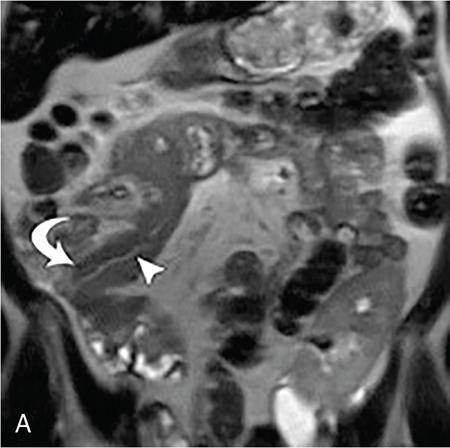
Kajal Patel, L. Murali Krishna Pancreatic transplantation is currently the only effective treatment for type 1 diabetes mellitus (93% of cases) allowing long-term glycaemic control without exogenous insulin injections. Additionally, it also counteracts the complications of diabetes mellitus, including diabetic neuropathy, retinopathy, vasculopathy and end-stage renal failure. The standard procedure is transplantation of a whole-organ cadaveric pancreas but less-invasive approaches such as islet cell transplantation are in development. Simultaneous pancreas–kidney (SPK) transplantation (78%) is done in most of the cases with same donor. In 16% cases, pancreas transplant can be done after kidney transplant also. But rarely, isolated pancreatic transplant (6%) can be done. The first pancreatic transplantation was performed in 1966 by the team of Dr. Kelly, Dr. Lillehei, Dr. Merkel, Dr. Idezuki Y and Dr. Goetz, 3 years after the first kidney transplantation. According to Scientific Registry of the United Network for Organ Sharing (UNOS) and the International Pancreas Transplant Registry (IPTR), the national 1-year patient, kidney and pancreas survival rates for recipients of an SPK transplant are 95%, 89% and 85%, respectively. Pancreas grafts have vascular and enteric connections that vary in their anatomic approach and understanding of this is critical for imaging. Imaging techniques are directed to display the pancreatic transplant arterial and venous vasculature, parenchyma and intestinal drainage pathway. Various surgical techniques have evolved over the years especially in the recent decade. Imaging of these patients should also be done based in the common surgical techniques and complications that arise of these surgeries. Though multiple modalities like ultrasonography (US), computed tomography (CT) and magnetic resonance (MR) imaging, radionuclide study and digital subtraction angiography (DSA) are available, each of them have their own advantages and limitations and usually a multimodality approach is required to diagnose the spectrum of complications. Preoperative evaluation typically considers the potential recipient’s age and ability to survive the operation. A relative contra-indication is the patient’s age (>55 years), due to the fact that the success rate is superior in younger individuals. Key components of the assessment include determining the presence of renal, cardiac, peripheral vascular, cerebro-vascular and psychiatric diseases. The pretransplant work-up consists of extensive laboratory, infectious and physiologic testing. Chest radiography is required for preoperative fitness of recipient. The extent of aorto-iliac calcification of recipients, a factor in choice of implant site, is evaluated with unenhanced CT. Donor factors such as age, sex, body mass index, cause of death, donation after cardiac death, serum creatinine and preservation time (cold ischaemia) can influence the outcome of pancreas transplantation. Usually, evaluation of donor pancreas is best done by the pancreatic transplant surgeon intraoperatively. Visual inspection of pancreas in terms of its size, texture, colour, fibrosis, fatty infiltration and its vascular supply is essential. There is very little role for preoperative imaging in the setting of deceased organ donation. The donor’s pancreas is harvested en bloc with its respective vascular support and a variable duodenal segment that contains the ampulla of Vater. The most common technique consists placing the pancreatic graft intraperitoneally in the right pelvic region with the duodenal segment facing cephalad and the renal graft in the left iliac fossa, extraperitoneally. Whole pancreatic graft transplantation can be performed with a duodenal segment; in this type of transplantation, donor’s duodenum is anastomosed with the recipient’s small bowel loop for enteric exocrine drainage and grafted portal vein is anastomosed with common iliac vein or inferior vena cava for systemic endocrine drainage (Fig. 9.20.1). Another way of restoring the endocrine drainage, grafted portal vein may be anastomosed with the recipient’s portal venous system and for exocrine drainage duodenal segment may be anastomosed with the urinary bladder. Duodenoenterostomy done by side to side anastomosis of donor’s duodenal segment to the recipient’s small bowel loop. Arterial supply is established by using the donor’s aortic patch, containing the splenic artery and the superior mesenteric artery (SMA), which is anastomosed to the recipient’s common or external iliac artery. Native pancreas of patient is left untouched in the upper abdomen. After placement of pancreatic graft by surgeon, pancreatic graft Doppler evaluation should be done intraoperatively. Intraoperative ultrasound probe is directly put on anastomotic artery and vein which show normal colour flow without evidence of thrombosis. In the case of occlusion, there is no evidence of colour flow. In the case of occlusion, surgeon does re-anastomosis of vessels once thrombi are removed. Due to its superficial location in pelvis and visualization of pulsation of vessels, visual inspection is sufficient for patency of vessels. So most of the time intra-operative Doppler study is not necessary. Postoperative imaging of pancreas transplantation is a challenge for the radiologist because of the altered surgical anatomy, identifying the pancreatic graft from adjacent structures and various postoperative complications that may arise posttransplantation. Imaging evaluation of the pancreas transplant grafts is commonly performed by a multitechnique approach. The most commonly utilized scanning techniques include US, CT and MR imaging. DSA and radionuclide study are routinely not performed nowadays. Ultrasound usually represents the first line imaging method in the assessment of the pancreatic graft, due to its portability, repeatability for ill and unstable patients in the immediate postoperative period, lack of ionizing radiation, and it provides a real-time vascular flow map which may allow detection of vascular anastomotic stenosis and reduced pancreatic graft perfusion. Its evaluation may, however, be limited due to the intraperitoneal position of the pancreas graft, in particular with the portal enteric approach with the organ in the right upper abdomen and intestinal gas overlap. Unless abnormally dilated, the duodenal component often cannot be separately evaluated by ultrasound. Additionally, ultrasound may be fundamental in guiding the percutaneous biopsy. Although the lack of an organ capsule generally results in an ill-defined appearance, the pancreatic transplant can be identified by its relatively cylindrical shape. In greyscale B-mode, the normal pancreatic graft presents homogeneous echotexture, lower than the native pancreas and the surrounding mesenteric or epiploic fatty tissue (Fig. 9.20.2). Colour and power Doppler US play a vital role in demonstrating pancreas transplant perfusion and vascular anatomy. We would also be able to visualize the Y arterial graft, graft vein, splenic artery and vein (Fig. 9.20.3A and B). Venous structures demonstrate a monophasic waveform within an anechoic lumen and velocities ranging between 10 and 60 cm/s. Normal arterial waveform exhibits a sharp systolic upstroke and a continuous diastolic flow. In the immediate postoperative period, the velocities of the arterial anastomosis may be very high as 400 cm/s due to possible postoperative oedema and/or due to kinking of the anastomosis. Usually in both the cases, the arterial anastomotic velocities gradually decrease on in follow-up. The resistive index (RI) may be of limited use to diagnose graft rejection, as the values may be as high as 0.9 and are variable throughout the gland. Due to the presence of renal capsule in a transplanted kidney, there is elevated vascular resistance when there is intrarenal oedema; however, due to the absence of capsule in the transplanted pancreas, the vascular resistance will be normal in spite of oedema secondary to pancreatitis or rejection. CT is generally required after an abnormal ultrasound or whenever the patient presents unexplained fever, abdominal pain or when abnormal laboratory data are found. Contrast-enhanced CT helps to evaluate the graft parenchyma, the enteric and vascular anastomosis and in diagnosing postoperative complications such as focal collections, vascular thrombosis or pneumoperitoneum. In noncontrast CT scan, pancreatic graft appears as a homogeneous isodense soft tissue organ. It is more difficult to differentiate between pancreatic graft and nonopacified and nondistended small bowel loop in plain/noncontrast CT scan. But you can always make out the surgical clips which are stapled on duodenal stump, which can be helpful for localization of pancreatic graft. Nonenhanced images should be acquired with the goal of locating the graft and possible early thrombus or haematoma. The protocol used should include (positive) enteric contrast that allows identification of intestinal loops adjacent to the graft and distinction from possible liquid collections (Fig. 9.20.4A). The donor’s duodenum is frequently collapsed and may fill (or not) with the given oral contrast. IV contrast material is administered infrequently, to avoid the risk of nephrotoxicity, if native renal function is impaired. About 120–150 mL of contrast medium greater than 350 mg iodine per millilitre is injected at the rate of 4–6 mL/s. Late arterial phase can be obtained with the bolus-tracking technique located in the common iliac artery (attenuation value of 150 HU) and is very useful in the assessment of parenchymal enhancement and arterial anastomosis; 50 seconds after the administration of intravenous contrast material, the portal venous phase evaluates the respective drainage and possible associated venous complications (Fig. 9.20.4B). CT images are evaluated by axial, multiplanar reformats and three-dimensional maximum intensity projection (MIP) and volume-rendered techniques. The normal pancreatic parenchyma will enhance uniformly more in the arterial phase than in the venous phase. The iliac arterial graft, peripancreatic and intrapancreatic arterial vasculature, as well as the anastomosis of the donor portal vein to the recipient iliac or superior mesenteric vein, should be delineated. Coronal reformats are the best to illustrate the intestinal anastomosis and to determine if the graft is placed inferiorly for a systemic venous drainage or superiorly for a portal venous drainage. MR is usually indicated in young patients in which cumulative radiation is an essential consideration. Contrast-enhanced MR angiography is used for evaluating the arterial and venous anatomy of pancreatic graft; however, it is difficult to access the enteric anastomosis and postoperative complications due to low spatial resolution. Also, it is a challenge to image sick patients requiring intense monitoring and those with metallic clips. An appropriate protocol includes axial T1-weighted (T1WI) (precontrast; in-phase and out-of-phase), coronal T2-weighted (T2WI) fast-spin-echo and axial T2-weighted fat-suppressed sequences. Furthermore, images after intravenous contrast administration (gadolinium-based) should be acquired in arterial and venous phases. Unenhanced MR imaging readily helps distinguish the pancreatic allograft from adjacent structures and is superior to CT without intravenous contrast material. In plain MRI of abdomen, pancreatic graft appears hyperintense as compared to liver and appears as homogenous structure on T1-weighted images. Normal pancreatic graft’s signal intensity is between that of fluid and muscle on T2-weighted images. Various pathologic process of graft pancreas is more related to increased glandular water content, so T2-weighted images are more sensitive to diagnose graft pathology (Fig. 9.20.5A). Axial and coronal images are useful in displaying pancreatic and peripancreatic graft oedema, as occurs in pancreatitis, and in characterizing peritransplant fluid collections (haematoma/seroma). The MR angiography helps to access the arterial and venous anatomy and can diagnose arterial or venous stenosis and venous thrombosis. The normal pancreas graft enhances briskly and homogeneously in the late arterial phase (Fig. 9.20.5B). Gadolinium-based contrast agents may pose the risk of nephrogenic systemic fibrosis in selected patients with advanced renal dysfunction. The normal pancreatic duct is generally not visible with US or CT but may be observed as a thin (≤3 mm) smooth line on T2W MRI.
9.20: Role of imaging in pancreatic transplant
Introduction
Goals of pancreatic transplantation
Types of pancreatic transplantation

History
Pretransplant workup
Assessment, procurement and implantation of the pancreatic graft for transplantation
Assessment
Procurement and implantation
Intraoperative imaging of pancreatic graft
Posttransplant pancreatic graft imaging
Ultrasound
Computed tomography

Magnetic resonance imaging and magnetic resonance angiography

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








