SMALL BOWEL IMAGING Poonam Narang, Himanshu Gupta The small bowel is a long tubular hollow abdominal viscus thrown into intricate folds and loops and packed into the abdominal cavity. It is approximately 5–7 m in length and extends from the gastric pylorus up to the ileo-caecal valve. It is anatomically divided into three parts: duodenum (meaning ‘in twelves’ as its length is roughly equal to the width of 12 fingers), jejunum (meaning ‘fasting’ as it was usually found empty after death) and ileum. The duodenum measures 20–25 cm in length and extends from gastric pylorus to the ligament of Trietz. It is a fixed ‘C’-shaped structure that cradles pancreatic head in its concavity. First part of the duodenum is a triangular conical structure with its base communicating with gastric antrum. Its apex points to the right and cranially, in close apposition with the gall bladder and the under surface of the liver. Second part of duodenum is almost vertical, lying in the right paravertebral gutter. The common bile duct courses obliquely posterior to it and drains at a papilla at its medial wall (the Ampula of Vater), after joining with the main pancreatic duct. Third part is horizontal, crossing the midline between the superior mesenteric artery (SMA) and aorta from right to left, caudal to pancreatic neck. Fourth part is again almost vertical, ascending left of the spine almost to the level of duodenal bulb and transitions to jejunum at duodenojejunal (DJ) flexure. Duodenum is mostly a retroperitoneal structure while the rest of the small bowel acquires a mesentery at the DJ flexure. The jejunum and ileum are intraperitoneal structures, suspended in the abdominal cavity from the posterior abdominal wall by the free end of their ‘fan-shaped’ mesentery and are thus relatively mobile. Though considered a part of small bowel, duodenum differs from the rest of the small bowel in structure as well as function. Plethora of the diseases that affect duodenum are distinct. Peptic ulcer disease affecting the first part does not affect the rest of the small bowel. Most other diseases affecting the duodenum arise from the surrounding structures like pancreas, gall bladder, bile duct, liver and the hepatic flexure of colon. Hence, on imaging, duodenal abnormalities should prompt a search for pathologies of the surrounding organs. While duodenum is structurally a distinct segment, the structural transition from jejunum to ileum is seamless, with no external demarcating point. Histologically, and also on imaging, all three segments have clear identities. Their mucosal lining, folds and presence of submucosal lymphoid follicles are allocated in accordance with their specific functions. Jejunal loops occupy left hemiabdomen and ileal loops, with ileo-caecal junction, lie in the right lower abdomen. Jejunum is about two-fifths of the length of the small bowel, and the ilium forms the rest of the three-fifths of the entire 5–7 m. ‘Terminal ileum’ is a term arbitrarily but commonly used in clinical practice to denote the portion of ileum just proximal to ileo-caecal junction harbouring abundant lymphoid tissue in its walls and playing host to a number of pathologies. At ileo-caecal junction, the ileum joins the caecum obliquely. A valve-like mucosal fold regulates the flow of digested food into the colon and prevents its reflux back. Its efficiency in preventing reflux is quite inconsistent in a large percentage of subjects. The arterial supply of the duodenum is by the branches of hepatic artery (branch of Celiac Axis artery), till the proximal half of second segment (marked by the Ampula of Vater); and by branches of the superior mesenteric artery beyond that point. Duodenal branches of respective superior and inferior pancreatico-duodenal arteries make an arcade along the medial margin of duodenum in the pancreatico-duodenal groove. The rest of the small bowel is supplied by the superior mesenteric artery and its branches while venous drainage is via superior mesenteric vein and its tributaries. These vessels traverse the mesentery, between the twofolds of the peritoneum from the posterior abdominal wall to reach the small bowel forming multiple arcades of vessels within the mesentery. Straight arteries, called the Vasa Recta, arise from the terminal arcade and enter the bowel wall, along its mesenteric border. Disease processes involving the specific artery affects the respective bowel segment that it supplies. Ischaemia, infarction and haemorrhage can be attributed to and localized correctly based on the relevant vascular anatomy. Inflammatory diseases cause engorgement of Vasa recta; and at times, identifying them as a lead point, one is able to localize the involved bowel segment correctly. Vascular ischaemia affects the antimesenteric part of the bowel earlier in the course of the disease. Therapeutic procedures too can be appropriately planned based on the vascular territory of the affected bowel segment. The mesentery forms a fan-shaped suspensory support, tethering the small bowel to the posterior abdominal wall, along a line running obliquely from ligament of Trietz (in LUQ) to ileo-caecal junction close to superior part of right sacro-iliac joint (in RLQ). This ‘root of mesentery’ averages about 15–20 cm in length. It is closely wrapped around small bowel loops and merges with the serosa. All lymphatic and vascular channels supplying to and draining from the small bowel traverse within the mesentery. Thus, pathology originating in one often affects the other by contiguity, haemodynamic alteration or haemo-lymphatic spread. The normal mesentery sandwiches a layer of fat between its two leaves. On imaging, it provides a good background against which identification of pathologies of fluid density (oedema) or soft tissue density (phlegmon, lymph nodes, masses) can be made. Mesenteric oedema often becomes the smoke leading up to the fire, for example: focal oedema adjacent to the site of an otherwise invisible bowel wall perforation. On ultrasound, such tell-tale inflammation is seen as an increase in the echogenicity of mesenteric fat adjacent to the site of bowel pathology. On CT, the hypodense fat becomes hyperdense whenever any pathological change occurs. Delineation of mesenteric vessels is also facilitated due to surrounding hypodense fat. Engorged Vasa recta are easy to identify on imaging as they stand out against the surrounding fat in the mesentery. Similar imaging findings can be extrapolated to MR as well. Similar to the rest of the gastrointestinal tract, the small bowel wall comprises four layers (strata) – from within outwards being mucosa, submucosa, muscularis propria and serosa. Mucosal folds (Valvulae Conniventes) and further finger-like projections of mucosa (Villi) help dramatically increase the surface area for absorption. Only valvulae conniventes are seen radiologically as concentric rings measuring 2–3 mm. The bowel wall measures 1–2 mm when distended and 2–3 mm when collapsed symmetrically in its entire circumference. Increased bowel wall thickness (in the background of adequate luminal distension) is a commonly used marker of pathology. Understanding how different pathologies target specific sites within the micro-structure of bowel wall, along with length of bowel involvement, could be helpful in drawing conclusion regarding the etiopathogenesis. For example, short segmental disruption of the entire thickness of bowel wall favours a unifocal, infiltrative pathology-like malignancy. Long segment submucosal oedema with preserved mucosal integrity/enhancement may be a haemodynamic phenomenon as in portal enteropathy or reactive as in infective enteritis. Depth and invasiveness of a disease process can be monitored for activity/progression, especially on imaging modalities like ultrasonography and MRI. The innermost anatomic layer of small bowel wall, its mucosa forms the absorptive surface which transports nutrients from the lumen into blood/lymph. Pathologies affecting the mucosa cause reduction (shortening) in the available area of absorption – the so-called short bowel syndrome. A variety of metabolic derangements can accompany the nutritional deficiencies caused thereby. Many other inflammatory and infiltrative diseases like lymphomas cause various alterations in the appearances of mucosal folds. Reduction, blunting, flattening, thickening, clubbing, nodularity, polypoid formation, etc. are various imaging appearances of the disease processes affecting the mucosal surface (Figs. 8.3.1.1–8.3.1.3). These abnormalities can be local, segmental, regional or generalized; involving the entire circumference, mesenteric or antimesenteric regions. The radiologist’s role thus lies in identifying the presence, location and extent of mucosal abnormality in addition to suggesting a possible aetiology based on the specific clinical settings. Small bowel has no sensory or motor nerve supply and is regulated by autonomic nervous system for its motility, secretions and absorption. Branches from vagus nerves and from dorsal sympathetic ganglion also traverse the mesentery to innervate the bowel wall at the mesenteric border. During the third week of intrauterine life, the process of ‘gastrulation’ results in formation of three basic layers, namely ectoderm, mesoderm and endoderm, which eventually evolve and differentiate into tissues and organs of all systems. Bowel, along with hepatobiliary and pancreatic system, develops from the ‘primitive gut tube’ that is formed when the endoderm folds upon itself. It then differentiates into three sections: foregut, midgut and hindgut. By 4–6 weeks, the rapidly growing intraabdominal organs outgrow the coelomic cavity. The midgut undergoes physiological umbilical herniation and rotation and then herniates back into the abdomen by the 10th week. Subsequently, the developed midgut organs undergo peritoneal incorporation and fixation to occupy their respective intraabdominal positions. This process takes place in three stages, and interruption at each stage results in specific anomalies of malrotation and peritoneal fixation. During the first stage, from week 6 to week 10, the midgut herniates ventrally into the umbilical cord in a vertical loop configuration. This loop has the superior mesenteric artery in the centre dividing the midgut into a prearterial (jejuno-ileal) segment superiorly and a postarterial (caeco-colic) segment inferiorly. This loop rotates 90 degree counter-clockwise, with superior mesenteric artery as its axis. The prearterial segment comes to lie to the right and the postarterial segment to the left in a horizontal plane. The second stage, from week 10 to week 12, results in a further 90-degree counter-clockwise rotation, and the small bowel loops now are positioned under the postarterial segment. By the end of the 10th week, the abdominal cavity grows large enough to accommodate the entire length of the foetal bowel. As the bowel loops now return to the abdominal cavity, the small bowel returns first followed by the large bowel. Subsequently, the small bowel loops make their final 90-degree counter-clockwise rotation, bringing the D-J junction to the left of the spine, while the colon makes a 180-degree rotation in the same direction to place the caecum in the right lower quadrant of the abdomen. Thus, by the end of stage II, the midgut derivatives have undergone a total 270-degree rotation (including the initial 90-degree rotation) with DJ junction to the left of midline, caecum in the right lower quadrant and transverse colon crossing in front of the duodeno-jejunal junction. Vitello-intestinal duct (VID) gets obliterated by the ninth week. The third stage (from 12 weeks until term) involves fixation of the mesenteries to the posterior abdomen. The completed normal rotation of the bowel produces a long mesenteric attachment for small bowel, extending from ligament of Treitz at the D-J junction in left upper quadrant to the ileo-caecal valve in right inferior quadrant known as the ‘root of mesentery’. The dorsal mesentery of some portions of the gut, like pancreas, duodenum, ascending and descending colon, becomes incorporated into the posterior abdominal wall, making these segments secondarily retroperitoneal (Figs. 8.3.1.4–8.3.1.6). Understanding of chronological embryology helps one to interpret respective imaging features of developmental anomalies and also to look for their associations. During imaging of a suspected case of malrotation, position of DJ flexure, ileo-caecal junction, relative location of jejunal and ileal loops and relation of superior mesenteric artery to the accompanying vein help in reaching the correct conclusion. It should be kept in mind that certain diseases (notably the celiac disease and other malabsorption syndromes) may invert the imaging morphology of jejunum and ileum leading to erroneous diagnosis. Evaluation of orientation of superior mesenteric vessels and the duodeno-jejunal junction would help resolve any ambiguity in such cases (Figs. 8.3.1.7 and 8.3.1.8). Developmental malrotation is often a precursor to small bowel volvulus, or obstruction by extrinsic bands in neonates and infants. In adults, on the other hand, often the malrotations are by themselves, asymptomatic. They get identified only incidentally, or while looking for symptoms due to their associations. Preduodenal peritoneal bands, commonly called as the Ladd’s bands, causing external compressions resulting in bowel obstruction are less common. Often, peritoneal fenestrations and deep fossae, associated with developmental midgut malrotations, present with internal bowel herniations like various para-duodenal and mesenteric herniations. One must be aware of their associations, so as to make the correct interpretation. Internal hernias may be a cause of intermittent postprandial abdominal pain, when reducible; or may result in acute bowel obstruction. They are identified by abnormal location and clumping together of the relatively mobile small bowel loops. High degree of suspicion and knowledge about their locations, helps one to diagnose them with more confidence. Occasionally, constrictions at the neck of hernia can also be identified. Fluoroscopic examination, with palpation to separate the opacified loops, provides the most confident method of their identification. The origin of clinical abdominal radiography dates back to 1896–97, soon after the discovery of X-rays by Wilhelm Roentgen in 1895, when E. Lindemann used X-rays to demonstrate gastroptosis and Walter B. Cannon used them to describe basic physiology of swallowing and peristalsis. The biggest shortcoming with plain abdominal radiographs was lack of inherent contrast with only occasional visualization of air-filled stomach. H. Rieder introduced the concept of contrast abdominal radiographs by giving patients large amounts of radio-opaque bismuth orally, followed by rapid serial abdominal radiographs for better luminal visualization. Later, C. Bachem and H. Gunther introduced barium as inert and nontoxic contrast agent for gastrointestinal imaging. Since their inception, conventional imaging techniques, like plain abdominal radiograph, fluoroscopy and single contrast barium studies, formed the cornerstone of abdominal imaging despite their limitations. In the mid-20th century, with the availability of better barium preparations and advancements in fluoroscopy and radiographic equipment, such as tilt tables, better films and image intensifiers, double contrast barium studies became the problem-solving technique due to its unparalleled ability to image gastrointestinal mucosa meticulously. This enabled the mucosal morphology to be imaged in detail, thus aiding detection of even minute mucosal lesions such as small ulcerations and early carcinomas. With the invention of flexible endoscopes in the late 20th century, UGI endoscopy and colonoscopy replaced abdominal radiography as the frontline investigation for evaluation of the upper GI tract (oesophagus, stomach and duodenum) and colon, respectively, with added advantage of direct lesion visualization and obtaining biopsy. Owing to its length and complex folded anatomy, small bowel is still inaccessible via flexible endoscopes except for a few centimetres of proximal jejunum and terminal ileum. One of the major drawbacks of barium studies and endoscopy was that they provided solely luminal details with lack of information about mural and extramural structures. This ushered the era of modern imaging modalities with cross-sectional capabilities like US, CT and MR. With ongoing advancements in equipment, imaging techniques and contrast media, these modalities are at the forefront of the modern GI imaging, offering better and detailed answers to clinical questions being asked. With recent introduction of capsule endoscopy, gastroenterologists are trying to find a place in noninvasive evaluation of small bowel, but it still has a long way to go. Till then, small bowel evaluation continues to be the radiologist’s domain. Imaging of the small bowel has traditionally played an important role in clinching a diagnosis due to its relative inaccessibility for direct endoscopy. Even with the advent of capsule endoscopy for mucosal evaluation, imaging retains its position in the diagnostic algorithm for its ability to evaluate full thickness of bowel wall and extraluminal structures, in addition to providing information regarding its lumen. Imaging also provides information about the bowel motility, directly or indirectly. Ultrasound and fluoroscopy provide real time visualization of peristalsis. CT scan with oral contrast may be used to monitor transit in certain specific situations, for example in postoperative settings, to differentiate paralytic ileus from mechanical obstruction. Small bowel anatomy as well as pathology can be assessed and compared on various imaging modalities under the following headings: Plain abdominal radiographs have a limited role in modern small bowel imaging. It gives relatively little diagnostic yield, and most modern departments have easy availability of US, CT and MRI. In setting of acute abdomen, plain abdominal radiographs have greatest value in evaluation of pneumoperitoneum; gas patterns and air-fluid levels in bowel obstruction; enteroliths; radio-opaque foreign bodies; and to a limited extent, in the evaluation of bowel wall pattern in cases of bowel ischaemia/colitis. A good supine abdominal radiograph helps to evaluate gas pattern and bowel calibre. The kilo-voltage should be low, preferably 60–75 kV, to maximize contrast. Exposure time must be short to minimize motion blur. It should cover the entire abdomen from domes of diaphragm to the inferior border of pubic symphysis, including obturator foramina (hernial orifices). The X-ray beam should be centred at the level of iliac crests with proper collimation. An adequate erect abdominal radiograph should suffice to demonstrate air-fluid levels and free peritoneal gas in cases of pneumoperitoneum, with erect chest x-ray (CXR), and left lateral decubitus radiographs being reserved for suspicious cases of small pneumoperitoneum. It is advisable to keep the patient in an erect position for at least 10–12 minutes before taking erect CXR or erect abdominal radiograph and in left lateral decubitus position before taking left lateral decubitus radiograph to allow free air time to rise to the highest point. Erect chest radiographs are more sensitive and reliable due to better visualization of free air forming sharp margins with diaphragm and liver surface, respectively. Normally, small amount of intraluminal gas is seen in nondilated small bowel loops. Three or more air-fluid levels measuring more than 2.5 cm in width are abnormal for small bowel and indicate stasis. Increased aerophagia or laboured breathing may sometimes give a peculiar appearance on radiographs called as ‘meteorism’. The small bowel loops appear distended with air, mimicking stasis. However, due to minimal fluid content, no abnormal air-fluid level is seen on accompanying erect radiographs. However, it is now recognized that the number, distribution and length of air-fluid levels on erect abdominal radiograph cannot reliably differentiate between obstruction and ileus and can be often misleading (Figs. 8.3.1.9 and 8.3.1.10). It must be mentioned here that plain X-ray abdomen has retained its popularity in healthcare settings that lack modern infrastructure like CT scanner in the Emergency Department. A rapid, low-dose CT scan of the abdomen gives much more information with higher degree of confidence as compared with that provided by a set of plain radiographs. Barring the cost and availability, low-dose noncontrast CT scan should replace plain radiography in patients presenting with acute abdomen. The earliest enteric contrast media used in GI tract imaging was iodized oil (Lipiodol). However, due to its oily nature, it did not coat the mucosa. At present, medical grade bismuth sulphate is the contrast media of choice for GI imaging. The reasons for using barium sulphate are: Barium studies have been the most reliable conventional techniques in small bowel imaging since their inception and still remain unmatched in detecting luminal dilatation or narrowing, subtle mucosal alterations and motility disorders. One of the major inherent limitation of conventional barium studies is the lack of information regarding extraluminal/extraintestinal structures. Duodenum is usually studied along with the stomach, using high-density barium suspension. Single or double contrast technique can be used, depending on the information sought. Only in cases where duodenal obstruction, diverticulum, fistula or leak needs to be demonstrated, a single contrast study using barium or gastrografin (in suspected cases of peritoneal leak) is performed. Double contrast studies require special manoeuvres to distend the lumen with air after coating the mucosa with a layer of barium. The patient is positioned in right or left lateral- oblique position, and images are taken under flouroscopic view to demonstrate barium coated, distended and then a collapsed duodenal bulb; and C loop till the DJ flexure. Duodenal ulcers usually deform and contract the symmetrical and conical duodenal bulb. Strictures or focal narrowing of the second part of duodenum can be due to congenital or inflammatory diseases. Widening of C loop indirectly reflects enlargement of pancreatic head. A small or large diverticulum along its medial wall adjacent to Ampula of Vater is seen in many subjects and is mostly incidental. Mucosal folds of the duodenum, seen in relief, can demonstrate focal or regional inflammatory/infiltrative processes. For fluoroscopic barium studies of the jejunum and ileum, a large amount (500–600 mL) of thinner suspension of barium sulphate is given orally; and flouroscopic images with local compression are taken at intervals of 20–30 minutes, till most of the barium reaches and distends the caecum; and ileo-caecal junction is demonstrated. To improve visualization of the ICJ, caecum can be insufflated with per-rectal air. This technique is called as per-oral pneumocolon and is mostly used in countries where diseases like tuberculosis that involve the ICJ are common. Enteroclysis too can be performed to better visualize the small bowel without opacifying the stomach and duodenum. The technique also achieves a continuous, unbroken column of barium without segmentation caused by intermittent gastric emptying. A nasogastric tube of 10F is introduced over a guidewire, to position its tip just beyond the ligament of Trietz. About 1200–1500 mL of barium is pumped manually, or by an automated pump under fluoroscopic vision to opacify the entire small bowel. It may be followed by 0.5% methyl cellulose if a double contrast study is desired. The rate of instillation is roughly 80 mL/minute but is titrated under vision so as to obtain adequate distension without causing the column to break or inducing bowel atony. Multiple supine and oblique views are obtained with compression to separate out the overlapping bowel loops. The procedure is long and entails a large amount of radiation exposure, while providing limited information about the lumen, extrinsic impressions and displacements only. At present, its best use is in demonstrating postoperative small bowel obstruction and in resolving some cases where cross-sectional imaging is ambiguous in demonstrating partial obstructive lesions. The above description is now more for its historical value, though the technique is still practised in less-privileged departments across the world. Endoscopic studies have largely replaced barium studies of the duodenum; while cross-sectional imaging has taken over the prime status in imaging of the jejunum and ileum. With conventional fluoroscopic studies and CT enterography dominating the field of small bowel imaging, ultrasonography (US) has been an underrated diagnostic imaging tool in evaluating bowel pathologies up till now, with its role limited to diagnosing appendicitis, intussusception and hypertrophic pyloric stenosis. Operator-dependent nature of the modality, technical challenges of imaging a mobile structure and gaseous contents are the major reasons for its relative unpopularity. These factors make bowel US technically challenging and may lead to misinterpretation by less-experienced radiologists. Nevertheless, with constant improvements in US machines, development of technical experience and integration of clinical details for targeted assessment, US can emerge as an indispensable diagnostic tool in evaluating patients with known or suspected small bowel pathology. Conventional transabdominal US, with a 2–6 MHz curvilinear probe, should be performed initially on a fasting patient, with special attention to the region of pain or probe tenderness, to look for any extraintestinal cause that can explain the patient’s symptoms. It can also detect any intraluminal pathologies, thickened bowel loop along with secondary findings such as perienteric inflammation, lymphadenopathy, ascites, collections, masses and hepatic metastatic lesions in cases of suspected bowel malignancies. This is followed by high-frequency (5–15 MHz) assessment of nondistended bowel using linear probes in either bowel or neonatal abdomen presets. Some authors recommend using 4–10 MHz curvilinear array initially, followed by problem solving 6–12 MHz for good balance between penetration and resolution. Scanning parameters are so optimized that all layers of bowel wall are visualized. Practically it is almost impossible to scan/evaluate the entire length of small bowel by US, thus a quadrant-wise approach is more practical, wherein the abdomen is divided into four equal quadrants by two imaginary perpendicular lines passing through umbilicus. Bowel loops in each quadrant are thoroughly evaluated for any potential abnormality followed by evaluation of the ICJ, terminal ileum and as much part of distal ileum as possible. Use a graded compression technique with gentle sweeping movement of the probe to separate bowel loops and displace intraluminal air. Using optimal sensitivity and doppler settings, colour or power doppler imaging may be used to assess the mural, mesenteric or intralesional vascularity. The bowel under probe should be assessed for motility, mural thickness and gut signature. When viewed at high frequency, normal bowel wall consists of five alternating concentric rings of low and high echogenicity usually called the ‘gut signature’ – innermost lumen-mucosa interface (hyperechoic), thin hypoechoic muscularis mucosa, echogenic submucosa, followed by hypoechoic muscularis propria and outermost echogenic serosa (Fig. 8.3.1.11). US is the only imaging modality that can demonstrate these histological layers of bowel wall distinctly. In various pathological conditions, this gut signature may be preserved, exaggerated, diminished, disrupted or completely lost (Figs. 8.3.1.12 and 8.3.1.13). Disruption of the normal gut signature usually points towards a malignant or infiltrative pathology. Diseased bowel wall must be evaluated for mural and adjacent mesenteric vascularity. Usually in normal bowel wall, the doppler signals are negligible, but with introduction of low-flow imaging software, the operator must be habituated with the appearance of normal bowel vascularity on their US machines. Active inflammation and hypervascular lesions show substantially increased vascularity in the affected bowel segments. On the other hand, thickened bowel loops without doppler signals in wall and adjacent mesentery may indicate ischaemia. Colour doppler imaging may allow differentiation of active mural thickening (increased blood flow) from chronic mural thickening/fibrosis (decreased blood flow). Bowel pathology may extend and involve the perienteric mesentery, adjacent bowel loops or solid organs. One must be vigilant enough to look for mesenteric fat oedema, collection, interbowel free fluid and lymphadenopathy when inflamed bowel segment is visualized. Hydrosonography and sonoenteroclysis are useful techniques in evaluating small bowel on US. Introduction of large volumes of echo-poor liquid oral contrast (water, mannitol, polyethylene glycol, etc.) via either oral route (hydrosonography) or fluoroscopically placed naso-jejunal tube (sonoenteroclysis) results in adequate luminal distension of small bowel loops and displaces the intraluminal gas aiding better bowel visualization. Diagnostic accuracy of sonoenteroclysis is comparable to barium enteroclysis for detecting small bowel lesions. In the past few years, there has been a constant rise in the use of intravenous microbubble US contrast in organs other than the liver. EFSUMB guidelines recommend 2.5–5.0 mL of Sonovue (Bracco Imaging) for contrast-enhanced US of bowel. High-frequency (>7.5 MHz) linear array probes are used for demonstrating the differential enhancement of bowel wall layers. Following intravenous injection of US contrast material, peak arterial enhancement occurs after 30–40 seconds, followed by venous phase lasting from 40 to 120 seconds. High-end US machines can produce dynamic enhancement curves for quantitative analysis by placing region of interest over bowel wall or mass lesions. Real-time imaging capability of US allows for evaluating the bowel motility. Rhythmic, phasic contractions with slow antegrade propulsions are usually seen in the small bowel. Large migratory contractions may occur spontaneously and are usually seen after fasting. Various bowel pathologies tend to alter the rate of peristalsis. Hypoperistalsis is an indicator of unhealthy bowel and can be seen in inflammation, obstruction, ischaemia or infiltrative pathologies. High-grade obstruction usually shows fluid distended bowel loops with nonpropulsive ‘to and fro’ movement of the intraluminal contents. During evaluation of acute abdomen in paediatric patients, some small bowel pathologies get so convincingly diagnosed that this modality deserves a place in their imaging algorithm. Target sign, telescoping bowel sign or doughnut sign of intussusception can guide one to directly treat or further image to look for a lesion at lead point. Colour doppler interrogation can suggest the level of ischaemia in these loops. Similarly, bowel strangulation and ischaemia can be readily identified in a herniated bowel loop. Follow-up of Crohn’s disease for activity is routinely done by colour doppler imaging in departments where sinologists are trained for the job. SMA and celiac axis arteries can be individually interrogated in cases suspected of bowel ischaemia. This is especially useful in cases that are already diagnosed on CTA/MRA and need to be followed up noninvasively. In GI bleeding, USG can diagnose presence of portal hypertension and varices to clinch the diagnosis. A mention of the relative orientation of SMA and superior mesenteric vein (SMV) has already been made in diagnosis in cases of midgut malrotation where the association is found in about 60% of cases. Whirling and twisting of the two is seen in the case of midgut volvulus. In tropical countries, finding ascaris worms within small bowel loops on US is not uncommon. In correct clinical context, this finding can help reach a conclusive diagnosis. Endoscopic US (EUS) has rendered the GI tract and surrounding structures acquiescent to US evaluation combining the range of endoscopy with the diagnostic abilities of US. It couples a 5–12 MHz US probe with an endoscope. There are two types of echo-endoscopes: radial and linear. Radial echo-endoscopes are used in high-resolution diagnostic imaging, as they provide a 360-degree view that most operators are familiar with. Linear echo-endoscopes have limited field of view but are equipped with therapeutic-working channel facilitating image-guided tissue sampling and intervention. The proximity and high resolution of EUS allow a detailed image-based analysis of lesions in and around the accessible GI tract. The technique is best suited for hepatobiliary and pancreatic evaluation where they are apposed to the bowel wall. In small bowel imaging, their utility is limited to the duodenum and proximal jejunum. EUS can depict about nine distinct layers of bowel wall, as against the abdominal US which has a capability of resolving up to five layers. Identification of the involved layers can aid in differentiating ulcerative colitis from Crohn’s disease in selected patients. This also helps in improved analysis of tumour penetration and hence the ‘T’ staging. Submucosal tumours can be differentiated from extraintestinal indentations, and they can further be characterized as solid, cystic or vascular lesions (Fig. 8.3.1.14). Peri-lesional infiltration and nodes can be accurately assessed. Malignant GIST (gastrointestinal stromal tumor) can be differentiated from benign ones on follow-up, by their size (>3 cm), irregular margins and heterogeneity of echotexture. Identification of peri-lesional nodes permits accurate nodal staging and is useful during follow-up. The increasing demand to shift from invasive or surgical interventions to minimally invasive alternatives has driven the development of multiple EUS-guided interventions, and EUS-guided tissue sampling is perhaps the most useful contribution of this technique. EUS-guided endoscopic resection can also be done for mucosal tumours. The technique requires training and familiarity with the orientation and is usually the domain of the endoscopists. Other limitations of EUS remain the availability of the technique and the shallow depth of field of view. Cross-sectional imaging has now become the mainstay of bowel imaging as they allow visualization of the entire bowel along with luminal, mural and extramural manifestations in a single examination. With recent advancements, like superior detectors, thinner collimation and reduced scan time, MDCT (multi-detector computed tomography) has now emerged as the first line investigation for most bowel conditions because of its wide availability, rapid execution, superb spatial and temporal resolution and high-quality multiplanar reconstructions. Modern postprocessing techniques, such as volume-rendered images, shaded surface displays, virtual enteroscopy and the introduction of artificial intelligence (computer-aided detection) have increased the sensitivity of CT and enhanced the confidence of radiologists in picking up smaller lesions with greater accuracy. However, like any other imaging technique, CT has its fair share of drawbacks. High-radiation dose that CT entails is of significant concern especially in young patients or patients with chronic bowel conditions who would require multiple serial scans during their treatment course. Other demerit is poor mucosal delineation and subtle alterations in mucosal morphology, which are best demonstrated on barium studies. Types of enteric contrast agents: (Table 8.3.1.3). Collapsed bowel loops may give false impression of bowel wall thickening, thus mimicking pathologies or may hide one. Enteric contrast agents are necessary to adequately distend, otherwise, usually collapsed bowel loops, for better evaluation. Choice of enteric contrast agent depends on the information being sought. Neutral enteric contrast agents, with their inherent low CT attenuation (0–30 HU), provide better evaluation of mucosal, submucosal pathologies and bowel wall enhancement patterns and are best for routine CT enterography/enteroclysis. Positive agents, due to their high CT attenuation, obscure GI bleeds, mucosal and mural details. They have fewer indications; that is in evaluation of site of small mechanical obstruction, bowel perforation, anastomotic leaks, sinus/fistula patency, differentiating bowel from adjacent masses, interloop collections or lymphadenopathy. Negative contrast agents like carbon dioxide are being used in virtual enteroscopy. Water as enteric contrast agent is cheap, easily available, can be consumed in large quantities and better tolerated; however, due to its rapid intestinal absorption, distension achieved by water is highly variable and sometimes inadequate. Better bowel distension is achieved by mannitol and methylcellulose as they retain water in bowel lumen. Milk as neutral contrast is preferred in paediatric patients as hyperosmolar agents like mannitol can cause dehydration. PEG can cause watery diarrhoea in some patients. Commercially available neutral agent, VoLumen (Bracco Diagnostics, Princeton, NJ) is a 0.1% w/v barium sulphate suspension in sorbitol and produces better distension than many other neutral agents. Usually 1–2 L of oral contrast produce adequate luminal distension; however, volume to be administered should be adequately tailored according to the safety profile of the specific agent, to achieve good luminal distension with minimal side effects. Smaller volumes, depending upon patient tolerance, are recommended in patients with history of bowel resection. Patients are advised to completely restrict solid food intake for about 6 hours prior to examination. To assure better compliance, they should be well-briefed about the procedure. They are encouraged to drink at least 1.3–1.5 L of neutral oral contrast over a period of 45–60 minutes immediately prior to the study. An intravenous assess is secured with 18G or 20G cannula and saline flush is given to check patency. Slow injection of 1 mL of Buscopan (hyoscine butylbromide) is given immediately before the scan to relax bowel smooth muscles and decrease peristalsis. Scan techniques include routine plain and ‘enteric phase’ imaging performed 45–50 seconds after giving ~1.5–2 mL/kg of iso or low osmolar intravenous contrast at the rate of 4 mL/s. Multiphase CT with arterial and delayed phases is indicated while evaluating occult GI bleeds or vascular malformations. Slice thickness of 0.9 mm is adequate with reconstruction interval of 0.45 mm. CT enteroclysis is a semiinvasive imaging technique that differs from CT enterography only in the mode of enteric contrast administration. CT image acquisition remains the same. It combines the advantages of enteroclysis (good luminal distension) with cross-sectional imaging in a single examination. Large volume of enteric contrast is pumped directly into the small bowel at high rates. This volume challenge to bowel ensures better and reliable luminal distension than CT enterography, thus aiding in better evaluation of mucosal lesion and mural enhancement patterns. A good CT enterography examination with adequately distended bowel loops can demonstrate luminal, mural as well as extraluminal pathologies. Better patient tolerance and noninvasive nature has made CT enterography a preferred imaging choice in modern noninvasive small bowel evaluation. A good CT examination of the bowel can help in detecting abnormal bowel loops positioning, intussusception (Fig. 8.3.1.15), intraluminal, mural (Figs. 8.3.1.16 and 8.3.1.17) and extramural pathologies. Arterial phase images are particularly useful in evaluating the small bowel arterial supply for stenosis, strictures, thrombosis and arterio-venous malformations (AVMs) (Figs. 8.3.1.18 to 8.3.1.20). Until recently, despite excellent inherent soft tissue resolution, MR imaging had limited role in GI tract evaluation. The major hurdle being longer acquisition time, resulting in image degradation from motion artefacts due to respiratory movements and bowel peristalsis. With advancements in MR hardware and development of faster breath-hold imaging sequences, scan times have reduced considerably with minimal motion artefacts and superior image quality. When combined with good luminal distention and intravenous administration of gadolinium-based contrast media, MR provides exceptional luminal, mural and extramural details along with vascular and functional information. Increasing awareness of radiation hazards associated with high-radiation dose in CT has furthered MR enterography to the forefronts of GI imaging especially when imaging paediatric, pregnant patients or patients with chronic bowel pathologies who require sequential imaging during their disease course. Advantages of MR over CT are lack of ionizing radiation, superior soft tissue contrast, dynamic information with respect to bowel motility and relatively safer intravenous MR contrast profiles. Limitations of MR include limited availability, higher cost, longer scan time, lower spatial and temporal resolution as compared to CT and known contraindications to MR such as claustrophobia, metallic implants and pacemakers. Gadolinium-based contrast agents, especially gadodiamide, are contraindicated in patients of chronic kidney disease or renal insufficiency with eGFR < 30 mL/min/1.73 m2 due to risk of fatal nephrogenic systemic fibrosis. For adequate bowel cleansing, patients are advised to take liquid-based diet for a day with four bisacodyl tablets in the evening prior to imaging. Preprocedural fasting of 4–6 hours is advised. All this minimizes food residue and debris in small bowel which may mimic luminal pathology while interpreting the scan. Bowel distension is necessary for evaluating the intraluminal and mural pathologies as collapsed small bowel segments can mimic pathological mural thickening or hide underlying pathologies. MR imaging of small bowel makes use of enteral contrast agents to provide homogenous and adequate intestinal distension and increase contrast between lumen, bowel wall and extraluminal soft tissue. Properties of a good enteral contrast agent include easy availability, low cost, least side effects and high contrast between lumen and bowel wall. Depending on the signal intensity on various sequences, MR enteric contrast agents can be divided into positive (bright on T1w images), negative (dark on T2w images) and biphasic (dark on T1w and bright on T2w images) contrast agents. Positive contrasts like gadolinium chelates, ferrous and manganese ions and food items like blueberry juice are not commonly used as enteric contrast due to high cost, unavailability and poor distinction of mural enhancement postintravenous contrast injection. Negative contrast agents include superparamagnetic iron oxides (SPIOs) and ultra-small SPIOs (USPIOs). They are used in MR pancreatico-cholangiography to suppress the high signal from bowel luminal contents. High signal intensity of pathology or inflammation in bowel wall and surrounding fat stands out against accompanying luminal low signal intensity on T2W images. However, negative contrasts are not preferred for bowel imaging as, apart from gastrointestinal side effects like nausea, vomiting and diarrhoea, the associated susceptibility artefact can mask the hypointense signal from normal bowel wall and hide low signal intensity lesions like carcinoids on T2W images. Biphasic agents are the most commonly used enteric contrast agents in MR and include osmotic agents like mannitol, polyethylene glycol, low-density barium sulphate (VoLumen) and nonosmotic agents like water, locust gum resin and methylcellulose. Osmotic agents retain water within the lumen producing better luminal distension than nonosmotic agents; however, this may lead to mild diarrhoea postexamination. On T2W images, there is marked contrast between the high signal intensity of the lumen against dark appearing normal bowel wall, thus aiding detection of transmural ulcers, sinuses and fistula. On fat-suppressed postintravenous contrast T1W images, their inherent low signal intensity provides outstanding contrast between low signal intensity lumen, intermediate signal intensity of normal bowel wall and high signal intensity of enhancing mural/extramural inflammation or neoplasm. Enteric contrast may not be required in patients with suspected high-grade obstruction because retained intestinal fluid adequately distends the loops proximal to obstruction, and additionally administering large volumes of fluid may cause patient discomfort, vomiting or even bowel perforation. Similar to CT imaging, MR imaging of the small bowel includes two techniques of small bowel distension: MR enterography (MRE) with oral administration of the enteric contrast and MR enteroclysis, wherein the enteric contrast is infused directly into the small bowel via a naso-jejunal tube inserted under fluoroscopic guidance.
8.3: Small bowel
Imaging anatomy
Arterial supply
Imaging relevance
Mesentery
Imaging relevance
Gut wall
Imaging relevance
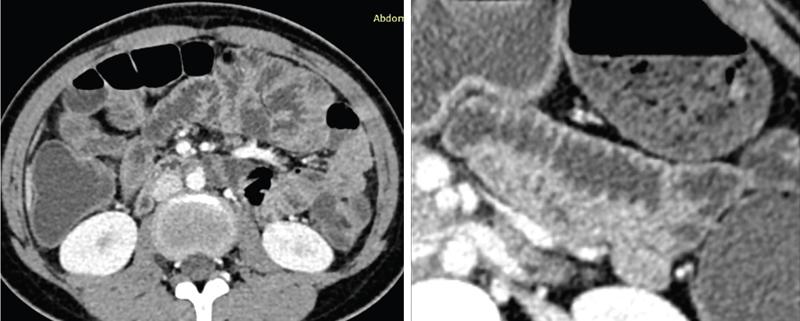
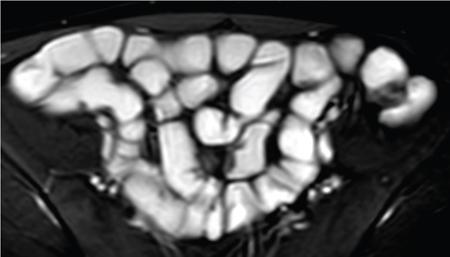
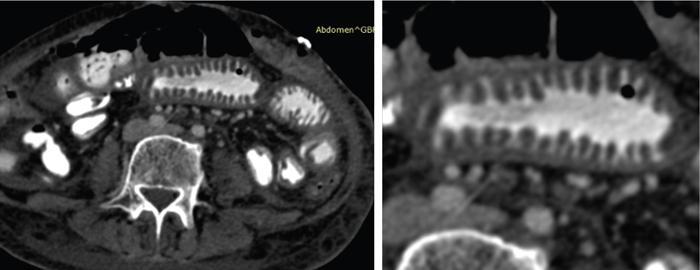
Nerve supply
Brief overview of gastrointestinal tract (GIT) development
Foregut
Midgut
Hindgut
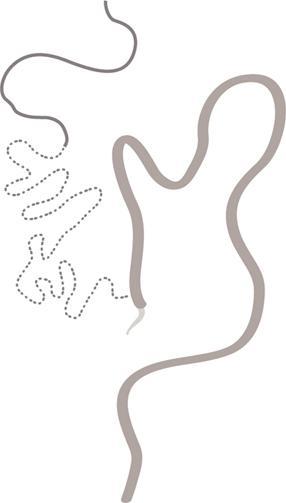
Embryology of the GI tract and their histological derivatives (Table 8.3.1.2)
Imaging relevance
Endoderm
Mesoderm
Neural Crest Cells
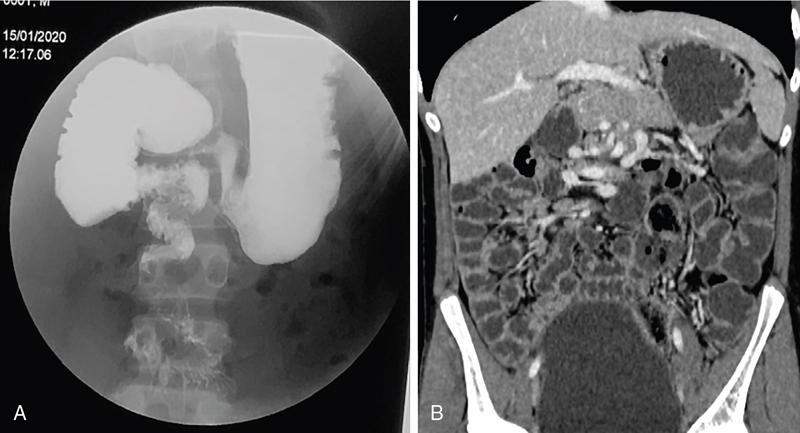
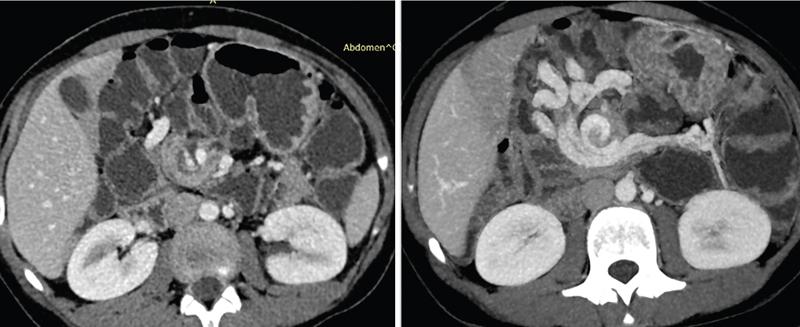
Brief history of gastrointestinal (GI) imaging
Imaging modalities in small bowel imaging
Small bowel imaging modalities
Conventional imaging
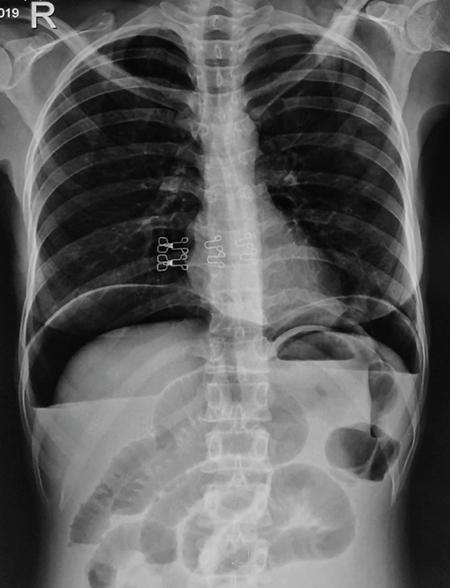
Plain abdominal radiograph
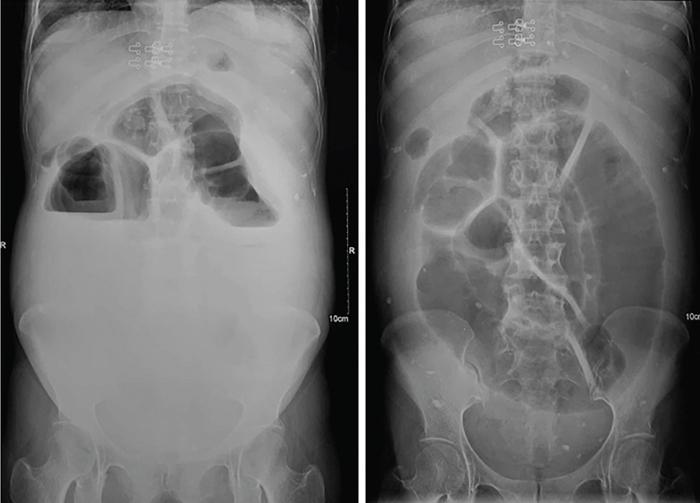
Fluoroscopic barium studies
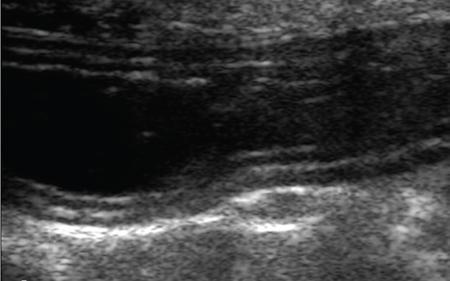
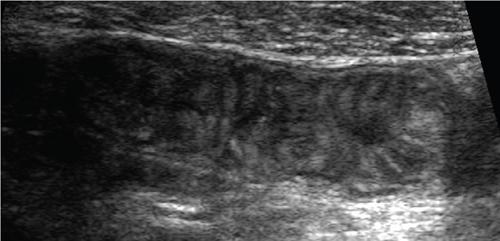
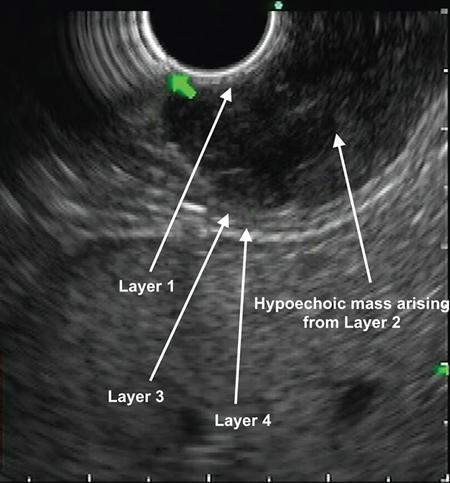
Ultrasonography in small bowel imaging
Computed tomography
Negative Agents (Negative CT Attenuation)
Neutral Agents (Near Water Attenuation, 0–30 HU)
Positive Agents (High CT Attenuation, >50 HU)
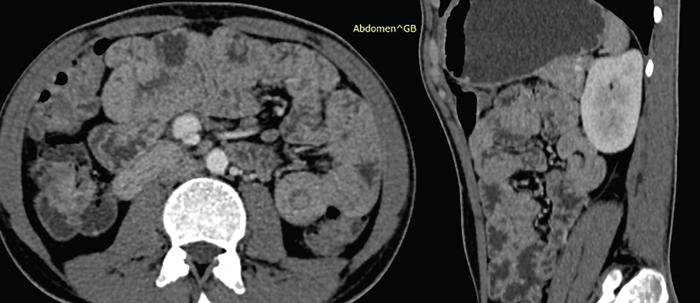
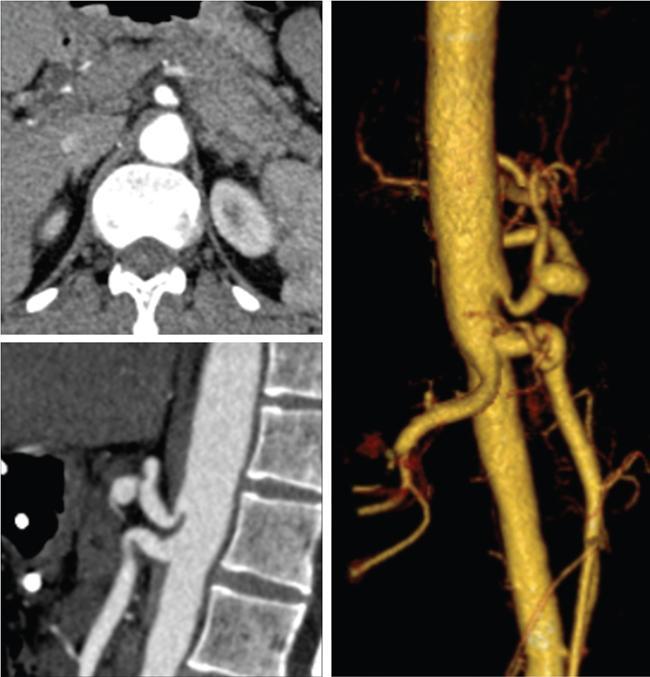
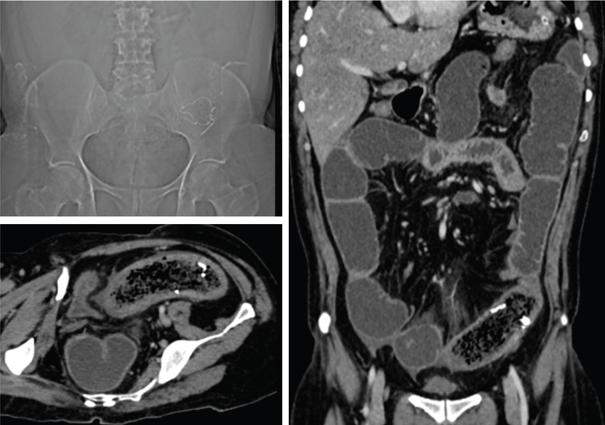
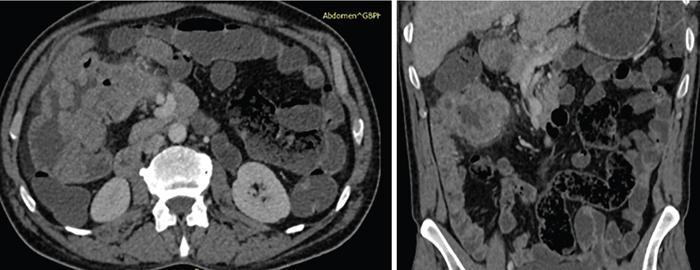
CT enterography
CT enteroclysis
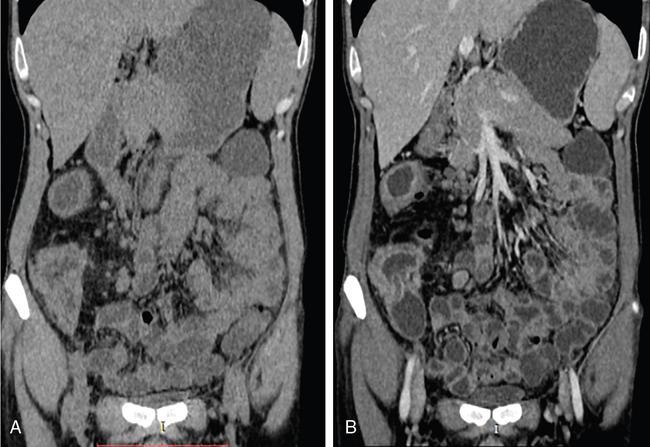
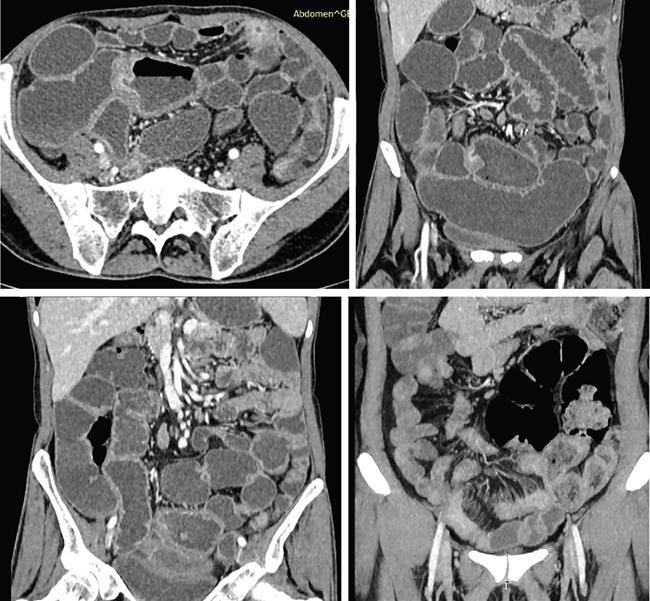
MR imaging of small bowel
Patient preparation and enteric contrast agents
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








