Small Bowel Obstruction: General Considerations
Etiology
Small bowel obstruction (SBO) is a common manifestation, and appropriate management continues to be a clinical challenge. The morbidity and mortality associated with acute SBO continue to be significant; however, there has been a decline in mortality from SBO in the last 50 to 60 years from 25% to 5%. The goal of treatment is to recognize the complications of ischemia early and develop an appropriate clinical plan. SBO can be caused by a variety of lesions, and the cause can be broadly classified based on their location. Intraluminal, intramural, and extraluminal causes have been described. The causes of SBO by age are listed in Table 25-1 .
| Cause | Neonates and Infants <2 Years | Children and Young Adults | Adults and the Elderly |
|---|---|---|---|
| Intraluminal | Meconium ileus Foreign bodies | Foreign bodies Ascaris lumbricoides | Foreign bodies Gallstones Food bolus |
| Intramural | Intussusception Congenital atresias and stenoses Henoch-Schönlein purpura | Crohn’s disease Benign and malignant neoplasms Tuberculosis | Crohn’s disease Benign and malignant neoplasm Radiation strictures Surgical anastomoses |
| Extramural | Midgut volvulus Inguinal hernia Congenital bands Postoperative adhesion | Inguinal hernia Adhesions Midgut volvulus | Adhesions (postoperative and inflammatory) Hernia Neoplasia |
Prevalence and Epidemiology
The leading cause of SBO in the United States is adhesions occurring after surgical procedures such as appendectomy, colorectal surgery, and gynecologic and upper gastrointestinal procedures. Other common causes are Crohn’s disease, neoplasm, and hernia.
SBOs can be classified based on the completeness into partial or complete (high grade vs. low grade) and based on complications as nonstrangulated or strangulated. If left untreated, strangulated obstructions cause death in 100% of patients. Mortality decreases to 8% if surgery is performed within 36 hours and to 25% if the surgery is postponed beyond 36 hours. The prevalence of SBO by cause is summarized in Box 25-1 . Numbers vary across studies.
- •
Adhesions: 67% to 74%
- •
Neoplasms: 5% to 13%
- •
Inflammatory bowel disease: 4% to 7%
- •
Hernia: 2% to 8%
- •
Miscellaneous: 4% to 12%
Clinical Presentation
The diagnostic approach should focus on recognizing an obstruction, distinguishing partial from complete SBO, distinguishing simple from strangulated SBO, and identifying the underlying cause. Patients with SBO usually present with a variable period of abdominal pain, often accompanied by nausea and vomiting. Colicky pain is more often associated in patients with simple obstruction and increases in severity. It can be continuous or interspersed with pain-free intervals. Strangulated obstructions commonly manifest as constant pain. The presence of vomiting is another feature of SBO. In more proximal obstructions, vomiting is an early finding, with or without significant distention of the abdomen. In more distal obstruction, abdominal distention is marked and vomiting is delayed as the bowel takes time to fill. Patients can have constipation or diarrhea, which is caused by secondary increased peristalsis distal to an obstruction. Whereas patients with partial obstruction might have diarrhea and can still pass flatus, patients with complete obstruction tend to have complete obstipation. Fever, hypotension, tachycardia, and leukocytosis are suggestive of strangulation.
On physical examination, patients will show abdominal distention. The degree of distention often depends on the level of obstruction. Bowel sounds can be hyperactive in an early stage of obstruction or hypoactive late in the course as well as with strangulated lesions. Strangulated SBO also is often associated with peritoneal signs. Peritonitis and perforation present as a silent tender abdomen and are late signs. The most important clinical findings in patients with SBO are shown in Box 25-2 .
- •
Abdominal pain
- •
Nausea and vomiting
- •
Abdominal distention
- •
Diarrhea or constipation; obstipation in complete small bowel obstruction
- •
Bowel sounds are hyperactive in early stages or hypoactive in late stages
Anatomy
The small bowel is a complex organ with multiple functions. It measures approximately 120 cm in length from the pylorus to the ileocecal valve. The potential surface area available for digestion is increased approximately 600 times by the presence of the circular folds, villous mucosa, and microvillar surface of the epithelium. The jejunum and ileum receive their blood supply from the superior mesenteric artery and its vascular arcades. Venous drainage is by the superior mesenteric vein, which joins the splenic vein posterior to the pancreas to form the portal vein. Small bowel loops are suspended by a mobile mesentery and covered by a lining of peritoneum that extends over the serosal surface of the bowel. Lymphatic drainage of the small bowel is into the regional lymph nodes, which follow the vascular arcades and drain into the cisterna chili. Loops of jejunum are mostly located in the left hypochondrium in an individual with normal gut rotation. The ileum is mostly in the midline of the pelvis. The terminal ileum is the narrowest part of the small bowel. Knowing the normal bowel gas pattern on abdominal radiographs is important because this determines the ability to detect deviation from the normal pattern and classify this further. The various bowel gas patterns are discussed in detail later in the section on radiographic imaging.
Pathology
Obstruction of the small bowel leads to proximal dilatation of the intestine secondary to accumulation of gastrointestinal secretions and swallowed air. This bowel dilatation stimulates cell secretory activity, resulting in more fluid accumulation. This leads to increased peristalsis both above and below the obstruction with frequent loose stools and flatus early in its course. Vomiting occurs if the level of obstruction is proximal. Increasing small bowel distention leads to increased intraluminal pressures. This can cause compression of mucosal lymphatics, leading to bowel wall lymphedema. With even higher intraluminal hydrostatic pressures, increased hydrostatic pressure in the capillary beds results in massive third spacing of fluid, electrolytes, and proteins into the intestinal lumen. The fluid loss and dehydration that ensue may be severe and contribute to increased morbidity and mortality. Strangulated SBOs are most commonly associated with adhesions and occur when a loop of distended bowel twists on its mesenteric pedicle. The arterial occlusion leads to bowel ischemia and necrosis. If left untreated, this progresses to perforation, peritonitis, and death. Proximal to the obstruction, bacteria proliferate in the gut. Microvascular changes in the bowel wall allow translocation to the mesenteric lymph nodes. This is associated with an increase in incidence of bacteremia due to Escherichia coli.
Imaging
SBO is a common clinical condition, and its diagnosis can be established if there is the classic triad of good clinical history, focused clinical examination, and targeted imaging and laboratory evaluation. The challenge is making the diagnosis of SBO, which is uncomplicated and can potentially be managed conservatively, from complicated SBO, in which bowel ischemia and strangulation are the concern. Vomiting, abdominal distention, and colicky or constant abdominal pain (individually or as a combination) are often the manifesting complaints. Imaging plays a very important role in making the diagnosis of this condition, and abdominal radiography, although not very sensitive or specific, is the first imaging test because of its widespread availability and low cost. Subsequent use of imaging modalities depends on the findings on radiographs and the level of clinical concern. Evaluation can be done using luminal contrast-enhanced studies or cross-sectional imaging. The goal of advanced imaging is three-fold: confirm the diagnosis, identify the cause, and evaluate for associated complications.
Radiography
Abdominal Radiography.
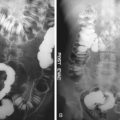
Conventional abdominal radiography remains the preferred method of initial radiologic examination of symptomatic patients suspected of SBO. The normal bowel gas pattern is either absence of small bowel gas or small amounts of gas within up to four variably shaped nondistended (<2.5 cm) loops of small bowel. A normal gas distribution and presence of stool in the large bowel should be recognized. A normal but nonspecific gas pattern describes a pattern of at least one loop of borderline or mildly distended small bowel (2.5 to 3 cm) with three or more air/fluid levels on upright or lateral decubitus radiographs. The colonic gas and stool pattern is either normal or has a similar degree of borderline distention. This kind of pattern can result from many conditions, such as low-grade obstruction, functional ileus, and medication-induced hypoperistalsis. The probable SBO pattern consists of multiple gas or fluid-filled loops of dilated small bowel with a moderate amount of colonic gas. The presence of colonic gas can indicate early complete SBO, incomplete SBO, or a nonobstructive ileus. A definite SBO pattern shows dilated gas or fluid-filled loops of small bowel in the setting of a gasless colon ( Figure 25-1 ). These findings are diagnostic of SBO. It is important to distinguish complete from partial SBO, because management is different. On plain radiographs, the presence of residual colonic gas after 6 to 12 hours is suggestive of partial SBO ( Figure 25-2 ). Despite the description of the various gas patterns that can be seen on radiographs, these are diagnostic in only 50% to 60% of cases. Box 25-3 lists common manifestations of SBO on plain radiography.
- •
Loops of distended small bowel (>3 cm)
- •
Dilated bowel loops proximal and collapsed bowel loops distal to obstruction
- •
Gas and fluid levels (>3 air/fluid levels on an erect radiograph measuring >3 cm)
- •
Increased distance between valvulae conniventes (“stretch” sign)
- •
Trapped gas between valvulae conniventes (“string-of-pearls” sign)
Intraluminal Contrast Radiography.
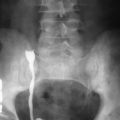
Barium can be safely used in the adynamic gut because it does not typically inspissate within it. Evaluation of the small bowel using barium can be done by either nonintubation techniques or intubation and insufflations. Nonintubation techniques include small bowel follow-through (SBFT) or per-enterostomy (colostomy or ileostomy) small bowel enemas. SBFT has limitations in the setting of SBO. The presence of significant bowel distention in patients with SBO can result in dilution of the ingested barium, resulting in incomplete opacification and poor mucosal detail. Also, in patients with bowel obstruction there is a prolonged transit time of the oral contrast agent that can be a limiting factor. The nonintubation techniques are unable to assess intestinal distensibility and fixity of bowel loops, which is another drawback. Despite these limitations, the SBFT technique is a viable alternative to enteroclysis in settings in which intubation is not possible because of technical or patient-related factors. Common manifestations of SBO on barium follow-through are listed in Box 25-4 .
- •
Small amount of contrast agent through narrow lumen at the site of obstruction (“beak” sign)
- •
Increased peristalsis to overcome obstruction leading to bulbous shape proximal to obstruction (snakehead appearance)
- •
Stretched mucosal folds
- •
Dilution of ingested barium in massively distended loops of small bowel (drawback of small bowel follow-through)
Barium Enteroclysis.
Enteroclysis is not practiced widely because it requires conscious sedation, nasointestinal intubation, and constant radiologist involvement. This technique involves the intubation of small bowel to a point beyond the pylorus and often beyond the duodenojejunal flexure, with infusion of nondiluted contrast medium. Based on findings of enteroclysis, SBO has been graded. This grading is being used for the characterization of bowel obstruction on modalities such as CT. In Table 25-2 the causes of SBO are classified on the basis of the findings of enteroclysis.
| Type of Obstruction | Description |
|---|---|
| Low grade | Sufficient contrast agent flow through obstruction to define mucosal folds distal to obstruction. |
| High grade | Delayed passage of contrast agent leads to dilution by intestinal fluid. Minimal flow does not enable definition of mucosal folds. |
| Complete | No contrast agent passes the obstruction. |
Enteroclysis has a diagnostic value in patients in whom the diagnosis of low-grade SBO is clinically uncertain. Its ability to distinguish low-grade obstruction from normal bowel makes it important in assisting diagnosis. Enteroclysis challenges the distensibility of small bowel and exaggerates the effect of mild or subclinical mechanical obstruction. The infusion technique promotes flow of contrast agent toward the site of obstruction despite diminished peristalsis. This distention facilitates evaluation of fixed and nondistensible bowel segments. Studies have shown that this technique can predict the presence of obstruction in 100%, the absence of obstruction in 88%, the level of obstruction in 89%, and the cause of obstruction in 86% of patients. SBO is excluded when unimpeded flow of contrast agent is seen in normal-caliber loops from the duodenojejunal junction to the right colon. Mechanical SBO is confirmed when a transition point is demonstrated. By enteroclysis criteria, 3 cm is the upper limit of normal for jejunal caliber and 2.5 cm is the upper limit for the ileum.
The level of obstruction is identified during the single-contrast phase of the study; the cause of obstruction is best evaluated during double-contrast evaluation in which mucosal detail is well appreciated. In partial SBO, enteroclysis has been almost 85% accurate in distinguishing adhesions from metastases, tumor recurrence, and radiation damage.
Enteroclysis can also gauge the severity of intestinal obstruction, an advantage over other modalities. In low-grade partial SBO there is no delay in contrast medium arriving at the point of obstruction, and adequate contrast medium passes distally such that the fold patterns of the distal bowel are well visualized. High-grade partial SBO is diagnosed when retained bowel fluid dilutes the barium and results in inadequate contrast density above the site of obstruction, allowing only small amounts of contrast medium to pass through the obstruction into the collapsed distal loops. Complete obstruction is diagnosed when there is no passage of contrast medium beyond the point of obstruction, as shown on delayed radiographs obtained up to 24 hours after the start of the examination.
Despite all its advantages, it is often impractical to perform this as an outpatient procedure. In the acute setting when time is limited, CT is often the initial method of examination.
Computed Tomography
CT is a valuable tool in the evaluation of patients with suspected SBO. A low overall sensitivity (63%) of CT for all grades of SBO has been reported. Sensitivity increases with high-grade SBO (81% to 100%) and conversely worsens with low-grade SBO (48%). CT can identify the location and severity of an SBO and detects the cause of the obstruction in 93% to 95% of cases. Complications such as closed-loop obstructions and strangulation are recognized, which would prompt immediate surgical intervention. Strangulation of bowel and ischemia occur as a complication of intussusception, volvulus ( Figure 25-3 ), torsion, hernia, or other types of closed-loop obstruction in which there is compromise of the mesenteric vascular pedicle. The presence of pneumatosis intestinalis on CT is an indicator of strangulation and bowel ischemia ( Figure 25-4 ). Decreased enhancement of a segment of bowel wall in the arterial phase with increased enhancement in the venous phase seems to be highly specific for bowel ischemia. Evaluation of the course of vascular arcades around the involved loops of bowel on CT helps identify patients who are at risk for the life-threatening complications of strangulation and ischemia. This is one of the major advantages of CT over other modalities for the diagnosis of this condition. CT is also useful in distinguishing high-grade obstruction from an ileus pattern with reported sensitivities up to 100% compared with 46% with plain radiographs.
The speed of multidetector CT (MDCT) and the isotropic data available with the newer generation scanners have revolutionized abdominal imaging. The widespread use of MDCT scanners and their availability has enabled rapid acquisition of scan data and has added significantly to CT diagnosis and evaluation of SBO. Multiplanar reformats can be used to identify the transition point of SBO more reliably and to assess adjacent structures, as well as the mural and extramural extent of small bowel lesions. This aids planning for surgical resection.
CT can be performed using barium and water as enteral contrast agents assist in visualization of mucosal detail. An intravenous contrast agent is used routinely in the absence of contraindications because it assists in the evaluation of bowel wall and mucosa, possible associated inflammatory or neoplastic processes, and mesenteric vasculature. In patients suspected of having ischemic bowel, scans should be performed in the arterial and venous phases to search for occluded arteries and veins, to depict vascular anatomy, and to assess bowel wall perfusion.
Controversy exists about what oral contrast agent should be used in the setting of a suspected SBO, and practices vary by institution. Some authors use a positive oral contrast medium in this setting as tolerated by the patient because it is helpful in determining if an obstruction is complete.
Analysis of dilution effects on CT can be particularly helpful in patients whose plain radiographs did not show any gas-filled dilated loops (so-called gasless abdomen ). It also can help in localizing the point of obstruction because the small bowel loops containing more dilute contrast medium are distal relative to those with more dense oral contrast.
Other investigators have deemed oral contrast unnecessary if SBO is evident on plain radiographs, because the contrast agent is unlikely to reach the obstruction and may make evaluation of bowel wall thickening difficult if it does reach the obstruction. If ischemia is suspected, water is used as the enteric contrast agent because positive contrast media interfere with vascular three-dimensional (3D) reconstructions. In general, water is better tolerated than positive contrast media in patients with SBO. The most common manifestations of SBO on CT are shown in Box 25-5 .
- •
Dilated proximal bowel loop (>2.5 cm) and collapsed distal loop (<1 cm)
- •
Passage of contrast agent into collapsed segment indicating partial obstruction
- •
Accumulation of feces and gas proximal to obstruction (feces sign) indicating mechanical obstruction
- •
Bowel wall thickening, portal venous gas, and pneumatosis indicating strangulation
Magnetic Resonance Imaging
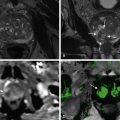
Although magnetic resonance imaging (MRI) can be used for evaluation of the small bowel, its role is somewhat limited in the acute setting. Advantages of MRI are the high soft tissue contrast and the acquisition of multiplanar images without exposing patients to ionizing radiation. This is especially important for young patients who are undergoing multiple evaluations (e.g., those with Crohn’s disease) pregnant patients.
For imaging of SBO, the major advantage of MRI is the direct visualization of the small bowel wall. Fast sequences enable acquisition of T1- and T2-weighted images within a single breath-hold. For T2-weighted images half-Fourier acquisition single-shot turbo spin-echo (HASTE) sequences with a fast acquisition time of approximately 1 second per image are used. For T1-weighted images, 2D or 3D spoiled gradient recalled echo (SPGR) before and after intravenous administration of a contrast agent are used. These ultrafast sequences have the disadvantage of being prone to chemical shift artifacts but are especially useful in patients who are unable to hold their breath. The use of parallel imaging techniques is another option to minimize scan time, by reducing the number of phase encoding steps per repetition time. This allows even shorter scan times by maintaining spatial resolution or alternatively allowing higher spatial resolution by maintaining short acquisition time.
For both T1- and T2-weighted sequences the addition of fat-saturated pulses has been shown to increase contrast between bowel and surrounding fat tissue.
The major problem of the described MRI methods is flow artifacts that are generated by peristaltic motion that could imitate intraluminal lesions. This can be reduced by antiperistaltic medication. Placing the patient in the prone position also can aid in reduction of artifact from abdominal wall motion and respiration. MRI of the small bowel is performed with oral contrast agents. Positive oral contrast agents, such as gadolinium chelates and ferrous and manganese ions are paramagnetic and reduce mainly T1 relaxation time. However, the use of positive contrast agents is limited because a hyperintense lumen often obscures pathologic processes of the bowel wall. Negative contrast agents, such as iron oxides, perfluoroocytl bromide, and barium sulfate produce low signal intensity on T1-wighted as well as T2-weighted images. T2 effects are predominant and caused by spin dephasing, which leads to a loss of signal intensity. One disadvantage of negative contrast agents are magnetic susceptibility artifacts by ferrous oxide on gradient-echo sequences.
Biphasic contrast agents show different signal characteristics in different sequences. Manganese and gadolinium chelates at high concentrations lead to hyperintensity on T1-weighted images and hypointensity on T2-weighted images, whereas water, mannitol-based solutions, polyethylene glycol, and barium sulfate create hypointensity on T1-weighted images and hyperintensity on T2-weighted images. Disadvantages of these contrast agents are the intestinal absorption of water and unwanted intestinal side effects of mannitol solutions.
MRI of the small bowel requires distention, because collapsed bowel loops can hide pathologic findings. There are two main approaches: imaging after administration of an oral contrast agent (MR follow-through) and imaging with distention by nasojejunal contrast application (MR enteroclysis).
For MR SBFT, image acquisition is preceded by oral ingestion of 600 to 1000 mL of contrast agent 20 minutes before the examination. A spasmolytic agent is added intravenously immediately before imaging is performed. T2-weighted and T1/T2 hybrid sequences are acquired in axial and coronal planes, until the terminal ileum is completely distended.
With the MR enteroclysis technique a biphasic contrast agent is given through a nasojejunal tube. The volume of contrast agent depends on the subject and varies between 1500 and 2000 mL. It is preferably applied using a peristaltic pump with an infusion rate between 80 and 150 mL. The main advantages of MR enteroclysis are improved intestinal distention and real-time imaging with ultrafast sequences. In the evaluation of SBO, MRI can be especially useful for distinction of nonobstructive or partially obstructive lesions using real-time evaluation with MR enteroclysis.
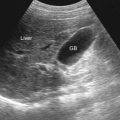
Ultrasonography
Ultrasonography is widely used in the assessment of acute abdominal pain. However, its use in the evaluation of SBO obstruction is limited. It is important to be aware of the findings of bowel obstruction on ultrasonography because this may be an incidental finding in a patient who is being evaluated for abdominal pain. In SBO, fluid accumulates in the bowel lumen. When there is no gas in the bowel lumen, fluid-filled bowel loops might be difficult to detect on plain radiographs. Contrast ultrasonography can be very useful to visualize SBO, because the intraluminal fluid is a natural contrast medium and helps demonstrate the origin of the obstruction. It can accurately detect the location of the obstruction. Characteristic features are rounded distended bowel loops with loss of definition and prominent valvulae conniventes. Ultrasonography is also able to assess the peristalsis and movements of intraluminal content. Box 25-6 shows the most common manifestations of SBO seen on ultrasound evaluation.
- •
Luminal diameter greater than 2.0 to 2.5 cm in jejunum and greater than 1.5 to 2.0 cm in ileum
- •
Dilated segment longer than 10 cm
- •
Increased peristalsis in dilated loops
- •
Collapsed colon
Imaging Algorithm
No single, generally accepted approach exists to evaluate patients with SBO. Plain radiography remains the initial modality of choice. However, radiographs may be diagnostic in only 45% to 60% of cases. The decision to use additional diagnostic imaging methods should be based on clinical presentation and findings on plain radiography. The definitive presence of SBO on plain radiograph confirms the diagnosis. Based on the clinical condition of the patient, if conservative management is initiated or if there are doubts about the diagnosis, standard CT has emerged as the imaging modality of choice for further evaluation of SBO ( Table 25-3 ). CT enteroclysis may be performed only if conventional CT is nondiagnostic. Ultrasonography and MRI can occasionally be useful in the diagnosis of SBO and evaluation of possible causes. However, CT is superior to these imaging modalities because it is more comprehensive and not limited by intraluminal air. Nuclear medicine studies and positron emission tomography with CT (PET/CT) do not play a role in the diagnosis of acute SBO and are reserved for assessment of underlying causes of SBO. An imaging algorithm is provided in Figure 25-20 .
| Modality | Accuracy (%) | Limitations | Pitfalls |
|---|---|---|---|
| Radiography | Sensitivity: 75 Specificity: 53 | Barium studies cannot be performed in patients with perforation or complete SBO Extended transit time in SBFT | Diagnosis might be missed if no gas present in the small bowel |
| CT | Accuracy: 94 Sensitivity: 92 Specificity: 96 | Ionizing radiation Risk for contrast reaction | Low sensitivity for low-grade, partial, or incomplete SBO |
| MRI | Accuracy: 88 Sensitivity: 90 Specificity: 86 | Expensive Time consuming Not available everywhere | Patient factors: Claustrophobia, pacemakers, metal implants |
| Ultrasonography | Accuracy: 84 Sensitivity: 83 Specificity: 100 | Air can obscure the field of view Difficult in obese patients | Operator dependent |
| Nuclear medicine | Sensitivity: 79 Specificity: 98 | Poor spatial resolution | Activity can be observed with other causes of inflammation. |
| PET/CT | Data not available | Ionizing radiation High cost | Differentiation of benign from borderline neoplasms can be difficult. |
Differential Diagnosis
A number of medical conditions can cause symptoms similar to those of SBO. Abdominal distention, vomiting, and constipation can be seen in patients with electrolyte disorders, uremia, diabetic ketoacidosis, and thyroid disorders, as well as intoxication with anticholinergics or tricyclic antidepressants. Peritonitis of any cause can mimic the symptoms of SBO.
Any condition resulting in small bowel dilatation or abdominal distention can be confused with SBO before clinical and radiographic investigations are completed and integrated. Obstruction typically results in distention that may include the colon in addition to small bowel. A concurrent inflammatory process, recent surgery, gastrointestinal infection, and electrolyte disturbances are predisposing factors for SBO, and appreciation of these comorbidities is important in scan interpretation. Cecal volvulus causes more proximal colonic obstruction and typically secondary small bowel dilatation, as well. Recognition of the distended cecum is the key to differentiating volvulus from an isolated SBO. Shock bowel can manifest as dilated loops of small bowel, but involvement of the colon and history should help differentiate the MDCT findings from SBO. Frank ischemia also can cause marked small bowel distention and simulate the appearance of obstruction. Ancillary findings of bowel wall thickening, segmental bowel perfusion abnormalities, and proof of arterial atherosclerotic disease provide evidence for an ischemic cause of the dilatation, however. Although Crohn’s disease may cause SBO because of stricture formation, stenotic-phase disease, or superimposed acute disease, it also may result in areas of dilatation and narrowing without a true obstruction.
Treatment
Medical Treatment
Initial nonsurgical management includes fluid administration, bowel decompression using nasogastric tubes, analgesics, antiemetics, and antibiotics, which should cover gram-negative and anaerobic organisms. Based on clinical features, laboratory and radiologic findings, nonoperative trial, and conservative management may be used for partial or simple obstruction. Resolution of the obstruction can occur in patients with these lesions in up to 72 hours. Surveillance using imaging techniques and regular clinical monitoring is done for these patients on conservative treatment, to detect any change that indicates complications.
Surgical Treatment
A strangulated SBO is a surgical emergency. In patients in whom the SBO is complete, the risk for strangulation is high and early surgical intervention is necessary. Patients with simple complete obstructions in whom nonoperative trials fail also need surgical treatment but experience no apparent disadvantage from delayed surgery.
- •
Recognize SBO: Clinical presentation and plain radiography (diagnostic in 45% to 60%).
- •
Determine the site of obstruction.
- •
Discriminate partial from complete obstruction.
- •
Discriminate simple from strangulated obstruction.
- •
Determine the cause (is there a cause that can be removed?).
- •
Detect and treat associated complications.
Prevalence and Epidemiology
The leading cause of SBO in the United States is adhesions occurring after surgical procedures such as appendectomy, colorectal surgery, and gynecologic and upper gastrointestinal procedures. Other common causes are Crohn’s disease, neoplasm, and hernia.
SBOs can be classified based on the completeness into partial or complete (high grade vs. low grade) and based on complications as nonstrangulated or strangulated. If left untreated, strangulated obstructions cause death in 100% of patients. Mortality decreases to 8% if surgery is performed within 36 hours and to 25% if the surgery is postponed beyond 36 hours. The prevalence of SBO by cause is summarized in Box 25-1 . Numbers vary across studies.
- •
Adhesions: 67% to 74%
- •
Neoplasms: 5% to 13%
- •
Inflammatory bowel disease: 4% to 7%
- •
Hernia: 2% to 8%
- •
Miscellaneous: 4% to 12%
Clinical Presentation
The diagnostic approach should focus on recognizing an obstruction, distinguishing partial from complete SBO, distinguishing simple from strangulated SBO, and identifying the underlying cause. Patients with SBO usually present with a variable period of abdominal pain, often accompanied by nausea and vomiting. Colicky pain is more often associated in patients with simple obstruction and increases in severity. It can be continuous or interspersed with pain-free intervals. Strangulated obstructions commonly manifest as constant pain. The presence of vomiting is another feature of SBO. In more proximal obstructions, vomiting is an early finding, with or without significant distention of the abdomen. In more distal obstruction, abdominal distention is marked and vomiting is delayed as the bowel takes time to fill. Patients can have constipation or diarrhea, which is caused by secondary increased peristalsis distal to an obstruction. Whereas patients with partial obstruction might have diarrhea and can still pass flatus, patients with complete obstruction tend to have complete obstipation. Fever, hypotension, tachycardia, and leukocytosis are suggestive of strangulation.
On physical examination, patients will show abdominal distention. The degree of distention often depends on the level of obstruction. Bowel sounds can be hyperactive in an early stage of obstruction or hypoactive late in the course as well as with strangulated lesions. Strangulated SBO also is often associated with peritoneal signs. Peritonitis and perforation present as a silent tender abdomen and are late signs. The most important clinical findings in patients with SBO are shown in Box 25-2 .
- •
Abdominal pain
- •
Nausea and vomiting
- •
Abdominal distention
- •
Diarrhea or constipation; obstipation in complete small bowel obstruction
- •
Bowel sounds are hyperactive in early stages or hypoactive in late stages
Anatomy
The small bowel is a complex organ with multiple functions. It measures approximately 120 cm in length from the pylorus to the ileocecal valve. The potential surface area available for digestion is increased approximately 600 times by the presence of the circular folds, villous mucosa, and microvillar surface of the epithelium. The jejunum and ileum receive their blood supply from the superior mesenteric artery and its vascular arcades. Venous drainage is by the superior mesenteric vein, which joins the splenic vein posterior to the pancreas to form the portal vein. Small bowel loops are suspended by a mobile mesentery and covered by a lining of peritoneum that extends over the serosal surface of the bowel. Lymphatic drainage of the small bowel is into the regional lymph nodes, which follow the vascular arcades and drain into the cisterna chili. Loops of jejunum are mostly located in the left hypochondrium in an individual with normal gut rotation. The ileum is mostly in the midline of the pelvis. The terminal ileum is the narrowest part of the small bowel. Knowing the normal bowel gas pattern on abdominal radiographs is important because this determines the ability to detect deviation from the normal pattern and classify this further. The various bowel gas patterns are discussed in detail later in the section on radiographic imaging.
Pathology
Obstruction of the small bowel leads to proximal dilatation of the intestine secondary to accumulation of gastrointestinal secretions and swallowed air. This bowel dilatation stimulates cell secretory activity, resulting in more fluid accumulation. This leads to increased peristalsis both above and below the obstruction with frequent loose stools and flatus early in its course. Vomiting occurs if the level of obstruction is proximal. Increasing small bowel distention leads to increased intraluminal pressures. This can cause compression of mucosal lymphatics, leading to bowel wall lymphedema. With even higher intraluminal hydrostatic pressures, increased hydrostatic pressure in the capillary beds results in massive third spacing of fluid, electrolytes, and proteins into the intestinal lumen. The fluid loss and dehydration that ensue may be severe and contribute to increased morbidity and mortality. Strangulated SBOs are most commonly associated with adhesions and occur when a loop of distended bowel twists on its mesenteric pedicle. The arterial occlusion leads to bowel ischemia and necrosis. If left untreated, this progresses to perforation, peritonitis, and death. Proximal to the obstruction, bacteria proliferate in the gut. Microvascular changes in the bowel wall allow translocation to the mesenteric lymph nodes. This is associated with an increase in incidence of bacteremia due to Escherichia coli.
Imaging
SBO is a common clinical condition, and its diagnosis can be established if there is the classic triad of good clinical history, focused clinical examination, and targeted imaging and laboratory evaluation. The challenge is making the diagnosis of SBO, which is uncomplicated and can potentially be managed conservatively, from complicated SBO, in which bowel ischemia and strangulation are the concern. Vomiting, abdominal distention, and colicky or constant abdominal pain (individually or as a combination) are often the manifesting complaints. Imaging plays a very important role in making the diagnosis of this condition, and abdominal radiography, although not very sensitive or specific, is the first imaging test because of its widespread availability and low cost. Subsequent use of imaging modalities depends on the findings on radiographs and the level of clinical concern. Evaluation can be done using luminal contrast-enhanced studies or cross-sectional imaging. The goal of advanced imaging is three-fold: confirm the diagnosis, identify the cause, and evaluate for associated complications.
Radiography
Abdominal Radiography.
Conventional abdominal radiography remains the preferred method of initial radiologic examination of symptomatic patients suspected of SBO. The normal bowel gas pattern is either absence of small bowel gas or small amounts of gas within up to four variably shaped nondistended (<2.5 cm) loops of small bowel. A normal gas distribution and presence of stool in the large bowel should be recognized. A normal but nonspecific gas pattern describes a pattern of at least one loop of borderline or mildly distended small bowel (2.5 to 3 cm) with three or more air/fluid levels on upright or lateral decubitus radiographs. The colonic gas and stool pattern is either normal or has a similar degree of borderline distention. This kind of pattern can result from many conditions, such as low-grade obstruction, functional ileus, and medication-induced hypoperistalsis. The probable SBO pattern consists of multiple gas or fluid-filled loops of dilated small bowel with a moderate amount of colonic gas. The presence of colonic gas can indicate early complete SBO, incomplete SBO, or a nonobstructive ileus. A definite SBO pattern shows dilated gas or fluid-filled loops of small bowel in the setting of a gasless colon ( Figure 25-1 ). These findings are diagnostic of SBO. It is important to distinguish complete from partial SBO, because management is different. On plain radiographs, the presence of residual colonic gas after 6 to 12 hours is suggestive of partial SBO ( Figure 25-2 ). Despite the description of the various gas patterns that can be seen on radiographs, these are diagnostic in only 50% to 60% of cases. Box 25-3 lists common manifestations of SBO on plain radiography.