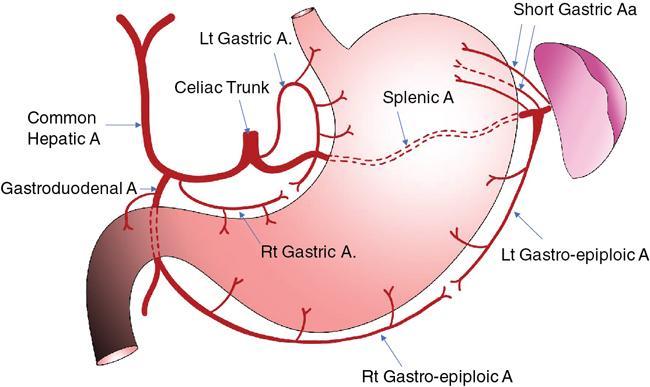
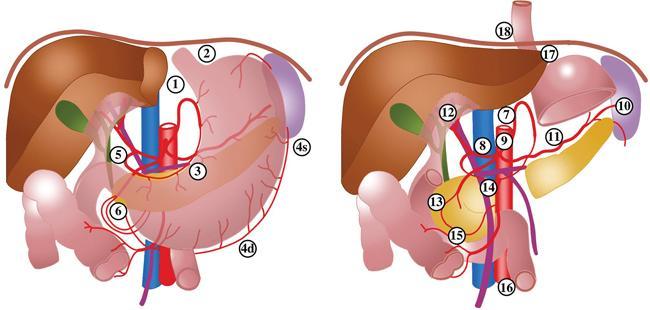
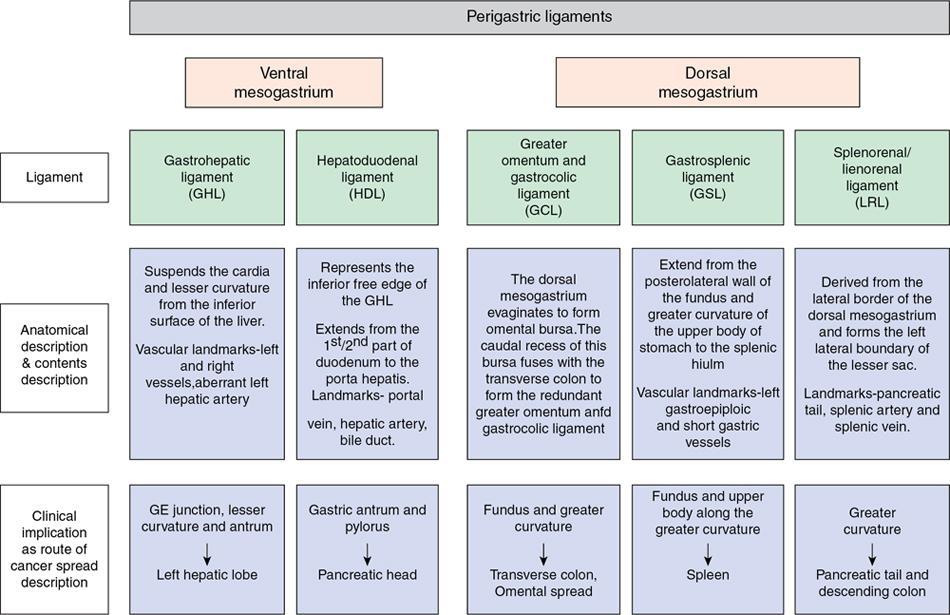
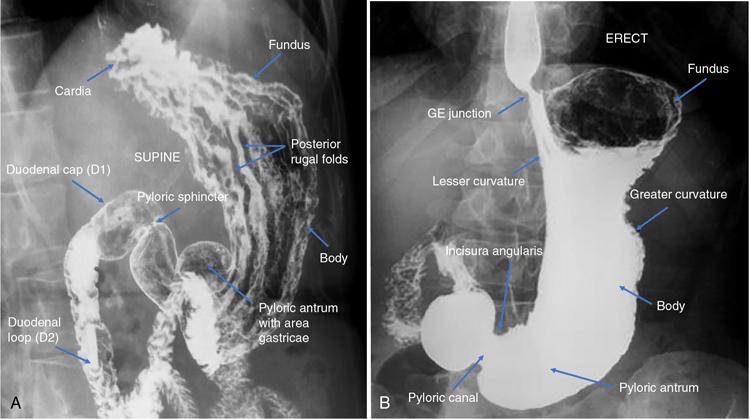
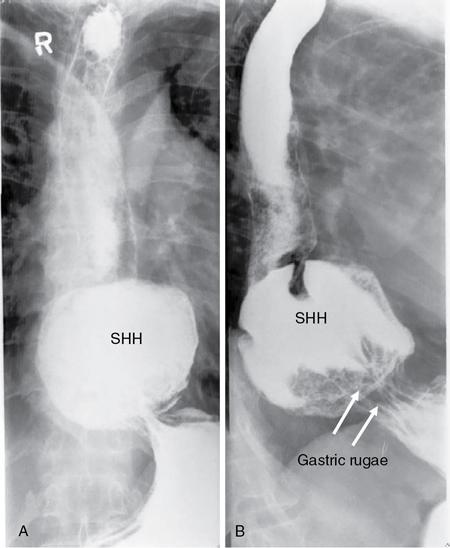
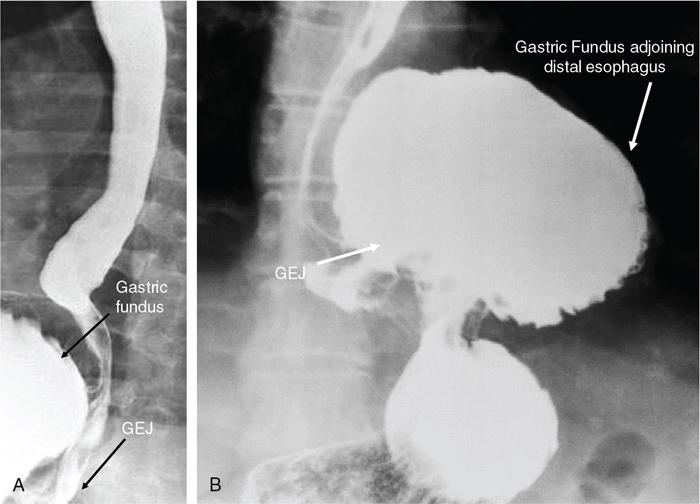
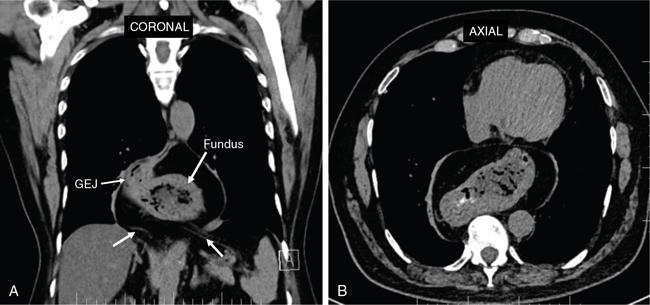
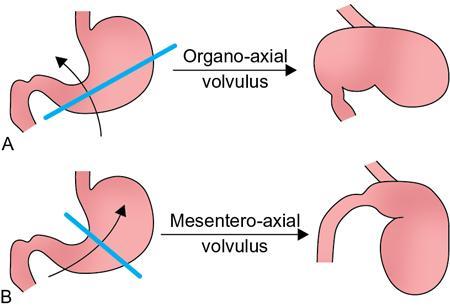
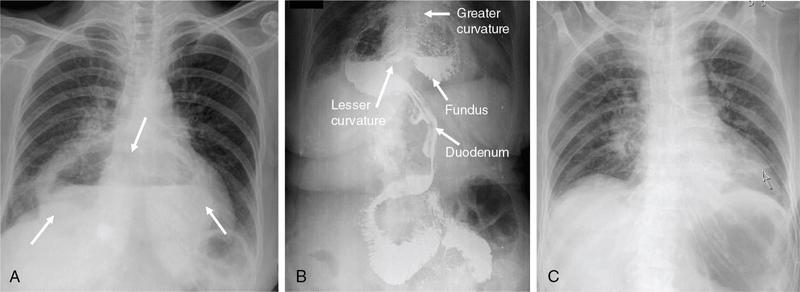
Swarup Nellore, Soumil Vyas, Ujwal Bhure, Ankit Jain, Richa Kothari, Daksh Mehta, Divya Kantesaria, Disha Lokhandwala, Karthik Ganesan The stomach is the most dilated portion of the gastrointestinal tract positioned between the oesophagus and the small intestine occupying the epigastrium, left hypochondrium and umbilical regions of the abdomen performing a multitude of functions including storage and mixing of food and controlling the passage of food into the intestine. Anatomically, the stomach is divided into the following parts (Fig. 8.2.1): the gastric cardia is the portion immediately adjoining the oesophageal opening into the stomach. The fundus is the dome-shaped part above the horizontal plane of the cardiac orifice which undergoes receptive relaxation and is the site of the autonomic pacemaker. The body is the largest part of the stomach extending from the cardiac orifice to the level of incisura angularis (notch like bend in the mid lesser curvature) containing majority of parietal cells (which secrete hydrochloric acid), chief cells (pepsinogen) and enterochromaffin-like cells (ECL). The pylorus is the tubular distal portion of the stomach which is further divided into the gastric antrum and pyloric canal. The pyloric antrum containing G-cells producing gastrin extends from the incisura angularis up to the pyloric sphincter which is an anatomical sphincter formed by concentric thickening of the circular muscle coat and encircles the narrow-slit like pyloric canal. The left dome of diaphragm and oesophagus lie superior to the stomach while the greater omentum and pancreas lie inferiorly. Spleen and liver lie on either side laterally while the transverse mesocolon is located inferolaterally. Diaphragm, greater omentum, left lobe of liver and anterior abdominal wall are anteriorly related to the stomach and contents of lesser sac including pancreas, spleen, splenic artery, transverse mesocolon, left kidney and adrenal gland lie posteriorly. The stomach is embryologically derived from the primitive foregut and is suspended anteriorly by the ventral mesogastrium and posteriorly by the dorsal mesogastrium. During development as the stomach rotates, the peritoneum grows and condenses to form perigastric ligaments, which contain blood vessels, lymphatics, lymph nodes, nerves and fat. The liver forms in the ventral mesogastrium, which develops into the falciform ligament, gastrohepatic ligament (GHL) and hepatoduodenal ligament (HDL). The spleen and pancreas form within the dorsal mesogastrium, which fuses with the posterior abdominal wall to form the gastrocolic ligament (GCL), gastrosplenic ligament (GSL) and splenorenal ligament (SRL). The perigastric ligaments are identified anatomically by the vessels contained in them (Fig. 8.2.2). The gastric cardia and lesser curvature of the stomach are attached to the inferior surface of liver by the lesser omentum by the gastrohepatic ligament respectively which contains the left and right gastric vessels. The inferior free edge of the gastrohepatic ligament extends inferiorly as the hepatoduodenal ligament between the porta hepatis and proximal duodenum gastrophrenic ligament extends between the stomach and the left hemidiaphragm. The gastrocolic ligament identified by the left and right gastroepiploic vessels, connects the greater curvature of the stomach to the anterior surface of the transverse colon. This ligament extends inferiorly as the greater omentum which is a double-layered peritoneum forming a drape anterior to the bowel loops. The gastrosplenic/lienogastric ligament bridges the posterolateral wall of the fundus and greater curvature along the proximal body of the stomach to the splenic hilum and contains the short gastric vessels. The gastrosplenic ligament is contiguous with the lienorenal ligament and both these structures constitute the lateral boundary of the lesser sac. Macroscopically the stomach shows a thick vascular mucosal lining in the form of longitudinal folds called gastric rugae. Microscopic layers of the stomach include mucosa, submucosa, muscularis externa and serosa. The mucosa includes surface mucus cells with simple columnar epithelium, gastric pits, gastric glands, lamina propria and muscularis mucosa. Submucosa includes connective tissue with submucosal Meissner’s plexus. Muscularis externa comprises three smooth muscle layers (longitudinal, circular and oblique) and Auerbach myenteric plexus. The outermost layer called serosa consists of loose connective tissue and visceral peritoneal lining. The arterial supply of stomach is constituted by the branches of celiac artery predominantly in the form of two anastomotic arcades along the lesser and greater curvature. Left gastric artery, a direct branch of the celiac trunk supplies the upper part of the lesser curvature while the lower part is supplied by the right gastric branch of the common hepatic artery arising at the upper border of pylorus. The left gastroepiploic artery, a branch of splenic artery, supplies the upper part of greater curvature and the inferior portion is supplied by the right gastroepiploic artery which is a branch of the gastroduodenal artery. The fundus additionally receives few small short gastric branches from the splenic artery. The veins follow the arteries in their nomenclature. Left and right gastric vein show direct drainage into the portal vein. The splenic vein derives flow from the short gastric and left gastroepiploic vein whereas the right gastroepiploic vein drains into the superior mesenteric vein. Pylorus can be surgically delineated by the prepyloric vein of Mayo lying on its anterior surface. Intrinsic nerve supply consists of the myenteric plexus of Auerbach and submucosal plexus of Meissner. Extrinsic nerve supply consists of sympathetic and parasympathetic components. Sympathetic nerve supply arises from T5 to T9 spinal cord segments supplying the celiac plexus via the greater splanchnic nerve. The plexuses then travel along the respective branches of celiac artery supplying the stomach. Parasympathetic nerve supply is derived from oesophageal plexus of vagus dividing into two vagal trunks. Right (posterior) vagus gives off the posterior gastric branch also called the criminal nerve of Grassi supplying the cardia and fundus of stomach. Right vagus then gives off a celiac branch and continues along lesser curvature of stomach as the posterior gastric nerve of Latarjet supplying the antrum, body and pylorus. Left (anterior) vagus gives off a hepatic branch then continues along the lesser curvature as the anterior nerve of Latarjet supplying the antrum, body and pylorus. Crow’s feet innervation to antropyloric area are important surgical landmarks preserved during highly selective vagotomy receiving branches from both major nerve trunks (anterior and posterior) at the incisura angularis. Anatomical lymphatic drainage is divided into three areas. Area I is the superior two-thirds of stomach draining via the nodes along left and right gastric vessels into the aortic nodes. Area II includes the right two-thirds of the inferior one-third which drains through the nodes along right gastroepiploic vessels via the subpyloric nodes into aortic nodes. Area III includes left one-third of greater curvature draining via short gastric and splenic nodes into suprapancreatic nodes and ultimately into aortic nodes. Gastric lymph node stations are divided into 4 levels with 16 lymph node stations: Paraesophageal nodes below the diaphragmatic hiatus (17) and above the diaphragmatic hiatus (18) are also included. The stomach first appears as a fusiform dilatation in the distal endodermal foregut in the 4th week of embryonic life. The dorsal and ventral mesogastrium suspend the developing stomach to the respective abdominal walls. Preferential growth along the dorsal border of the developing stomach leads to the formation of a convex dorsal border (greater curvature) and a concave ventral border (lesser curvature). The stomach subsequently undergoes a sequence of rotations. The first (90 degrees) clockwise rotation occurs around the longitudinal axis which brings the lesser curvature to the right and greater to the left. This rotation also brings the dorsal mesogastrium towards the left and ventral to the right creating a space behind the stomach called as the lesser sac or omental bursa. The second clockwise rotation occurs around the anteroposterior axis, with the caudal or pyloric part moving upwards and to the right while the cephalic or cardiac portion moves towards the left and slightly downward causing the stomach to assume its final anatomic position. This rotation causes the dorsal mesogastrium to bulge downwards and grow further to eventually become a double-layer apron called the greater omentum. The developing liver divides the ventral mesogastrium into the falciform ligament extending from the ventral abdominal wall to the liver with its lower free edge forming the ligamentum teres, the visceral peritoneum surrounding the liver and the lesser omentum with its two parts – the hepatogastric ligament and hepatoduodenal ligament. The dorsal mesogastrium gives rise to the redundant greater omentum, gastrocolic ligament, gastrosplenic ligament and lienorenal ligament. These perigastric ligaments help us in deciphering patterns of the spread of gastric malignancies and in taking decisions regarding their management and prognostication. These will be further explained in detail under the section of gastric malignancies. An abdominal radiograph is often the initial imaging test for evaluation of acute abdominal pain. The gastric bubble is seen below the left hemidiaphragm in situs solitus. Presence of gastric outlet obstruction may show a distended stomach with gasless small bowel. Hollow visceral perforation is diagnosed by free air seen under domes of diaphragm. Also, the presence and position of various tubes like the nasogastric tube can be confirmed using a radiograph. A fluoroscopic upper gastrointestinal (GI) examination is the radiological study of oesophagus, gastro-oesophageal junction, stomach, duodenum up to duodenojejunal junction by oral administration of contrast. Barium sulphate is a good contrast medium for GI studies as it is radio-opaque, non-absorbable, inert to tissues and can be used for double-contrast studies. Patient should be nil by mouth at least 4 hours prior to the examination. In a single contrast examination, the emphasis is on mucosal relief, luminal distention with contrast material and compression. In the erect position, a small amount of contrast is given to swallow while the oesophagus is visualized under fluoroscopy. The table is then made horizontal and the patient is rotated in a clockwise manner as seen from the foot end of the patient, thus ensuring good coating of the stomach mucosa. Mucosal relief images are then obtained in supine and prone positions to demonstrate the mucosal fold pattern and possible filling defects. After giving some more contrast, the patient is turned oblique with the right side dependent and spot images of the duodenal cap and C loop are taken both in distended and empty states. The patient is again positioned erect and more images of duodenal cap and loop are taken. Further contrast is given to optimally distend the lumen and assess for possible contour abnormalities, wall rigidity and strictures. Compression techniques help assess for filling defects and lesions, in the compressible areas of the stomach. Images are taken in multiple positions – supine, prone, right anterior oblique, right lateral, left posterior oblique in recumbent and right anterior oblique in erect position. Gastric peristalsis and emptying can be observed at fluoroscopy. Advantages of the single contrast technique are that it can be performed quickly, well-tolerated and requires less patient cooperation as compared to double-contrast studies. It can even be performed in physically debilitated patients. Barium is contraindicated in suspected cases of perforation, aspiration, fistula or recent GI biopsy. Single contrast examination can be performed using water-soluble iodinated contrast media in these cases and immediate postoperative patients. Earlier, ionic contrast like Gastrograffin was used. However, due to its high osmolarity, nowadays, non-ionic contrast is preferred as it causes less electrolyte imbalance due to its low osmolarity. Also, it delineates the GI tract very well due to less dilution. Double-contrast studies provide better evaluation of the mucosa than do single-contrast studies. Here, the emphasis is on coating the mucosa with barium and distending the lumen with gas. A well-performed double-contrast study is biphasic and also incorporates some single-contrast techniques. The initial part of the examination is the same as a single contrast examination to obtain the mucosal relief images. Then, gas-forming powder that produces carbon dioxide within the stomach lumen is given with more barium. With the double-contrast technique, the mucosa is coated with a high-density barium suspension and the lumen distended with gas. The patient is rotated in a clockwise manner as seen from the foot end of the patient, thus ensuring good coating of the stomach mucosa. Spot images of the distended stomach are taken followed by the duodenal cap and loop in oblique right-side dependent position. Patient is brought back to erect position erect and more images of stomach, duodenal cap and loop are taken. Further contrast is given to distend the lumen. Residual fluid or food debris in the stomach impairs stomach coating, and lack of adequate coating may preclude visualization of lesions. In addition, optimal gaseous distention is important as underdistention will cause a false appearance of abnormally thickened, and overdistention can obliterate abnormal folds. With normal gastric anatomy, the gastric fundus is best visualized in the upright left-posterior-oblique position, the body in the supine anteroposterior position and the antral-pyloric region in the supine left-posterior-oblique position. The normal gastric mucosal surface should be smooth on double-contrast studies. Areae gastricae appear as reticular networks of polygonal tufts which, owing to the presence of barium in the narrow intervening grooves, are coated with white lines. These are seen most often in the antrum or body of the stomach but can also be seen in the fundus. Areae gastricae are identified on double-contrast studies in 70% of patients and are seen more often in elderly patients. Polygonal tufts should normally range in size from 2 to 3 mm in the antrum and 3 to 5 mm in the body and fundus. Normal gastric folds are more prominent in the proximal to mid stomach and more undulating along the greater curvature as compared with the lesser curvature. Antral folds should be typically effaced with the double-contrast technique. Gastric cardia is characterized by three or four stellate folds radiating from a central point at the gastro-oesophageal junction; this is known as the cardiac rosette and is best visualized in the supine right-lateral position. Ultrasonography (USG) is the modality of choice to visualize hypertrophic pyloric stenosis in infants. Although USG does not play much of a role in adults for imaging of stomach due to reflection of sound waves by air, luminal distension with water may enable to identify mucosal – submucosal pathologies and to assess perigastric relationships in pathologies. CT can not only evaluate the location and anatomy of the stomach, but also provide additional information about the relationship of the stomach to surrounding structures. An optimal CT examination of the stomach includes good stomach distension with a well-visualized wall. Gastric distention can be achieved by the oral administration of negative or positive luminal contrast. Positive contrast agents include a 1%–2% barium suspension or a 2%–3% solution of a water-soluble iodinated contrast agent. Water-soluble agents should be used in cases of suspected perforation. Positive agents provide a bright lumen with better identification of luminal encroachment or diverticula but may limit detailed evaluation of gastric wall enhancement. Positive contrast also may not mix well with gastric contents, producing a pseudotumor appearance. On the other hand, neutral or negative contrasts agents, usually water, produce a low attenuation lumen which allows for more detailed evaluation of the gastric wall and its enhancement pattern following intravenous contrast and is preferred for three-dimensional imaging. Multidetector CT with thin collimation allows for postprocessing in any orthogonal plane. The CT data of the stomach can be manipulated to simulate endoscopic images (virtual gastroscopy). This display technique accentuates the stomach wall and folds. Prior to evaluating the stomach with CT, oral contrast is administered at repeated intervals as well as immediately prior to scanning. Multiphasic scanning after intravenous contrast may be employed at 25–35 seconds (late arterial), 55–75 seconds (venous phase) after contrast injection. Normally, the gastric wall may enhance to approximately 120 Hounsfield Units after intravenous contrast administration. The mucosa may enhance more than the relatively lower-attenuation muscularis propria. Abnormal enhancement of the wall can highlight pathologic processes. The CT appearance of the stomach also depends upon the degree of distention. When well distended, the body and fundus is less than 5 mm thick, though the normal antrum may appear slightly thicker. The presence and pattern of wall enhancement, degree and location of wall thickening, and length of an involved segment may be assessed. Comprehensive MRI examination of the stomach and duodenum by the combined use of T2-weighted single-shot and T1-weighted gradient echo (GRE) – with and without fat suppression, and gadolinium-enhanced 3D GRE helps to detect the spectrum of gastric diseases on MRI. These sequences minimize artefacts from bowel peristalsis, increase the sensitivity of detection of peritoneal and serosal disease by suppressing the high signal of intra-abdominal fat, widen the dynamic range of abdominal tissue signal intensities and distinguish between intraluminal bowel contents and bowel wall. The gastric rugae are well seen on T2-weighted single-shot spin-echo images and their enhancement appears as bands of enhancing tissue arranged perpendicular to the external contour of the gastric wall. The normal gastric wall enhances more intensely than other segments of bowel on immediate postgadolinium SGE images. Water is frequently used as an oral contrast agent when imaging the upper GI segment-stomach and duodenum. Noninvasive dynamic study for assessment of gastric motor function and wall motility can also be done using MRI for diagnosis of gastroparesis. Gastric emptying scintigraphy (GES) is a well-established radionuclide procedure to evaluate patients with suspected gastric motility disorder, more so for non-invasive assessment of gastroparesis in patients with symptoms of postprandial fullness, nausea, vomiting, abdominal pain, bloating, early satiety, loss of appetite, as well as weight loss. Gastroparesis, commonly seen in diabetics, is a condition that affects the normal spontaneous movement of the gastric muscles, leading to impairment of gastric motility, because of which the stomach cannot empty itself of food in a normal fashion and time. GES can also provide useful information with regards to assessment of impaired gastric motility in patients with GERD (gastro-oesophageal reflux disease) unresponsive to routine therapy, diabetics with poor glycemic control, and also in patients with colonic inertia who are being considered for colectomy (since benefits of colectomy are mitigated in patients with concurrent impaired gastric emptying). On the other hand, GES is also useful for assessment of rapid gastric emptying (GE), which is a major factor in dumping syndrome, often seen post peptic ulcer surgery, with or without vagotomy. The solid or liquid component of a meal is radiolabelled with a radiopharmaceutical, which is consumed by the patient and subsequently measured gastric radioactive counts by scintigraphy correlate with the volume of the meal remaining in the stomach. Solid-phase GES is used for the evaluation of gastric motility disorder/gastroparesis. Liquid-phase gastric emptying is generally not clinically useful since it is often in the normal range in spite of deranged emptying for solids even with severe gastroparesis. The preferred radionuclide meal for the gastric emptying scintigraphy includes egg-white radiolabelled with 0.5–1.0 mCi of Technetium-99m (99mTc) [approximate caloric value of 255 kcal]. Two large eggs (egg-whites), two slices of bread, fruit jam, water (120 mL approximately), and the radiopharmaceutical (0.5–1.0 mCi of 99mTc–sulphur colloid). For patients allergic, to eggs, alternative meals like oatmeal or Liquid Ensure nutrient supplement can be used. Reporting should include the percentage of radiotracer retained in the stomach at defined time intervals. The normal reference values for comparison are as follows: Grading the severity of the delay in gastric emptying based on gastric retention at 4 hours of the study: Gastro-oesophageal reflux (GER) is a common condition in infants and children, wherein there is reflux of stomach contents into the oesophagus, due to possible incompetence of gastro-oesophageal sphincter, which can lead to development of symptoms like regurgitation, heartburn, cough and dyspepsia. Children can present with recurrent respiratory infections, iron deficiency anaemia and failure to thrive. It is often a self-limited process in infants and children which usually resolves by 12–18 months of age. On the other hand, gastro-oesophageal reflux disease (GERD) is a chronic pathological process which can be troublesome. Scintigraphic evaluation of gastro-oesophageal reflux, popularly known as ‘milk scan’, is an established imaging modality and has sensitivity of 75%–100%. This radionuclide test for gastro-oesophageal reflux is physiological and quantitative and commonly used for diagnosis in neonates and infants. Four hours fasting is preferred. A liquid meal consisting of formula, milk, or orange juice mixed with the radiopharmaceutical is administered either orally or by nasogastric tube. 99mTc–sulphur colloid. Dose: 0.2 to 1 mCi. After administering the liquid meal and allowing burping, the baby is positioned supine under the gamma camera detector head, including chest and upper abdomen in the field of view. A radioactive marker can be placed at the mouth. Anterior serial dynamic planar images are acquired for around 60 minutes with a framing rate of 5–10 seconds per frame. Delayed anterior static image of the chest may be acquired additionally. The acquired frames are reviewed in cinematic display for visual estimation and to look for reflux of the radiotracer into the oesophagus. Reflux is seen as distinct spikes of radiotracer activity into the oesophagus. For quantitative estimation, the volume of each episode of reflux, frequency of episodes and rate of reflux clearance from the oesophagus are taken into consideration. Percentage of reflux is calculated by drawing an ROI over the oesophageal spike of radiotracer activity and the counts are calculated and expressed as a fraction of the gastric activity counts. Values greater than 5% are suggestive of reflux. Semiquantitative evaluation grades each reflux event: Based on this, four categories can be formulated: The greater the number of high-level reflux events and the longer they last, more severe is the gravity of the problem. Pulmonary aspiration should also be looked for, though the sensitivity of detection is poor. Liquid gastric emptying can be evaluated during the same study up to 2 hours. Normal values for liquid gastric emptying are: Endoscopy is the preferred examination to evaluate for majority of the upper GI symptoms. It is very sensitive in detecting mucosal lesions and simultaneously, biopsies can be taken. In cases of upper GI bleed, endoscopy can not only detect the site and cause of the bleed, but also further treatment like adrenaline/sclerosant injection or ligation can be done. EUS can provide detailed assessment of the 5 layers of the gastric wall and is thought to be the most accurate non-surgical method for local tumour staging (Fig. 8.2.9). For subepithelial (submucosal) gastric lesions, EUS can assess the echogenicity of a lesion, the exact layer of the gastric wall which is involved (accurate T-staging of a tumour) and assess for the presence of blood flow with Doppler ultrasound to help ascertain the likely aetiology. EUS-guided fine needle aspiration may also provide additional diagnostic information. Hiatus hernia is a common positional abnormality of stomach, characterized by herniation of abdominal contents, commonly the gastro-oesophageal junction and stomach, into the mediastinum above the diaphragm through a widened oesophageal hiatus. The oesophageal hiatus is an elliptical opening with complex anatomy formed by the right and left crus of the diaphragm. However, the most common anatomical description is of the right crus splitting into two bundles, the right and left, which form a sling encircling the distal oesophagus and may be reinforced by the left diaphragmatic crus. The distal oesophagus is typically anchored to the oesophageal hiatus by a main restraining structure which is the phrenicoesophageal ligament/membrane (the fascia of Laimer) which is a condensation of endo-thoracic and endo-abdominal fascia and this membrane circumferentially inserts into the oesophageal musculature in close proximity to the squamocolumnar junction (SCJ). The ligament maintains the competence of the oesophageal hiatus and prevents rostral herniation of the gastro-oesophageal junction and stomach. During normal physiological swallowing, the longitudinal layer of the oesophageal muscularis propria contracts with associated oesophageal shortening and stretching of the phrenicoesophageal ligament/membrane resulting in the transient elevation of the oesophagogastric junction and portion of the stomach above the hiatus. At the end of the swallowing mechanism, the elastic recoiling of the phrenicoesophageal ligament/membrane restores the migrated segment to its normal position. With ageing-related wear and tear, the ligament becomes lax with progressive depletion of elastin fibres resulting in proximal migration of the gastro-oesophageal junction and stomach. Other potential risk factors of fibromuscular degeneration are caused by increase in the intra-abdominal pressure as occurs in obesity and pregnancy. The majority of the symptomatic cases of hiatal hernia present with the clinical symptoms of gastro-oesophageal reflux disease (GERD) with most characteristic manifestations of acid regurgitation, heartburn, dysphagia, epigastric or chest pain and even chronic iron deficiency anaemia. Large hernias may also present with early satiety. Conventionally, hiatus hernias are classified into sliding and paraesophageal varieties. The current comprehensive anatomic classification has evolved to include a categorization of hiatal hernias into Types I–IV and is necessary especially regarding the treatment approach, as indications for the surgical method are quite different between sliding and paraesophageal hernias. Majority of hiatus hernias (95%) are sliding hiatus hernias (type I), the remaining type II–IV hernias as a group are referred to as paraesophageal hernias (PEH), with more than 90% of these comprising type III, and the least common being of type II. The assessment of hiatus hernia can be done by various modalities including radiological, manometry and endoscopy studies. The diagnosis of a large hiatus hernia is straightforward, however, identifying a small one is challenging due to mobility of the gastro-oesophageal junction during physiological process like deglutition and respiration. According to the SAGES Guidelines, only those investigations which will alter the clinical management of the patient should be performed. Radiological investigations are usually indicated for diagnosis and presurgical evaluation. Plain chest radiographs demonstrate opacity within the chest typically in the midline overlapping the cardiac shadow or paramedian location (Fig. 8.2.14A), with or without an air-fluid level. Barium swallow radiography is still considered an easy tool for diagnosis of hiatus hernia and provides essential information about the size, location, motility dysfunction, stricture secondary to GERD and stenosis. The evaluation of hiatus hernia requires understanding of anatomical radiographic landmarks like phrenic ampulla, A ring and B ring (or Schatzki’s ring) on a barium study (Fig. 8.2.14B). The phrenic ampulla is a globular structure seen above the diaphragm and below the tubular oesophagus during a contrast swallow. The A ring represents a muscular ring visible at the upper margin of the phrenic ampulla and physiologically corresponds to the highest-pressure zone within the lower oesophageal sphincter, this is often seen as a transient indentation on the oesophageal lumen. The B ring identified in about 15% of individuals, representing the mucosal ring corresponds to the squamocolumnar junction (Z line) and divides the phrenic ampulla into the oesophageal vestibule (A ring to B ring) and the sliding hiatus hernia (B ring to the stomach below the diaphragm). The B ring is a persistent constriction which is not usually not more than 2–3 mm and despite mucosal, can sometimes be symptomatic and require dilatation. By convention, there must be a distance of 2 cm or more between the B ring and the hiatus, to call it a hiatus hernia as physiological migration up to 2 cm is known in normal individuals. Although not reliable, barium studies may also provide information about a short oesophagus, which is identified as a tubular straightened oesophagus with the oesophagogastric junction fixed far above the diaphragm and may be stenotic, the intrathoracic herniated stomach revealing a more elongated funnel/bell – shaped configuration. Cine fluoroscopy provides further additional information about the bolus transit through gastro-oesophageal junction. The major drawbacks of the procedure are non-visualization of B-ring in majority of the cases, radiation exposure, barium/iodine hypersensitivity, contraindication in pregnancy and increase risk of aspiration in paraesophageal hernia causing pneumonitis. Computed tomography (CT) scan is not routinely advised for the primary diagnosis of hiatus hernia but is indicated in cases of suspected complications like volvulus in paraesophageal hernia, obstruction, perforation and strangulation. Multi-slice CT with reconstruction in sagittal, coronal planes and 3D reformations have increased the sensitivity of CT in detection of hiatal hernia. Oesophagogastroduodenoscopy gives added advantage of real-time visual assessment of the mucosa of the oesophagus, stomach and duodenum. The common diagnostic criteria in endoscopic is proximal migration of gastro-oesophageal junction by >2 cm. Endoscopy can also determine the presence of erosive esophagitis or Barrett’s oesophagus. Further, the inability or difficulty of negotiating the scope into duodenum in the presence of a large hiatal hernia is diagnostic of paraesophageal hernia with volvulus. Evaluation of gastric viability is also important in patients undergoing emergency surgery for incarcerated hernias. The disadvantage of the procedure is lack of reproducibility; retching or belching of the patients during examination which can alter the location and anatomy of GEJ and excess air insufflation of the stomach which may exaggerate the size of the hernia. Oesophageal manometry is measured at the level of the diaphragmatic crura, however, manometric landmarks differ from that of barium study and endoscopy, and identified by pressure inversion point. In patients with a paraesophageal hiatal hernia, placement of the manometry catheter across the lower oesophageal sphincter and below the diaphragm can be difficult. The procedure is planned before surgery to exclude motility disorders like achalasia and to verify the integrity of oesophageal peristalsis. pH testing is not essential in the diagnosis of a hiatal hernia, but is necessary for a quantitative analysis of reflux in a case of hernia which can be relieved by antireflux surgery. Confirmation of abnormal gastro-oesophageal reflux on upper endoscopy, or increased oesophageal acid exposure on pH monitoring is necessary prior to operative intervention in patients with a sliding hiatal hernia. Nuclear medicine studies, transoesophageal echocardiogram (TEE) and endoscopic ultrasound (EUS) can also demonstrate hiatal hernias but are not routinely used for diagnosis. The main aim of treatment in symptomatic patient is to reduce the acid reflux. Lifestyle modifications are the first line of management and include weight loss, elevating the head of the bed preferably by 8 inches during sleep, avoidance of meals for atleast 2–3 hours before bedtime and elimination of ‘trigger’ foods. According to the SAGES Guidelines updated in April 2013, the treatment protocols are provided on the basis of types of the hiatus hernia. Type I (sliding) hiatus hernia are usually managed with proton pump inhibitors for 8 weeks, once or twice daily depending on the symptoms. Other alternatives are H2 receptor antagonists and antacids. If the patient is not relieved symptomatically by medication, surgical repair like laparoscopic fundoplication (Nissen or Toupet) can be done. Steps of fundoplication comprise surgically relocating the gastro-oesophageal junction below the diaphragm, maintaining an oesophagogastric angle to prevent gastro-oesophageal reflux, constructing a gastric wrap around the distal oesophagus to support the distal oesophageal sphincter and increase its resting pressure, and finally approximate the diaphragmatic hernia to narrow the widened hiatus. In all symptomatic Type II, III and IV hiatus hernia, surgical repair is strongly recommended as the primary treatment, particularly those with acute obstructive symptoms or which have undergone volvulus. Routine elective repair of completely asymptomatic paraesophageal hernias may not always be indicated and determinants for surgery should include the patient’s age and co-morbidities. These fundoplication surgeries are described later in the text. Gastric volvulus is an uncommon entity and characterized by abnormal rotation of the stomach (usually >180°) along the long or short axis, leading to variable degree of gastric obstruction, which can be of a closed-loop type resulting in strangulation. The exact incidence of gastric volvulus is unclear as patients with a chronic presentation may never be diagnosed.2 Approximately, 10%–20% cases of gastric volvulus occur in children less than 1 year, while the other 80% cases are detected in adults, of which 80%–90% are often encountered in the fifth decade of life.3 No significant predilection is seen with either sex or races. Gastric volvulus can be classified on the basis of aetiology, axis of rotation and duration of onset. While classification based on axis of rotation by Singleton is widely accepted and more relevant to the radiologist, classification based on duration of onset of symptoms is more relevant in clinical assessment. On the basis of axis of rotation (Fig. 8.2.15), gastric volvulus is divided into organoaxial volvulus, mesenteroaxial volvulus and the rarest combined/mixed type. Organo-axial volvulus, by far, the most common type of gastric volvulus, encountered often in the elderly, is characterized by rotation of the stomach along its long axis, that is, cardio-pyloric axis, and resulting in an ‘inverted stomach’ with a horizontal orientation, in the form of anterosuperior displacement of the antrum and posteroinferior rotation of the fundus, so that the greater curvature is displaced superiorly and lesser curvature caudally in the abdomen (Fig. 8.2.16). The site of obstruction in this type of volvulus is at the cardia or at pylorus. It has a higher predisposition towards strangulation and ischaemia. Organo-axial volvulus also shows association with the paraoesophageal hernia or diaphragmatic defects, which allows abnormal movement of the stomach along the long axis. If the degree of rotation is less than 180 degrees, the patient may have an incomplete or partial volvulus which is not completely obstructing and may be asymptomatic; it may be more appropriate to describe it as organo-axial position rather than volvulus. Mesenteroaxial volvulus is less commonly encountered (30%) and accounts for about 29% of cases, is reported more often in young adults or children. It occurs when the stomach rotates around its short (transverse) axis, that is, line connecting the midpoint between the lesser curvature and the greater curvature of the stomach, leading to a vertically oriented stomach, with anterior rotation of the pylorus, antrum and resultant displacement of the antrum above the gastro-oesophageal junction (Figs 8.2.17 and 8.2.18). The site of obstruction is usually at antropyloric region. It is usually not associated with a diaphragmatic defect and is often idiopathic. The third and rarest type of gastric volvulus is the combined type when the stomach shows both rotation along the short and long axes. Based on aetiology of rotation, gastric volvulus can be subdivided into either primary or secondary. Primary gastric volvulus representing 25%–30% of the cases, is more common in the adults. It occurs in the subdiaphragmatic location due to abnormality in the gastric fixation, exclusively because of disruption, laxity or absence of the gastric ligaments which anchor the stomach in place. These ligaments namely the gastrohepatic, gastrocolic, gastrophrenic, gastrosplenic and gastropancreatic ligaments, along with gastro-oesophageal junction and pylorus, provide anchorage and fix the stomach in place intra-abdominally, and prevent abnormal rotation of the mesentery. The primary gastric volvulus usually presents with the mesenteroaxial type of anatomical configuration. Around 70% of patients present with secondary gastric volvulus occurring due to abnormal rotation around the lead point formed by associated disease. The most common association of gastric volvulus is seen with paraoesophageal hiatus hernia. Other causative factors of volvulus include congenital or traumatic diaphragmatic hernia, diaphragmatic paralysis, eventration, connective tissue disorders, previous surgery, adhesions, peptic ulcer, neoplasm, splenomegaly or absence of the spleen and colonic overdistension. On basis of clinical presentation and speed of onset, gastric volvulus can be acute, subacute or chronic. Acute gastric volvulus is a surgical emergency and usually presents with sudden onset epigastric pain, distension, non-bilious vomiting or severe retching, depending on the degree of obstruction. The Borchardt triad of acute volvulus comprises severe epigastric pain and distention, retching but inability to vomit, and difficulty or impossibility of passing a nasogastric tube and may be observed in 70% of cases. Other symptoms include hematemesis due to mucosal sloughing in stomach gangrene or mucosal tear due to retching, dysphagia and dyspnea. Even after prompt treatment, mortality of acute gastric volvulus can be up to 30%–50%, likely secondary to gastric ischaemia, perforation or necrosis resulting from severe gastric obstruction (closed-loop obstruction) causing vascular compromise, making it a life-threatening surgical emergency. Chronic gastric volvulus represents spectrum of diseases from long-standing partially obstructed volvulized stomach with incomplete gastric obstruction on the one side, and intermittent volvulus of stomach with recurrent episode of acute symptoms as the other presentation. Chronic gastric volvulus usually presents with intermittent complaints of vague epigastric pain, non-bilious vomiting, dysphagia or early satiety, dyspepsia and sometimes regurgitation. Due to nonspecific clinical symptoms, diagnosing gastric volvulus is very challenging on the first instance and requires a high clinical suspicion. Diagnostic imaging, along with clinical findings are usually required to achieve the correct diagnosis, and also plays major role in excluding the other common differential diagnosis, like pancreatitis, cholecystitis or pneumonitis etc. The diagnostic modalities useful in suspected cases of gastric volvulus are plain radiographs, upper gastrointestinal contrast (barium) studies, computed tomography and upper gastrointestinal endoscopy. Plain chest and abdominal radiographs may reveal a radiolucent hollow viscus, with or without an air-fluid level, in the chest (when associated with PEH) or upper abdomen. The presence of a nasogastric tube may assist in identifying a malpositioned stomach, and administration of contrast via the nasogastric tube may be confirmatory in this regard. Furthermore, chest radiographs may identify evidence of underlying anatomic abnormalities predisposing to gastric volvulus, including elevated hemidiaphragm due to phrenic nerve palsy or diaphragmatic eventration and rib fractures suggesting prior thoracoabdominal blunt trauma. The plain radiographs may show two air-fluid levels in the antrum and fundus, or a single air bubble with no additional luminal gas in the supine position. A retrocardiac air-fluid level may be seen secondary to hernia and presence of intramural air (gastric emphysema) can be visualized as a radiolucent stripe in the gastric wall. At times a large hiatus hernia may be complicated by volvulus (Fig. 8.2.19). Passage of orally administered contrast like barium can demonstrate the anatomy and also assess the degree of obstruction. As mentioned above, the position of the gastric curvatures and the gastro-oesophageal junction are useful in differentiating the anatomical subtype of volvulus. Computed tomography may be more feasible in an acute emergency setting in order to identify abnormal gastric position, axis and multiplanar reconstructions, especially in the coronal plane, can well elucidate the findings and an associated complication like ischaemia. CT has an overall 90% accuracy in the diagnosis with identification of gastro-oesophageal junction and pylorus lying in close proximity to each other and the transition point at the pylorus known to have 100% sensitivity and specificity. Abnormal antral folds may be seen secondary to the twisting. Stenosis at the neck of the hernia is the CT finding with the second-highest sensitivity (77%–80%) and specificity (94%–97%). CT findings of oedematous or hypoenhancing gastric wall, pneumatosis, pleural effusion or pneumoperitoneum, as indicators of gastric ischaemia are not common but specific when identified. An upper gastrointestinal endoscopy confers both diagnostic and therapeutic benefit, although it may establish the diagnosis in only 28%–45% patients and also, the endoscopic procedure may reduce the volvulus missing the diagnosis by an unwary endoscopist. However, it still remains the best method to detect mucosal ischaemia. Flexible endoscopy also has the advantage of placing a nasogastric tube as it may not be possible with a blind technique at the bedside in the setting of organoaxial volvulus and an obstructed gastro-oesophageal junction. Nasogastric decompression forms the initial management of gastric volvulus with emergent laparotomy or laparoscopy to assess gastric viability, resect gangrenous portion if any and perform de-rotation and gastropexy (fixation of the stomach to the diaphragm and/or the anterior abdominal wall) with or without gastrostomy with repair of secondary factors like paraoesophageal hernia. Erosions are focal shallow areas of ulcerations confined to the epithelium or lamina propria without extending through the muscularis mucosae into the submucosa. NSAIDs are the most common cause of gastric erosions while other incriminated factors include Helicobacter pylori infection, alcohol, viral infections, Crohn’s disease, stress and iatrogenic trauma. Erosions may be visible on double-contrast barium examination obtained with good mucosal coating, as multiple tiny 1- to 2-mm collections of barium, often with a surrounding mound of oedema appearing as a filling defect in the barium pool (Fig. 8.2.20). They are usually located in the gastric antrum and tend to occur along thickened folds.
8.2: Stomach
Introduction
Anatomy
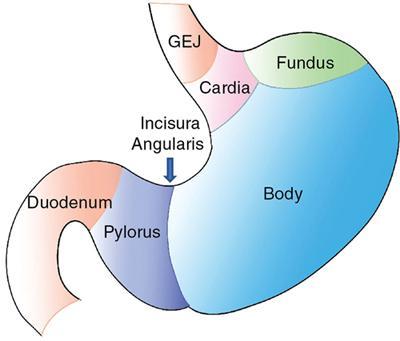
Visceral relations
Peritoneal relations (perigastric ligaments)
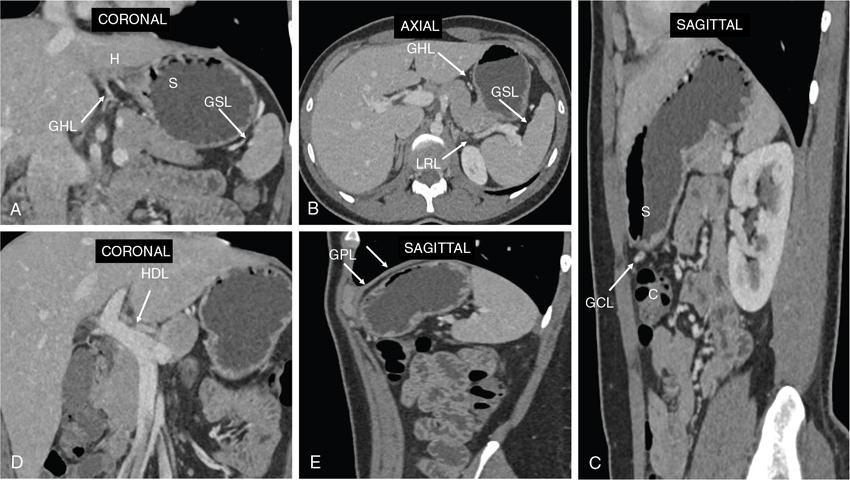
Histology
Arterial supply (Fig. 8.2.3)
Venous drainage
Nerve supply
Lymphatic drainage of the stomach (Fig. 8.2.4)
Embryology of stomach
Imaging techniques
X-ray
Fluoroscopy
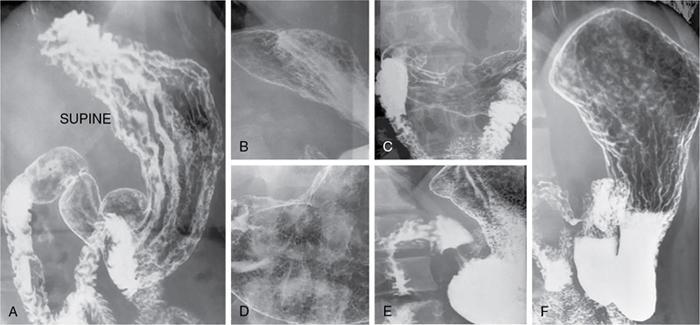
Single contrast upper GI examination
Double contrast upper GI examination (Figs 8.2.5 and 8.2.6)
Ultrasonography (USG)
Computerized tomography (CT)
Magnetic resonance imaging (MRI)
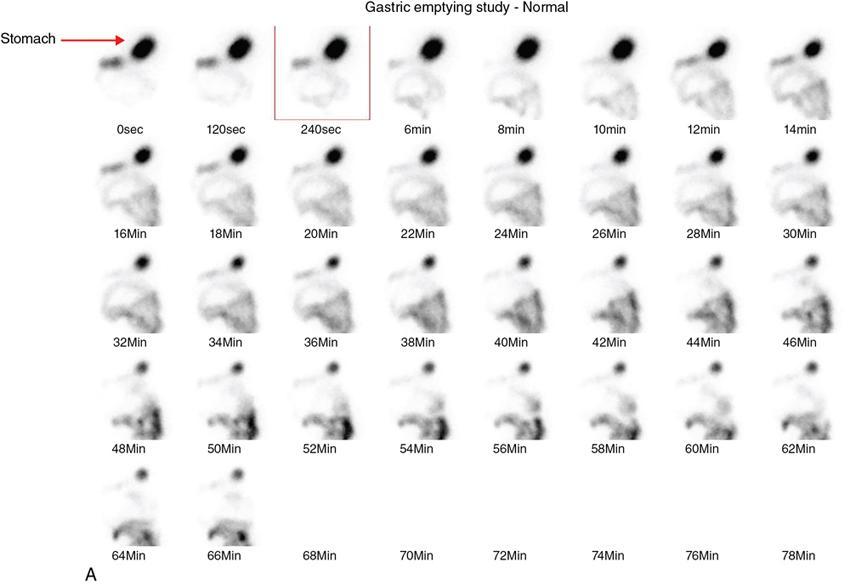
Gastric emptying scintigraphy (Fig. 8.2.7)
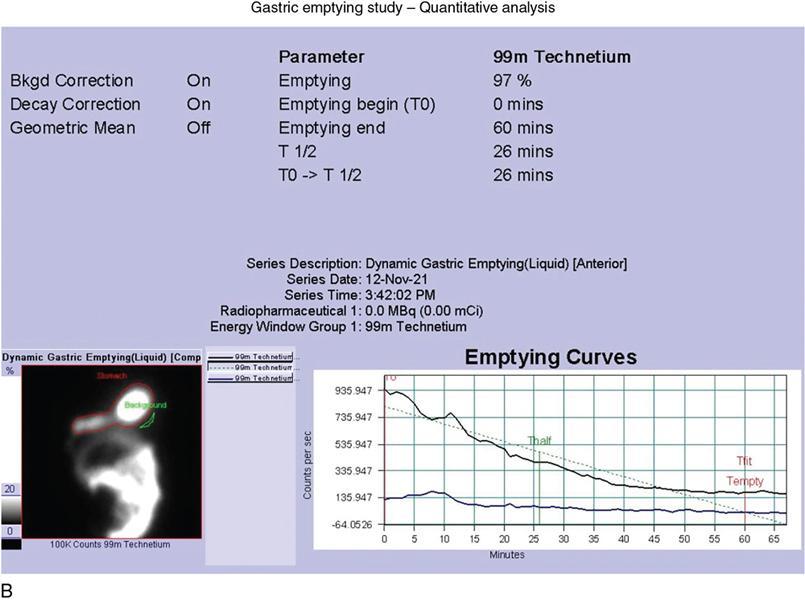
Radionuclide meal preparation
Composition of the meal
Image acquisition
Data processing
Interpretation criteria
Time Point
Lower normal limit for gastric retention
Upper normal limit for gastric retention
0 minute
A lower value suggests rapid gastric emptying
A greater value suggests delayed gastric emptying
0.5 hour
70%
1 hour
30%
90%
2 hour
60%
3 hour
30%
4 hour
10%
Gastro-oesophageal reflux study/milk scan (Fig. 8.2.8)
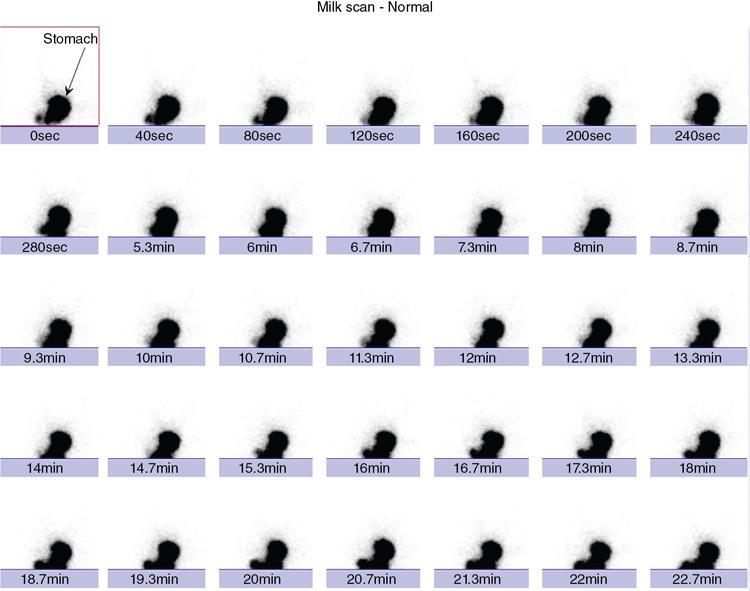
Patient preparation for the test
Radiopharmaceutical
Image acquisition
Data processing and interpretation
Endoscopy
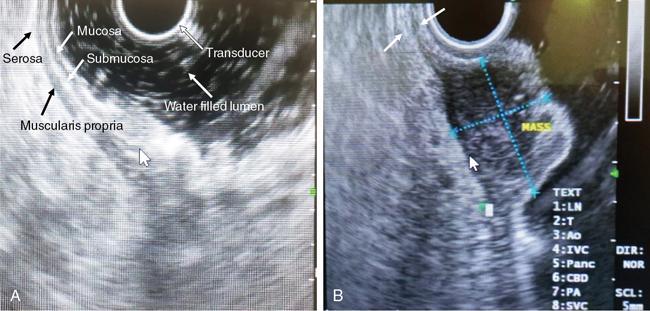
Endoscopic ultrasound (EUS)
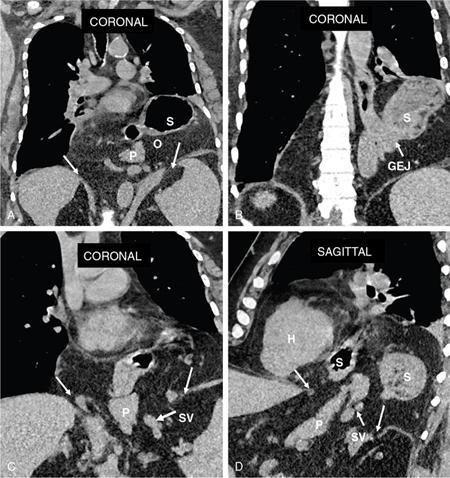
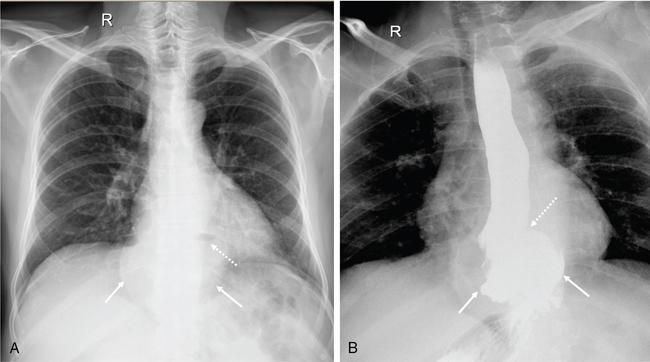
Hiatus hernia
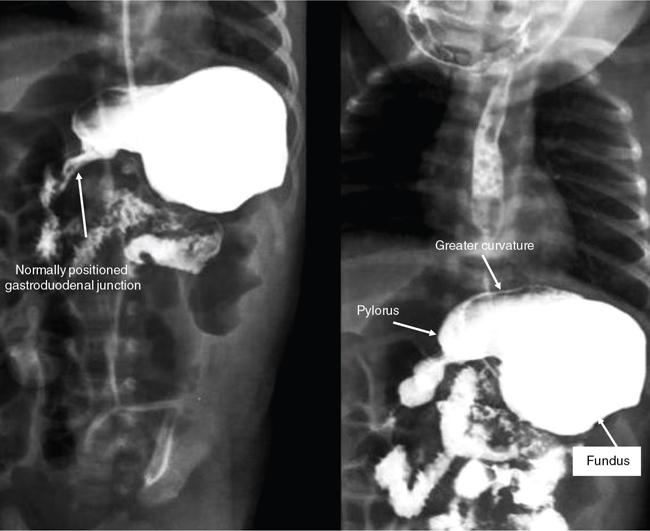
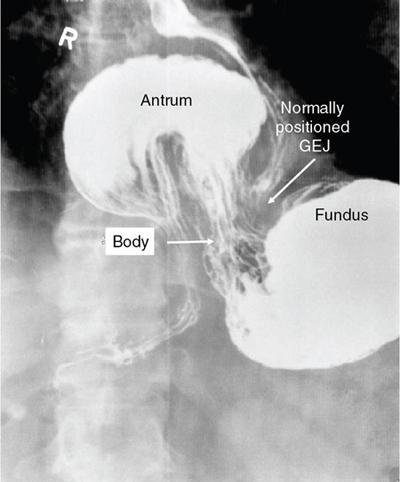
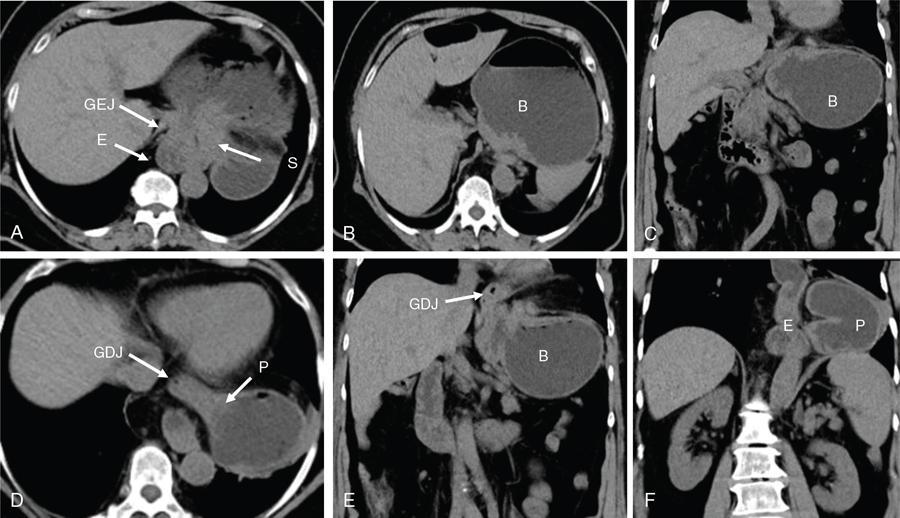
Gastric volvulus
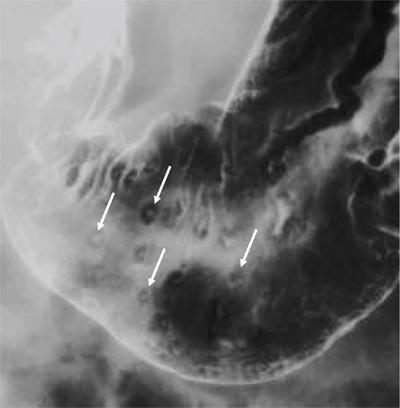
Gastric erosions
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








