Subarachnoid hemorrhage of unknown cause represents approximately 10% to 15% of nontraumatic subarachnoid hemorrhages. The key factors in determining the management strategy for a presumed nonaneurysmal subarachnoid hemorrhage are the distribution, location, and amount of subarachnoid blood. Hemorrhage distribution on computed tomography can be categorized as follows: perimesencephalic, diffuse, sulcal, and primary intraventricular. The extent of the workup required in determining the cause of hemorrhage depends on the distribution of blood. The authors review the potential causes, differential diagnoses, and acute and long-term follow-up strategies in patients with subarachnoid hemorrhage of unknown cause.
Key points
- •
Subarachnoid hemorrhage of unknown cause is a fairly common occurrence and comprises 10% to 15% of all nontraumatic subarachnoid hemorrhages.
- •
Such cases can be categorized into distinct groups based on the location and distribution of subarachnoid blood.
- •
Each group of subarachnoid hemorrhage of unknown cause carries unique imaging recommendations in acute and follow-up contexts.
Introduction
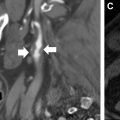
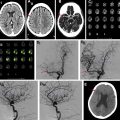
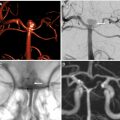
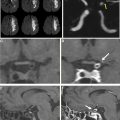
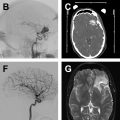
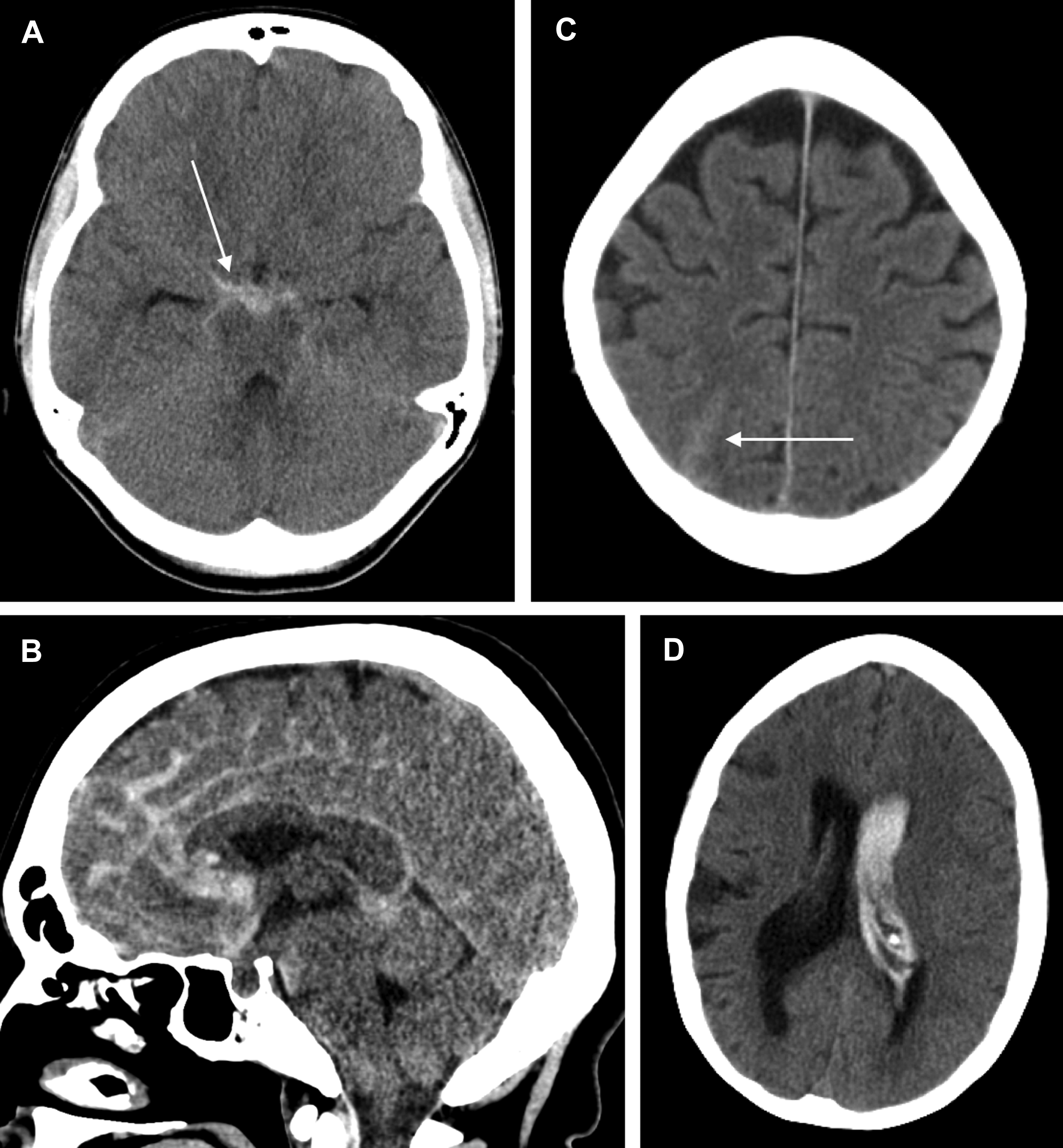
Subarachnoid hemorrhage (SAH) of unknown cause is a relatively common occurrence that represents 10% to 15% of all nontraumatic SAH. The pattern of hemorrhage as demonstrated on presenting computed tomography (CT) may aid in determining the optimal management strategy for SAH of unknown cause. Four patterns of SAH of unknown cause have been described based on the anatomic distribution of blood, each of which can be thought of as distinct clinical entities with characteristic neuroimaging findings, potential underlying causes, and management paradigms: nonaneurysmal perimesencephalic SAH (pSAH), nonaneurysmal diffuse SAH with aneurysmal pattern (dSAH), sulcal or convexal SAH (sSAH), and primary intraventricular hemorrhage (IVH). Examples of each of these are demonstrated in Fig. 1 . Understanding the imaging features and unique characteristics of each of the aforementioned patterns of SAH of unknown cause may aid practitioners in determining which subsequent imaging studies are required in the acute and follow-up settings. In this review, the authors discuss the potential causes and the various neuroimaging characteristics of each of the aforementioned SAH of unknown cause entities. Furthermore, they also discuss follow-up imaging considerations relating to each entity.
Nonaneurysmal perimesencephalic hemorrhage
Background and Potential Causes
Nonaneurysmal pSAH is described as an SAH with blood limited primarily to the perimesencephalic cisterns without angiographic evidence of an aneurysm. For an SAH to be considered a pSAH it has to meet 4 imaging criteria: (1) the center of the hemorrhage must be located in front of the brainstem, (2) no or minute amounts of blood in the interhemispheric or lateral Sylvian fissures, (3) minimal intraventricular hemorrhage, and (4) absence of intraparenchymal hemorrhage. Importantly, these criteria must be found on an imaging study performed within 3 days of the ictus, after which time hemorrhage redistribution may occur. Prior reports have shown that pSAH is readily recognized with excellent agreement between observers. , , The prognosis of patients with pSAH is excellent and rebleeding rarely occurs.
Although pSAH may be the result of vascular lesions including saccular aneurysms, arterial dissection/dissecting aneurysms, posterior fossa or spinal vascular malformations, or hypervascular tumors such as hemangioblastomas, most of the cases of pSAH are without evidence of a structural lesion to account for the hemorrhage. , Nevertheless, the cause of idiopathic pSAH remains uncertain. One plausible theory that has been proposed is that these bleeds may be venous in origin, which may be explained by the fact that many patients with pSAH report performing some variant of a Valsalva maneuver at the time of the initial ictus. , The Valsalva maneuver can result in increased intrathoracic pressure, which blocks internal jugular venous return, thus resulting in increased intracranial venous pressure, potentially increasing the likelihood of venous rupture. Further supporting this theory is the fact that some patients with nonaneurysmal pSAH have variant venous drainage patterns. Patients with pSAH have been found to have more primitive drainage patterns of the basal vein of Rosenthal including drainage directly into the dural sinuses rather than into the Galenic system. This alternative drainage pattern and resulting direct connection of the perimesencephalic and basal veins with the dural sinuses may predispose to increases in venous pressure and venous rupture during sudden increases in venous pressure. Other potential venous mechanisms for pSAH have been described and include stenosis of the vein of Galen straight sinus. ,
Initial Imaging Considerations
Although the ideal initial imaging strategy for patients with pSAH remains uncertain, improvements in CTA techniques have made this the study of choice for initial evaluation of pSAH. Prior reports have demonstrated a negative predictive value of CTA to be 98.6% and potentially as high as 100% with exclusion of cisterna magna pSAH. The role of digital subtraction angiography (DSA) in the initial imaging evaluation of patients with pSAH is also a point of controversy. Prior studies of the diagnostic yield of DSA after a negative CT angiography (CTA) found that aneurysms were present in less than 1% of cases in CTA-negative pSAH. Resultingly, some investigators have opined that a single CTA at initial evaluation without a follow-up DSA may be the appropriate, cost-effective imaging strategy in the radiographic management of patients with pSAH. ,
Nevertheless, separate reports have noted a low sensitivity (roughly 60%–70%) of modern CTA in detecting tiny aneurysms, and therefore, relying only on a single negative CTA in excluding a source of SAH may be inappropriate. Furthermore, CTA is likely not effective in excluding other causes of pSAH such as minute vascular malformations, vasculitis, dissections, and blister aneurysms. , Importantly, the diagnostic yield of a cerebral DSA in the setting of a CTA-negative pSAH can be as high as 10%. , Given the excellent safety profile of diagnostic cerebral angiography and the potential for devastating neurologic sequelae that may result from inappropriately excluding one of the aforementioned pathologic entities, it is preferable in most cases to perform cerebral DSA, even in cases of CTA-negative pSAH. ,
Follow-up Imaging Considerations
Angiographic imaging (computed tomographic angiography, magnetic resonance angiography, and digital subtraction angiography)
The previous paradigm for the diagnostic evaluation of pSAH is 2 negative cerebral angiograms performed within a month of one another. Over the course of recent years, however, studies have demonstrated the diagnostic yield of a second short-term or long-term repeat cerebral angiographic evaluation in the setting of pSAH to be less than 2%. It is therefore likely that a single negative DSA in the context of pSAH is sufficient in excluding an underlying vascular pathology as the cause. Long-term follow-up angiographic studies subsequent to a negative cerebral angiogram at presentation also suffer from a low diagnostic yield and are therefore generally not performed. ,
Brain MR imaging
Brain MR imaging is commonly performed in patients with pSAH and negative angiographic imaging so as to exclude other causes of pSAH such as an angiographically occult vascular malformation or small vascular tumors. However, several large studies have found that the diagnostic yield of a follow-up MR imaging with and without contrast is 0%. , Previous reports of patients with perforator infarcts seen on MR exist, although it is likely that such infarcts may be related to localized vasospasm following pSAH rather than the cause of pSAH itself. In light of these data, routine brain MR imaging following a negative initial angiographic study for patients with pSAH is generally not recommended.
Spinal axis imaging
MR imaging of the cervical spine is commonly pursued to rule out a cervical cord tumor or vascular lesion, which could result in a pSAH distribution of blood products. , Rarely, spinal dural fistulae may result in pSAH. Previous studies on spinal axis imaging for pSAH have found that the diagnostic yield is close to 0%. , A prior study of 51 patients with angiographically negative pSAH did not find any cases in which spinal axis imaging demonstrated a potential source of hemorrhage. Although whether or not to pursue spinal axis imaging in cases of pSAH should be considered on a case-by-case basis, in general, spinal neuroimaging is not recommended in patients without suspicion for spinal involvement (ie, back or neck pain).
Advanced vessel wall imaging
The role of high-resolution vessel wall imaging in the characterization of cerebrovascular diseases has continued to gain traction in recent years. Although a venous cause of pSAH seems to be most likely, it may be that microdissections or microaneurysms related to tiny perforators are the underlying cause of hemorrhage. High-resolution vessel wall imaging enables for superb spatial resolution and can delineate such pathologies as compared with traditional imaging modalities. However, one recently published study of 7 patients with angiogram-negative pSAH found that the diagnostic yield of high-resolution vessel wall imaging in such cases was close to zero. Studies with larger sample sizes are needed in order to corroborate these results.
Nonaneurysmal diffuse subarachnoid hemorrhage with aneurysmal pattern
Background and Potential Causes
Angiographically negative dSAH is defined as SAH with a similar pattern of blood distribution as seen in aneurysmal SAH but without the imaging presence of an underlying lesion as the likely cause of the bleed. The distribution of blood in cases of dSAH includes the interhemispheric and Sylvian fissures, potentially with sulcal and interventricular blood. , Angiographically negative dSAH is relatively common and represents 15% to 20% of spontaneous SAH cases. Although the underlying cause of most cases of angiographically negative dSAH by definition remains uncertain, a ruptured subarachnoid or cortical vein has been purported to be a potential cause.
In most of the dSAH, however, saccular aneurysms represent the underlying etiology. On CTA, it is important to use the epicenter of the bleed in order to determine the location of the potential lesion. In example, dense SAH in the interhemispheric fissure suggests an aneurysm of the anterior cerebral artery or anterior communicating artery, whereas dense blood in the Sylvian fissure highly suggests an MCA aneurysm. Other, nontraumatic causes of dSAH include blister aneurysms or dissecting aneurysms, both of which require careful interrogation of all vascular imaging. , Less commonly, vascular malformations, vasculitis, tumors, and skull base trauma may be the cause of dSAH.
Initial Imaging Considerations
dSAH is diagnosed on initial noncontrast CT imaging studies, which is typically followed with CTA. Recent advances in CT technology, including dual-energy CT has enabled CTA to be an effective modality in the initial angiographic evaluation of dSAH. In contrast to the controversial nature of DSA in the context of pSAH, DSA is a requirement in all patients with dSAH in the setting of a negative CTA. , Because endovascular therapy has become the standard of care in treating ruptured cerebral aneurysms in most cases, many are of the opinion that patients with dSAH should undergo catheter-based cerebral angiogram as soon as possible in lieu of undergoing CTA imaging. This likely represents the most cost-effective strategy in the initial management of these patients. Intriguingly, however, it seems that many centers are moving toward using CTA as the primary diagnostic tool in patients with dSAH, given the noninvasive nature and improving technology with high sensitivities and specificities. However, even with improving CTA technology, ruptured aneurysms may still be missed on initial studies, which further emphasizes the importance of subsequent DSA.
Role of Follow-Up Imaging
Angiographic imaging (computed tomographic angiography, magnetic resonance angiography, and digital subtraction angiography)
In cases of a CTA-negative dSAH, subsequent DSA is requisite in order to rule out any potential treatable causes that may otherwise be undetected on CTA. Prior reports have demonstrated that up to 13% of patients with dSAH who undergo a second angiogram are found to have an aneurysm. , Ruptured aneurysms carry a poor natural history, and therefore detection is of utmost importance so that the appropriate treatment may be pursued. This fact further emphasizes the importance of obtaining a second DSA in the context of a negative initial angiographic study. In some cases of dSAH, a third angiogram may be warranted in the context of 2 previously negative studies. , , Interestingly, the diagnostic yield of a third DSA has been shown to be as high as 5% to 10%, suggesting that, indeed, a third DSA should be considered in patients with 2 prior negative studies. , ,
Brain MR imaging
MR imaging is commonly performed on patients with angiogram-negative dSAH in order to interrogate for the presence of a small vascular mural lesion or a small hypervascular neoplasm. However, the diagnostic yield of a follow-up MR imaging in patients with angiographically negative dSAH has been reported to be close to 0%. Follow-up brain MR imaging in such patients is therefore not recommended in most cases.
Spinal axis imaging
In contrast to brain MR imaging, spinal axis imaging in patients with angiogram-negative dSAH may play a more significant role. Akcakaya and colleagues reported on a series of 40 patients with angiographically negative dSAH, 2 (5%) of whom had a spinal lesion thought to be the cause of the SAH. One patient was found to have a spinal vascular malformation, and the other was found to have a hypervascular neoplasm. The small but definite diagnostic value of spinal axis imaging in such patients has been corroborated by separate reports. , Although there is a small likelihood of spinal pathology resulting in dSAH, spinal imaging in patients with dSAH may be considered, particularly in patients with symptoms that may suggest spinal involvement such as the presence of neck pain.
Advanced vessel wall imaging
A paucity of data exists relating to the role of advanced vessel wall imaging in evaluation of patients with angiogram-negative dSAH. To date, at least a single study performed by Coutinho and colleagues who imaged 11 patients with dSAH has been performed. In this study, 2 patients were found to have focal abnormalities contiguous with the vessel wall, which were likely representative of thrombosed or small, blister-like aneurysms. Additional studies with larger sample sizes are necessary in order to further interrogate the role of high-resolution vessel wall imaging in cases of angiographically negative dSAH.
Sulcal/convexal subarachnoid hemorrhage
Background and Potential Causes
Sulcal or convexal SAH is isolated to the cerebral sulci without extension of blood products into the intraventricular or basal cisterns. In such cases, the hemorrhage may be visualized on CT alone, or on hemosiderin-sensitive MR sequences including gradient recall echo or susceptibility weighted imaging (SWI). sSAH has been demonstrated to represent roughly 5% of all spontaneous SAH. , sSAH carries a broad differential diagnosis and includes potential causes such as reversible cerebral vasoconstriction syndrome (RCVS), cerebral amyloid angiopathy, trauma, moyamoya disease, posterior reversible encephalopathy, superficial vascular malformation, and vasculitis among other potential causes. , To date, the largest series of patients with sSAH included a sample size of 88 patients, with vasoconstriction syndrome and cerebral amyloid angiopathy being the most common causes, each making up approximately 30% of causes of sSAH. Interestingly, in approximately 20% of cases, the cause was noted to be indeterminate. , These data have been corroborated by separate reports.
Imaging Evaluation
Angiographic imaging (computed tomographic angiography, magnetic resonance angiography, and digital subtraction angiography)
Initial imaging evaluation of patients with sSAH should consist of noninvasive vascular imaging such as CTA or magnetic resonance angiography (MRA) so as to identify an underlying vascular lesion such as a small aneurysm, superficial vascular malformation, vasculitis, or vasoconstriction. Noninvasive diagnostic vascular imaging in patients with sSAH is associated with high diagnostic yield, ranging from 57% to 66%. ,
In general, DSA is not indicated in the evaluation of sSAH and should only be considered after noninvasive imaging methods (CTA, MRA, and MR imaging) have failed to demonstrate an underlying culprit. DSA performed after a negative CTA or MRA has been demonstrated to have a small but definite diagnostic yield of less than 5%. , , Consequently, repeat angiography after an a negative initial angiogram is not recommended in most cases but may be considered in cases where initial, noninvasive imaging has failed to demonstrate an underlying cause.
Brain MR imaging
Brain MR imaging plays an instrumental role in the evaluation of sSAH. MR imaging may be helpful in identifying nonvascular causes of sSAH, in addition to determining if a chronic sSAH exists or there is an infarct associated with the original ictus. In terms of the diagnosis and imaging characterization of cerebral amyloid angiopathy and thrombosed mycotic aneurysms, brain MR imaging with SWI sequences remains an essential modality. Moreover, brain MR imaging also plays a crucial role in identifying vascular pathologies that may not be readily visualized on angiographic studies, such as cavernous malformations, posterior reversible encephalopathy syndrome, and cerebral venous thrombosis. Repeat brain MR imaging may be considered in cases where no clear underlying culprit is readily seen on initial imaging.
Advanced vessel wall imaging
High-resolution vessel wall imaging continues to be recognized as a useful modality in differentiating various vascular causes of sSAH. In example, vasculitis and cerebral vasoconstriction syndrome are often clinically similar to one another, and both can present with sSAH. In cases where CSF testing and serum biomarkers are negative or equivocal, high-resolution vessel wall imaging may aid in differentiating between RCVS and vasculitis. Both pathologies may demonstrate multifocal vascular narrowing on angiographic imaging, although in vasculitis, circumferential enhancement of the vessel wall is typically visualized, whereas RCVS does not commonly have vessel wall enhancement at all.
Isolated intraventricular hemorrhage
Background
Primary intraventricular hemorrhage is a rare occurrence comprising less than 1% of intracerebral hemorrhages and refers to hemorrhage isolated to the ventricular system without associated intraparenchymal hemorrhage or basilar cistern SAH. Hypertension represents the most prevalent cause for primary intraventricular hemorrhage with most series reporting that approximately 50% to 80% of patients with primary intraventricular hemorrhage have severe hypertension. Most other series have reported that vascular malformations comprise a sizable minority of cases of primary intraventricular hemorrhage with about 10% to 30% of cases being related to the presence of a vascular malformation. However, this number has been reported to be as high as 52.3% in some instances. Another less-commonly observed cause of primary intraventricular hemorrhage is the presence of a coagulopathy, which is found in roughly 1% to 10% of patients. In roughly one-fifth of cases the cause of intraventricular hemorrhage is unknown. ,
Imaging Considerations
Angiographic imaging (computed tomographic angiography, magnetic resonance angiography, and digital subtraction angiography)
In order to exclude a potential vascular cause of primary intraventricular hemorrhage, obtaining a routine CTA or MRA is, in general, recommended. Approximately 20% of patients will have a vascular lesion such as a small aneurysm or choroidal arteriovenous malformation that is the culprit of the hemorrhage, as mentioned. In some cases these lesions may not be readily apparent on initial CTA or MRA imaging studies. DSA may therefore be considered in cases in which CTA or MRA is negative and an offending lesion is not observed on brain MR imaging. However, the yield of DSA in context of a negative MR imaging, MRA, or CTA remains low at roughly 5% but is nevertheless nonnegligible. , , ,
Brain MR imaging
Patients with primary intraventricular hemorrhage should undergo brain MR imaging so as to rule out the presence of an underlying neoplasm or mass. The presence of intraventricular flow voids or subependymal enhancement should be examined for, which may suggest the presence of an underlying vascular lesion. The yield of repeat brain MR imaging, however, is typically low.
Summary
Here the authors discuss the unique imaging characteristics of 4 patterns of SAH with an unknown cause including perimesencephalic SAH, nonaneurysmal diffuse SAH with aneurysmal pattern, sulcal/convexal SAH, and primary intraventricular hemorrhage. The distribution, location, and amount of subarachnoid blood are paramount features in determining optimal imaging strategies in both acute and follow-up contexts. Following an initial negative angiographic study, patients with perimesencephalic, primary intraventricular, and sulcal SAH generally have a lower diagnostic yield of repeat cerebral angiography than those with diffuse SAH. Nevertheless, DSA may be considered in such cases so as to not miss the presence of an occult lesion that is not readily visualized on noninvasive imaging studies. MR imaging plays a particularly important role in cases of sulcal/convexal and primary intraventricular hemorrhage with negative angiographic studies. High-resolution vessel wall imaging is useful in differentiating between causes of sulcal/convexal SAH; however, more data are required to determine its usefulness in other patterns of idiopathic SAH.
Clinics care points
- •
Although digital subtraction angiography has low diagnostic yield in cases of perimesencephalic SAH, it should nevertheless be pursed in cases of CTA-negative pSAH in order to rule out any potential treatable causes.
- •
DSA is requisite in cases of dSAH, regardless of whether a preceding CTA is positive or negative.
- •
MR imaging, MRA, and CTA should be the primary imaging modalities used in evaluating sSAH, whereas DSA should be more strongly considered in cases where these studies fail to demonstrate an underlying cause.
- •
High-resolution vessel wall imaging is useful in differentiating between underlying causes of sSAH.
- •
MR imaging, MRA, and CTA are first-line modalities in evaluating patients with isolated IVH, although DSA should be considered in cases where these studies are negative.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree