SUPRAHYOID NECK AND RELATED SPACES: INTRODUCTION AND GENERAL PRINCIPLES
KEY POINTS
- This chapter presents the organizational framework for production and interpretation of images of the suprahyroid neck.
IMAGING APPROACH
Techniques and Relevant Aspects
The suprahyoid neck is studied in essentially the same manner as the nasopharynx is evaluated with magnetic resonance imaging (MRI) and computed tomography (CT). The specifics of using these studies in this anatomic region are reviewed in Chapter 184. Problem-driven protocols for MRI and CT are presented in Appendixes A and B.
There is essentially no definitive use for ultrasound in studying the suprahyoid neck outside or parotid and submandibular gland problems that are not the subject of this chapter (refer to Chapter 175).
The approach with radionuclide studies depends on the specific aim of the examination. Most of the current usage is limited to known or suspected cancer evaluation with Fluorine-18 2-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) and bone/gallium scans for inflammatory pathology. Specifics of the use of this physiologic imaging in relation to anatomic imaging are discussed in Chapter 5. FDG-PET should be performed with discernment rather than routinely; its use in this region is very limited, owing largely for the lack of predictability of the metabolic activity of tumors that frequent the region (Figs. 5.12 and 13.12).
Catheter angiography has a limited role in diagnosis and, more importantly nowadays, a critical role in endovascular therapy in lesions that involve the deep spaces of suprahyoid neck that are sometimes transcranial. CT angiography is sufficient for almost all required diagnostic angiography.
Pros and Cons
Introduction
CT and magnetic resonance (MR) are used for the majority of imaging of the suprahyoid neck. Dental-related artifacts can seriously degrade both CT and MR images through the region at the level of the mid oral cavity. The problem is generally worse on CT. The problem is accentuated on MRI when fat suppression is used. These problems usually do not significantly impact medical decision making or treatment planning in suprahyoid neck pathology. MRI sections above the hard palate are not frequently significantly degraded by jaw and other motion.
Angiography is used occasionally in both diagnosis and treatment settings. Catheter angiography is necessary if angioarchitecture and temporal flow dynamics of a lesion, such as might be used to identify a paraganglioma, or an active vascular leak are in question. If not, CT and/or MR angiography will usually suffice; 320 multidetector computed tomography (MDCT) or its equivalent will further reduce the need for diagnostic catheter angiography. Angiography is almost always used following imaging studies, although it is occasionally the primary examination (e.g., intractable bleeding) or a prelude to endovascular procedures.
Radionuclide studies, particularly single photon emission computed tomography (SPECT) gallium and bone scanning, are useful in highly selected cases of infectious disease. FDG-PET is used selectively in cancer patients. It should not be used routinely. Specific radionuclide technique depends on the indications; this is discussed elsewhere (Chapter 5).
Developmental Abnormalities
Developmental Abnormalities are studied primarily with CT when bone detail is critical to diagnosis and three-dimensional planning or craniofacial reconstruction. MR is frequently used to look for an intracranial connection of the parapharyngeal or other deep face process and associated intracranial congenital anomalies (Fig. 8.23).
Potentially cystic congenital lesions with a complex relationship to the nasopharynx and related deep facial spaces are very effectively imaged with MRI, especially in children where one would like to limit exposure to ionizing radiation (Fig. 142.1).
If MRI, CT, or the clinical evaluation suggests a venolymphatic or other vascular malformation, then angiography is occasionally done to confirm this impression and/or used to direct endovascular or percutaneous ablative therapy. Such treatment may be definitive or an adjunct to surgical removal.
Inflammatory Disease
Complex inflammatory disease of the deep spaces making up the suprahyoid neck, such as that associated skull base osteomyelitis, is studied with contrast-enhanced CT and MRI. Radionuclide bone gallium scanning is used occasionally as a supplemental diagnostic study as well as to monitor the progress of antimicrobial therapy (Figs. 13.5, 13.12, 16.1, 16.2, 142.2).
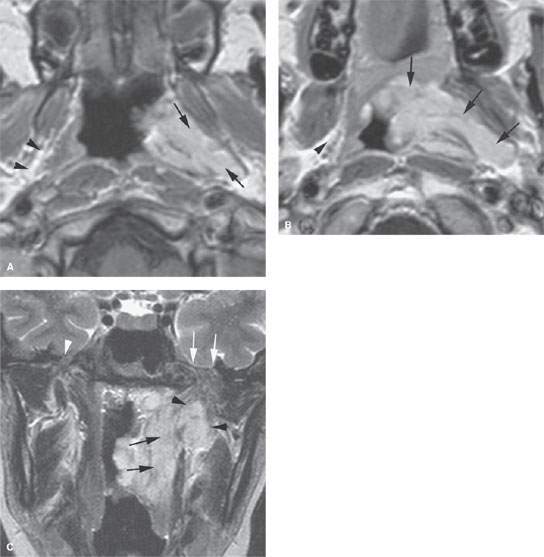
FIGURE 142.1. Contrast-enhanced magnetic resonance (MR) of a patient with a venolymphatic malformation of the nasopharynx and parapharyngeal space (PS). A: The enhancing malformation infiltrates the parapharyngeal fat expanding that space (arrows) compared to the normal fatty prestyloid parapharyngeal space (PSPS) on the right (arrowheads). B: Contrast-enhanced T1-weighted image at the soft palate level showing the malformation to extend into the soft palate and illustrating that the boundaries we create with names are to some extent artificial. C: T2-weighted coronal image showing a submucosal component of the mass medial to the PS (black arrows) and a second component in the PSPS (black arrowheads). The foramen ovale on the left was widened (white arrows) compared to the normal foramen on the right (white arrowhead), suggesting intracranial spread that was not confirmed by this MR study. (NOTE: This patient demonstrates how MR can be used to exclude intracranial structures as an origin of masses presenting in the PS. It also illustrates how imaging techniques can sort out mucosal and submucosal components of the disease relative to those spreading deeper within the parapharyngeal and masticator spaces.)
Masses of Uncertain Etiology
Both CT and MR can be used to demonstrate the presence, possible nature, and extent of a usually submucosal nasopharyngeal or oropharyngeal mass lesion arising in one or more of the spaces of the suprahyoid neck. In some cases, both CT and MR study are necessary for this indication. A variety of sometimes unusual lesions, either benign or malignant, can be encountered in this clinical context. As the initial biopsy results may be inconclusive or negative, MR can be particularly useful to further characterize the lesion. In most patients, MR is the best starting point.
Both MR and CT can supply information needed by the clinician for optimal treatment planning. The studies are complementary. CT studies are easier to execute and more reproducible than MRI because of the short data acquisition time that may result in less image motion degradation; motion degradation on MR studies is much less a problem than at anatomic sites below the hard palate. MDCT systems also now offer high-quality multiplanar imaging, with the image quality often equivalent in diagnostic information to multiplanar MR images. The evaluation of cortical bone on CT is superior to that of MRI, and this can be a critical factor. CT detects calcifications better than MRI, but this is only infrequently significant in suprahyoid neck masses diseases.
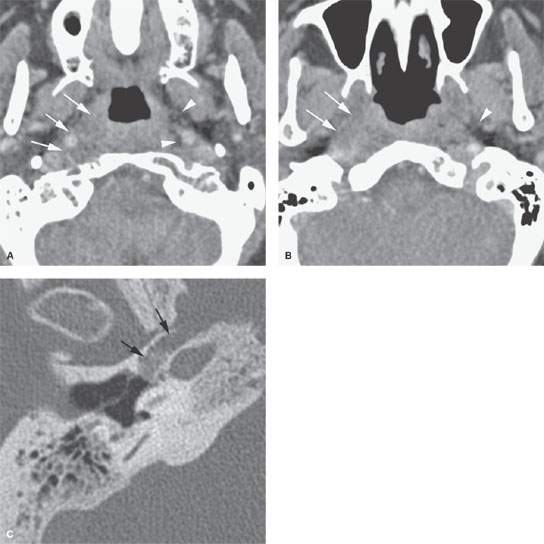
FIGURE 142.2. Parapharyngeal processes are not always mass lesions. The parapharyngeal space (PS) may be involved in aggressive inflammatory lesions and cancers arising in the nasopharynx. This contrast-enhanced computed tomography study demonstrates such disease. A: There is a diffuse although relatively well-circumscribed infiltrating process involving the prestyloid and retrostyloid parapharyngeal space (arrows) on the right compared to the left, where the normal fat (arrowheads) remains intact in these spaces. B: A section superior to that seen in (A) showing obliteration of the fat in the prestyloid parapharyngeal space (arrows) and preservation of the fat (arrowheads) on the left and that the disease essentially tracts along the eustachian tube. C: Bone windows of the temporal bone showing the inciting cause to be necrotizing otitis externa spreading from the hypotympanum into the PS.
FDG-PET has no consistent role in evaluating masses in this region; it should be used selectively when it has a reasonable chance of altering medical decision making or outcomes.
Controversies
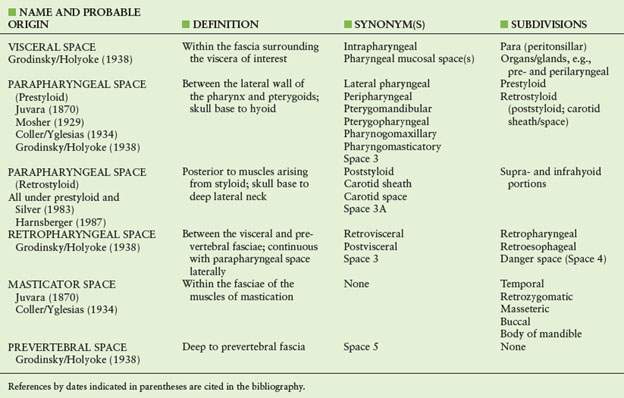
The parapharyngeal space (PS) and other deep tissue spaces and related portions of the suprahyoid neck have been logically divided into prestyloid and retrostyloid (poststyloid) and other subdivisions. This is a very useful concept, especially when considering possible tissues of origin for suprahyoid neck masses. These designations have been around for over 70 years1–12 (Table 142.1). Contemporary renaming of these spaces does not seem appropriate, mainly because these new naming systems are driven by imaging specialists and might impede communication between diagnostic imagers and surgeons, with the latter having used the established terminology for many generations.
NORMAL ANATOMY
Introduction
There are numerous anatomic studies of the fascial sheaths in this region dating back well over 100 years.1–7 These are descriptions of the relatively thick and strong pharyngobasilar fascia, the more flimsy buccopharyngeal fascia, the prevertebral fascia, and the carotid sheath (Table 142.1). CT and MRI show the tissue density of these connective tissue condensations for the most part to be inseparable from the structures they surround, such as muscles and blood vessels. Fat occupies the intervening spaces and provides the tissue contrast necessary to define the spaces, their muscular boundaries, and the structures that pass through the spaces as seen on CT and MR studies (Fig. 142.3). The rendering of these spaces on highly spatially resolved MRI is particularly striking. On such images, it is often possible to resolve the signal of pharyngobasilar fascia from that of surrounding muscles and fat (Fig. 142.4A). The more fat, the better the distinction of the spaces and their contained structures. In older patients, atrophy of the deep musculature with fat replacement makes these deep planes and spaces especially conspicuous.8
Fascial Organization of Deep Spaces
The prevertebral fascia is a well-developed structure that covers the longus colli, rectus capitus and longus capitus muscles. It extends from the base of the skull to the lower limit of the longus colli muscles at approximately the T3 vertebral body1–7 (Fig. 142.3). Swallowing and neck movement require that the pharyngeal structures and carotid sheath move easily relative to the spine and prevertebral space. The pharynx is essentially a muscular tube suspended from the skull base. The fat in the retropharyngeal and parapharyngeal spaces allow for this necessary movement and pharyngeal expansion as well.
The pharyngobasilar fascia holds the lumen of the nasopharynx open. The fascia encloses the nasopharyngeal mucosa, surface lymphatics, and some of its musculature (Fig. 142.4). The fascia lies medial to the superior pharyngeal constrictor and extends above the muscle’s most superior fibers to attach to the base of the skull.
The medial part of the buccopharyngeal fascia is the epimysium of the superior pharyngeal constrictor. This becomes the medial boundary of the PS. Its lateral counterpart is a reflection of deep cervical fascia that covers the deep surface of the parotid gland and pterygoid muscles and, therefore, the masticator space (MS). That lateral component of the bordering fasciae becomes the lateral boundary of the PS. Both of these fascial layers are sparse and loosely applied to their respective muscles of origin to accommodate the changes in size and movement of the pharynx that occurs during mastication and swallowing. These fasciae are not normally clearly visible on CT or MR or even anatomic whole-organ sections (Fig. 142.4).
Detailed Description of Specific Areas
Parapharyngeal Space
The PS is a loose network of fibrofatty tissue that permits free movement of the muscles of mastication and swallowing. Its fat content produces the distinct plane lying between the pharyngeal and pterygoid muscles of the MS. The PS is always visible and always appears relatively symmetric at the nasopharyngeal level (Fig. 142.5). Obliteration of the PS is a strong sign of aggressive pathology.
The PS is divided into prestyloid and retrostyloid (poststyloid) subdivisions. This is a very useful concept, especially when considering possible tissues of origin for parapharyngeal masses. These designations have been around for over 70 years1–12 (Table 142.1).
TABLE 142.1 VARIOUS NAMES OF DEEP SPACES IN THE HEAD AND NECK
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree