Etiology, Prevalence, and Epidemiology
Takayasu arteritis, also known as pulseless disease or Martorell syndrome, is an idiopathic chronic granulomatous arteritis affecting medium and large arteries with a predilection for the aorta and its main branches. It is seen most commonly in Japan, Southeast Asia, India, Mexico, and Brazil. Although it occurs most commonly in girls and women from eastern Asia, it has been recognized worldwide in both sexes. The incidence in North America is very low, estimated to be 2.6 cases per million population per year. The female-to-male ratio is about 10 : 1 in Japan but is likely lower in non-Asians (approximately 3 : 1 in South Africa). The patients are usually between 10 and 40 years of age at the time of initial diagnosis.
In a national survey in Japan from 1998 to 2000, Takayasu arteritis was shown to involve the aortic arch in 59% of cases, descending aorta in 45%, ascending aorta in 30%, left subclavian artery in 60%, left common carotid artery in 40%, brachiocephalic artery in 19%, and pulmonary arteries in 15%. It involved the abdominal aorta in 28% of cases and the renal arteries in 13%. Approximately 28% had concomitant thoracic and abdominal aortic lesions. Abdominal aorta and renal artery involvement is considerably more common in other south Asian countries. Coronary arterial involvement is not uncommon and may be seen in 10% to 30% of all patients at conventional angiography and up to 53% at coronary computed tomography (CT) angiography.
Clinical Presentation
Constitutional symptoms of fever, myalgias, arthralgias, and weight loss are typically present for months to years before the more specific features of the disease become evident. These are usually related to vascular stenosis, the most common being diminished or absent pulses in as many as 96% of patients, typically in association with limb claudication and blood pressure discrepancies. Clinical examination reveals vascular bruits in 80% to 94% of patients, often multiple and affecting mainly the carotid, subclavian, and abdominal arteries. Other common findings are hypertension in 33% to 83% of patients, usually reflecting renal artery stenosis, which is seen in 28% to 75% of patients; aortic regurgitation caused by dilatation of the ascending aorta and separation of the valve leaflets; and left-sided heart failure associated with hypertension, aortic regurgitation, and dilated cardiomyopathy.
Evidence of pulmonary involvement almost always occurs in patients who have systemic findings; rarely, it is the first manifestation of disease. Many patients are asymptomatic despite the presence of clinical signs or radiologic findings of pulmonary disease. Chest pain and hemoptysis occur occasionally.
Various classification schemes have been proposed. The best known is the one by Numano’s group, which divided the disease into six types :
- •
Type I involves only the branches of the aortic arch.
- •
Type IIa involves the aorta only at its ascending portion or at the aortic arch. The branches of the aortic arch may be involved as well. The rest of the aorta is not affected.
- •
Type IIb affects the descending thoracic aorta with or without involvement of the ascending aorta or the aortic arch with its branches. The abdominal aorta is not involved.
- •
Type III is concomitant involvement of the descending thoracic aorta, the abdominal aorta, or the renal arteries. The ascending aorta and the aortic arch and its branches are not involved.
- •
Type IV involves only the abdominal aorta or the renal arteries.
- •
Type V is a generalized type, with combined features of the other types.
In this classification scheme, involvement of the coronary and pulmonary arteries is indicated as C + or P + , respectively.
Pathophysiology
The pathogenesis of Takayasu arteritis is unclear, and familial onset indicates a genetic contribution beyond ethnicity. The disease has been shown to be associated with different human leukocyte antigen (HLA) alleles in different populations and may coexist with autoimmune disorders, including systemic lupus erythematosus, rheumatoid arthritis, and sarcoidosis. Although the HLA association suggests the possibility of an autoimmune process, no specific autoantigens have been identified. Takayasu arteritis has also been associated with infections caused by Mycobacterium tuberculosis and streptococcal species.
The main histologic finding consists of patchy panarteritis involving the large elastic vessels of the systemic and pulmonary circulations. On microscopic examination the vasculitis may be divided into an acute inflammatory phase and a healed fibrotic phase. The acute phase is characterized by vasculitis of the vasa vasorum in the adventitia; infiltration of the media by lymphocytes; and thickening of the intima by mucopolysaccharides, smooth muscle cells, and fibroblasts. The inflammation tends to be most marked in the adventitia, with infiltration of B and T lymphocytes. In the chronic phase there is fibrosis with destruction of elastic tissue and narrowing of the arterial lumen in a patchy distribution, often affecting multiple areas. Marked inflammation and more acute progression may cause degeneration of smooth muscle cells with loss of elastic fibers and subsequent aneurysm formation or rupture of the involved arteries.
Manifestations of the Disease
Radiography
The most common radiographic abnormalities consist of a wavy or scalloped contour of the descending thoracic aorta and dilated of the aortic arch. Aneurysms of the ascending and descending thoracic aorta are seen less frequently.
Computed Tomography
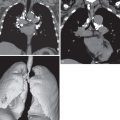
Multidetector CT angiography with or without electrocardiography gating has a high sensitivity and specificity in the diagnosis of Takayasu arteritis. It depicts the presence and extent of luminal stenosis or aneurysm formation and provides information about vessel wall thickness and inflammation and presence of mural calcification. Precontrast CT often shows high attenuation wall thickening relative to the aortic lumen. Postcontrast imaging in early-stage Takayasu arteritis shows aortic wall thickening with the “double-ring” sign. This sign consists of a poorly enhanced internal ring (swollen intima) and an enhancing outer ring (inflamed media and adventitia). In patients with active inflammation, delayed-phase CT obtained 20 to 40 minutes after intravenous injection shows circumferential enhancement of the vessel wall. Aortic ( Fig. 49.1 ) or branch vessel stenosis ( Fig. 49.2 ) is the most common luminal change and occurs in up to 90% of patients. Vascular occlusion ( Fig. 49.3 ) and aneurysm formation ( Fig. 49.4 ) are less common disease manifestations, with the latter being associated with fatal rupture or aortic dissection ( Fig. 49.5 ). Aortic wall calcification is usually transmural (see Fig. 49.5 ), occurs in up to 27% of patients, and is typically seen in chronic Takayasu arteritis. Pulmonary artery involvement occurs in 15% to 63% of patients ( Fig. 49.6 ). The most common abnormalities consist of stenosis or occlusion of segmental or subsegmental branches, usually in an upper lobe. These abnormalities are associated with localized areas of low attenuation and decreased vascularity on high-resolution CT and perfusion defects on technetium-99m–macroaggregated albumin perfusion scintigraphy. The main limitations of CT are radiation dose and need for iodinated contrast material. Coronary artery involvement is common, affecting greater than 50% of all patients. Common manifestations include ostial and nonostial coronary artery stenoses and occlusions ( Fig. 49.7 ) and, less commonly, aneurysm formation.