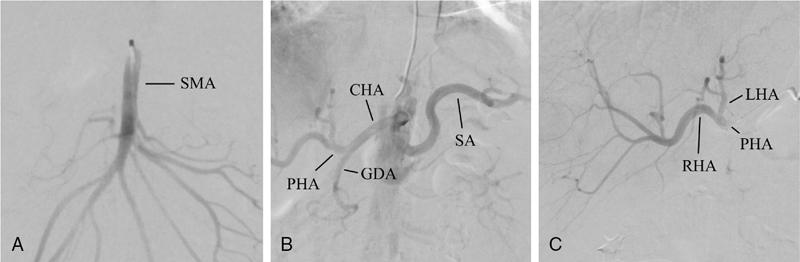
Rory L. Cochran, Sanjeeva Kalva This chapter discusses the rationale, clinical considerations and practical aspects relevant to the different types of transarterial (TA) therapies commonly used for the management of primary and secondary liver neoplasms. Other interventional oncology techniques, such as percutaneous ablation (e.g. chemical, microwave, radio- and cryo-), are discussed elsewhere. Liver-directed TA therapies represent a dynamic class of indispensable tools within the interventional oncologist’s arsenal. Although conventional TA treatments have evolved significantly since the early proof of concept studies decades ago, all currently used modalities share a common approach: transcatheter, intraarterial delivery of a therapeutic agent (e.g. bland embolic, chemotherapeutic). TA therapy may be used alone or combined with other modern anticancer treatments. Table 9.25.1 lists the major forms of TA therapy. The conceptual basis of TA therapy is built upon the unique perfusion characteristics of the normal liver and the altered blood supply observed in many liver cancers. Normal hepatic perfusion arises from a mixture of portal venous and hepatic arterial blood, with approximately two-thirds arising from the portal venous system and the remainder contributed by the hepatic arterial system. Interestingly, most primary and secondary liver neoplasms predominately derive their blood supply from the hepatic arterial system. This unique characteristic led to the development and refinement of the TA therapies available today. Apart from the technical aspects necessary to perform a given approach, the specific treatment objective varies based on the theory behind the technique. For transarterial hepatic embolization procedures (bland embolization or chemoembolization), the treatment objective is to occlude a tumour’s arterial blood supply to promote tissue ischemia and necrosis. Complete occlusion is often necessary to maximize the ischemic effects on a given tumour. In contradistinction, radioembolization is optimally performed when embolization is incomplete, enabling the formation of reactive oxygen species. Optimal radiation-induced tissue necrosis requires oxygen for free radical generation. Thus, for TA therapies not utilizing ionizing radiation, complete arterial occlusion is often the goal (embolize to stasis), whereas in radioembolization, incomplete embolization is typically preferred. The desired level of arterial embolization with emerging viral-based and immunologic therapeutic approaches remain to be determined. TA therapies are unique amongst cancer management strategies, serving as a form of local, regional or locoregional therapy, intended to preferentially treat tumour cells in their microenvironment, theoretically minimizing many of the potentially toxic chemotherapeutic side effects. Indeed, TA therapy is distinct from conventional systemic anticancer therapy where agents are administered intravenously, most often via a central venous catheter, exposing much of the body to a given therapeutic. Locoregional therapy, such as in chemoembolization, may theoretically reduce the systemic toxicities associated with many anticancer agents while permitting the administration of higher intratumoural doses locally. Modern cancer care continues to grow increasingly complex, affirming the importance of a multidisciplinary approach supported by a team of specialists. Towards brevity, this chapter focuses on the important details related to TA treatments for neoplasms either arising from or metastasizing to the liver. Comprehensive information focusing on the diagnosis, staging, prognosis and treatment for these malignancies is covered elsewhere. It is important, however, to emphasize that in many patients with liver predominant metastases (e.g. oligometastatic colorectal cancer) or patients with primary liver cancer (e.g. solitary hepatocellular carcinoma), surgical resection can be curative. Unfortunately, many patients present either later with more advanced disease or they are deemed not suitable surgical candidates. As a result, a considerable number of patients continue to benefit from advances in TA therapy. Table 9.25.2 lists the main hepatic lesions effectively treated with TA therapy. Hepatocellular carcinoma (HCC) Large adenomas Large haemangiomas Intrahepatic cholangiocarcinoma (ICC) Neuroendocrine tumours (NET) Colorectal cancer (CRC) Uveal melanoma Other (breast, sarcoma, ovarian, etc.) Preprocedural noninvasive imaging, either computed tomography (CT) or magnetic resonance imaging (MRI), should ideally be performed within 30 days of the prospective treatment date. Temporal proximity demonstrates high correlation of findings between cross-sectional imaging and angiography. For melanoma, primary liver and neuroendocrine tumours, multiphase contrast enhanced imaging in the arterial and portal-venous phases should be performed. Also, depending on tumour type, positron emission tomography (PET/CT) may be appropriate. The most important cross-sectional imaging features relevant to preprocedural planning are shown in Table 9.25.3. During the preprocedural patient evaluation period, it is important that the patient and provider share a common understanding of the treatment objectives. For some patients, TA therapy may be curative, while for others the treatment may be purely palliative. Thus, TA treatment intent should be classified as one of the following: The many factors influencing the decision to proceed with TA therapy are identical to those for a patient undergoing standard diagnostic mesenteric angiography. The most important patient specific factors informing the decision to proceed are listed in Table 9.25.4. Is eGFR >30? Contrast material, sedation medications, local anaesthetics INR <1.5 Platelets >50K Benzodiazepines or opioids Performance Score (ECOG) ≤2 Total bilirubin <2 mg/dL Child-Pugh Score – up to B7 Presence of biliary obstruction Biliary stent/biliary drainage catheter Sphincterotomy Bilioenteric anastomosis Evaluation of a patient’s renal function, usually via an estimated glomerular filtration (eGFR), is required prior to administering contrast material. Most providers consider an eGFR >30 an acceptable threshold for administering intravenous contrast material. Nonionic iodinated contrast material is the preferred contrast agent for standard angiography. Gadolinium-based contrast agents and carbon dioxide gas may also be used under specific circumstances. Details regarding their use are discussed elsewhere. It is important to inquire about any known adverse drug reactions or drug allergies to iodinated contrast material, local anaesthetics (e.g. lidocaine), sedatives (e.g. benzodiazepines or opioid analgesics) or antibiotics. Evaluation of hepatic dysfunction is important in the preprocedural period. Liver function tests are routinely performed in the patient evaluation period, which can inform the provider of a given patient’s overall liver function and guide treatment decisions. Several different scoring systems have been developed to evaluate clinically significant liver disease. Most early studies were used either as a means to predict surgical outcomes in cirrhotic patients (Child-Turcotte-Pugh score) or predict mortality in end-stage liver disease patients (model for end-stage liver disease). The Child-Pugh score ranges from 1 to 15, placing patients in one of three classes (A, B or C) and is derived from both subjective and objective data. The criteria used in the Child-Pugh scoring system include: Child-Pugh class A represents the least severe liver disease and class C represents the most severe. In contrast to the Child-Pugh system, the model for end-stage liver disease (MELD and later updated MELD- Na) scoring system ranging from 6 to 40 that utilizes only objective data and is used to predict 3–month survival. The MELD-Na criteria include: Mobile device applications and online websites are available that enable efficient calculation of both scores (mdcalc.com). For routine mesenteric angiography performed under standard sterile technique, we do not advocate for prophylactic antibiotics. Prophylactic antibiotics may be considered in specific patient populations. In particular, special consideration should be made for patients with a history of prior biliary intervention or abnormality. Prior work has demonstrated that patients with acquired abnormalities of the biliary system, such as from prior endoscopic papillotomy, indwelling biliary stent or bilioenteric anastomosis, appear to be at increased risk of developing a liver abscess following TA therapy. In an effort to mitigate infectious complications within this cohort, some providers advocate for prolonged periprocedural antibiotic treatment with bowel flora coverage, yet the clinical evidence remains controversial. Although many factors inform appropriate patient selection and the type of TA treatment, a patient’s overall functioning in daily life, or performance status, also remain very important. Performance status, which broadly measures how the patient’s disease impacts their daily activities, represents a critical factor influencing overall management decisions. Incorporating performance status into the management paradigm guides safe and effective treatment choices and minimize potential harms. The most commonly used metric is the Eastern Cooperative Oncology Group (ECOG) performance status scale, which was developed in the early 1980s and is summarized in Table 9.25.5. Technical factors and patient specific factors can also influence a given study or treatment. Two important modifiable variables include: site of access and patient sedation. Although radial arterial access has gained significant favour over the past decade in large part due to patient comfort, the common femoral artery remains the dominant access site for patients undergoing TA therapy. This is due to the technical challenges associated with longer catheters required for hepatic arterial catheterization using radial artery access. Patient sedation also becomes uniquely important when performing the diagnostic angiogram step of the study. More specifically, it is important for the operator to remain cognizant of the degree of patient sedation in order to obtain effective patient cooperation during initial image acquisition. If the patient becomes too sedated and is unable to perform effective breath holds, there will be significant motion artefact secondary to respiration limiting the quality of the diagnostic images, potentially leading to diagnostic errors and off target treatments. Most often there is minimal discomfort during the diagnostic imaging component of a TA therapeutic intervention that would require substantial sedation. General anaesthesia is uncommonly used for TA therapies, typically reserved for unique cases (e.g. strong patient preference or paediatric patients). Monitored moderate sedation may be accomplished with a short acting benzodiazepine, such as midazolam, while immediate analgesia may be addressed with a short acting opioid, such as fentanyl. Given all TA therapies rely on a common approach, here we briefly describe the practical aspects of conventional mesenteric angiography followed by some procedural considerations. The following procedural steps were adapted from Liu et al. as a practical guide: Contrast material injection for the major mesenteric angiograms previously described should be performed using a power injector to provide adequate opacification. Table 9.25.6 lists some practical parameters for mesenteric angiography. On the day of a given procedure, the patient should fast for 4–6 hours. At many institutions, including our own, patients are often prophylactically given medications before, during and after a treatment which is aimed at minimizing pain, nausea and inflammation. Prophylactic cocktails are often tailored to the patient based on their medical history, contraindications and other factors. Commonly used prophylactic agents are listed in Table 9.25.7. Acetaminophen Oral 975 1x Ondansetron Haloperidol Dexamethasone IV IV IV 4 1 8 1x 1x 1x Ondansetron Haloperidol Metaclopramide Promethazine IV IV IV IV 4 1 5–10 6.25 1x 1x 1x 1x Acetaminophen Tramadol Oxycodone Ketorolac Oral Oral Oral IV 650 50–100 5–10 15 q6h q6h q4h 1x
9.25: Transarterial therapy for liver tumours
Introduction
Physiologic basis of ta therapy
Patient selection
Hepatic tumours
Cholangiocarcinoma
Metastasis
Tumour size
Tumour number
Location(s) using the Couinaud segment classification system
Morphology
Vascular involvement (e.g. portal vein thrombosis or vascular invasion)
Presence of extrahepatic disease
Factor
Details
Renal function (eGFR)
Known allergies
Current anticoagulation or bleeding diathesis
Current sedatives/analgesics
Functional Status
Liver function tests
Prior violation of ampulla of Vater
Grade
Description
0
Fully active, able to carry on all pre-disease performance without restriction
1
Restricted in physically strenuous activity but ambulatory and able to carry out work of a light or sedentary nature, for example, light housework, office work
2
Ambulatory and capable of all selfcare but unable to carry out any work activities; up and about more than 50% of waking hours
3
Capable of only limited selfcare; confined to bed or chair more than 50% of waking hours
4
Completely disabled; cannot carry on any selfcare; totally confined to bed or chair
5
Dead
Technique
Artery
Injection Rate (mL/sec)
Total (mL)
Abdominal aorta
20
20
Celiac axis
5
25
SMA
5
25
Hepatic
Hand
10
Periprocedural Timing
Class
Medication
Route
Dose (mg)
Frequency
Pre
Analgesic
Intra
Antiemetic
Post
Antiemetic
Analgesic
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Radiology Key
Fastest Radiology Insight Engine






