Inclusion criteria
Exclusion criteria
Age ≥45a years
No informed consent
T1–2 (≤2.0a cm; mammography, ultrasound)
Non-compliance
cN0 (examination, ultrasound)
Age <45a years
Histopathology: ductal-invasive
Tumour size >2.0a cm
Unifocal
cN+
Multifocal lesions
Lobular or non-invasive
Metastases
All patients should undergo a triple assessment (mammography, ultrasound and histopathology) and the results should be discussed in a multidisciplinary team meeting (comprising breast surgeons, medical and clinical oncologists, radiologists, histopathologists, etc.). These experts work collaboratively and within their scope of practice in order for the most appropriate decision to be made regarding the best possible treatment for the patient before selecting the patient for TARGIT.
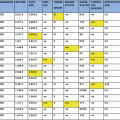
The above criteria for patient selection are taken from the eligibility criteria for the TARGIT-A trial, but it should be noted that both the American Society for Radiation Oncology (ASTRO) (Smith et al. 2009) and the European Society for Therapeutic Radiology and Oncology (ESTRO) (Polgár et al. 2010) have released guidelines (Table 17.2) for the selection of patients for accelerated partial breast irradiation (APBI) outside the context of a clinical trial. It is important to note that these guidelines are based upon all trials and methods of APBI, which are similar (but not identical) to TARGIT. They conclude that there appears to be a group of patients in whom the use of PBI could be deemed safe as the sole method of radiotherapy treatment but for some modalities there is limited recurrence and survival data.
Table 17.2
Comparison of ASTRO and ESTRO guidelines with TARGIT-alone eligibility criteria
Organisation/trial | Age (years) | Tumour size (cm) | Histopathology | Exclusion criteria |
|---|---|---|---|---|
ASTRO | ≥60 | ≤2 | Ductal invasive or other favourable subtypes | cN1, R+, L1, EIC, mf, HR negative |
ESTRO | >50 | ≤3 | Ductal invasive or other favourable subtypes | cN1, R+, L1, EIC, mf |
TARGIT-A | >45 | ≤2 | Ductal invasive | L1, R+, cN+, mf, EIC |