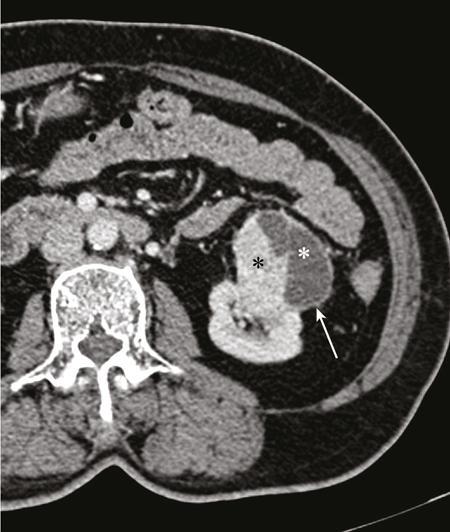
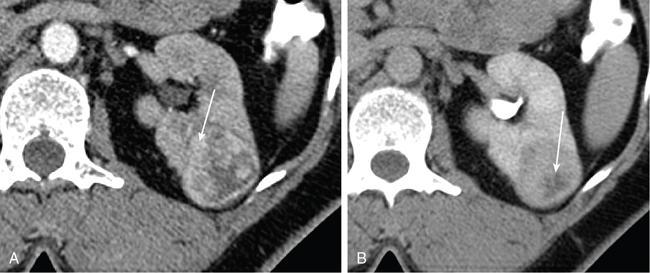
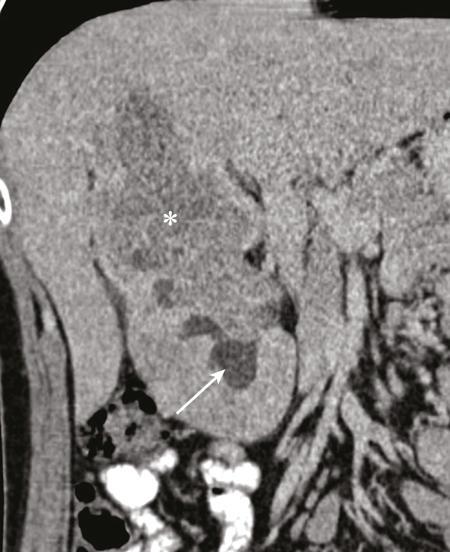
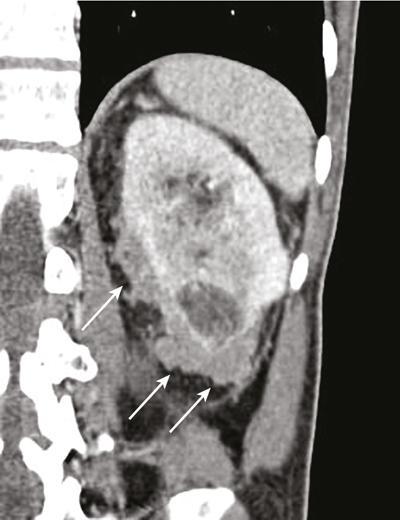
IMAGING OF RENAL MALIGNANCIES IN ADULTS Aparna Katdare, Palak Bhavesh Popat, Nilesh P. Sable, Ganesh Bakshi, Suyash Kulkarni Detection of renal masses has increased remarkably in recent times owing to wide-spread use of cross-sectional imaging. Imaging plays an important role in characterizing renal masses and is indispensable in treatment planning in renal malignancies. The most commonly encountered malignant renal masses in practice include renal cell carcinoma (RCC), urothelial carcinoma, lymphoma and metastases. Urothelial carcinoma and lymphoma have been covered in detail in separate chapters. RCC constitutes nearly 90% of all renal malignancies with a higher incidence in North America and Europe than India, Africa and China. With an increase in the number of cross-sectional studies performed, especially in Western population, the incidental diagnosis of RCC has increased in recent times, with such incidentally diagnosed lesions presenting at earlier stages with better prognosis and reduced rates of recurrence. The median age of presentation of RCC is 64 years according to the Surveillance, Epidemiology and End Results (SEER) program database and almost a decade earlier in Indian population, with a definite increasing risk of RCC with increasing age. Males are affected about 2–3.5 times more than females. In the Indian population, patients have been seen to present at a later stage as compared to the Western population. Amongst the various postulated risk factors, there is convincing evidence that smoking, hypertension, obesity and acquired renal cystic disease increase the risk of RCC. Alcohol intake and physical activity have been found to be associated with reduced risk of RCC. Trichloroethylene and cadmium exposure have been postulated as likely causes as well. Two to four per cent of RCCs are hereditary, with association of various genetic disorders such as Von Hippel Lindau (VHL) syndrome, hereditary papillary renal carcinoma, hereditary leiomyomatosis RCC, Birt-Hogg-Dube syndrome, chromosome 3 translocation and tuberous sclerosis (TCS1, TCS2). The WHO classification of 2016 (Table 11.25.1.1) stratifies tumours of the kidney into different subtypes based on cytoplasmic and architectural features, tumour location, background renal disease and molecular alterations. Clear cell renal cell carcinoma Multilocular cystic renal neoplasm of low malignant potential Papillary renal cell carcinoma Hereditary leiomyomatosis and renal cell carcinoma-associated renal cell carcinoma Chromophobe renal cell carcinoma Collecting duct carcinoma Renal medullary carcinoma MiT family translocation renal cell carcinomas Succinate dehydrogenase-deficient renal carcinoma Mucinous tubular and spindle cell carcinoma Tubulocystic renal cell carcinoma Acquired cystic disease-associated renal cell carcinoma Clear cell papillary renal cell carcinoma Renal cell carcinoma, unclassified Papillary adenoma Oncocytoma 8310/3 8316/1a 8260/3 8311/3* 8317/3 8319/3 8510/3a 8311/3a 8311/3 8480/3a 8316/3a 8316/3 8323/1 8312/3 8260/0 8290/0 Metanephric adenoma Metanephric adenofibroma Metanephric stromal tumour 8325/0 9013/0 8935/1 Nephrogenic rests Nephroblastoma Cystic partially differentiated nephroblastoma Paediatric cystic nephroma 8960/3 8959/1 8959/0 Clear cell sarcoma Rhabdoid tumour Congenital mesoblastic nephroma Ossifying renal tumour of infancy 8964/3 8963/3 8960/1 8967/0 Leiomyosarcoma Angiosarcoma Rhabdomyosarcoma Osteosarcoma Synovial sarcoma Ewing sarcoma Angiomyolipoma Epithelioid angiomyolipoma Leiomyoma Haemangioma Lymphangioma Haemangioblastoma Juxtaglomerular cell tumour Renomedullary interstitial cell tumour Schwannoma Solitary fibrous tumour 8890/3 9120/3 8900/3 9180/3 9040/3 9364/3 8860/0 8860/1a 8890/0 9120/0 9170/0 9161/1 8361/0 8966/0 9560/0 8815/1 Cystic nephroma Mixed epithelial and stromal tumour 8959/0 8959/0 Well-differentiated neuroendocrine tumour Large cell neuroendocrine carcinoma Small cell neuroendocrine carcinoma Phaeochromocytoma 8240/3 8013/3 8041/3 8700/0 Renal haematopoietic neoplasms Germ cell tumours The morphology codes are from the International Classification of Diseases for Oncology (ICD-O) {917A}, Behaviour is coded/0 for benign tumours; /1 for unspecified, borderline or uncertain behaviour; /2 for carcinoma in situ and grade III intraepithelial neoplasia; and /3 for malignant tumours. The classification is modified from the previous WHO classification (756A), taking into account changes in our understanding of these lesions. aNew code approved by the IARC/WHO Committee for ICD-O. Of these, the most common entity is the clear cell subtype, while papillary and chromophobe subtypes are less common. The different subtypes show varied biological behaviour, treatment response and prognosis. The 2017 guidelines by American Urological Association (AUA) as well as 2019 guidelines by European Society of Medical Oncology (ESMO) recommend multiphase cross-sectional imaging by either CT or MRI for renal mass characterization and staging. AUA 2017 guidelines recommend renal mass characterization on the basis of tumour complexity, contrast enhancement and presence or absence of fat. MRI has an upper hand over CT in characterizing subtle mass enhancement, cystic lesions and lesions less than 2 cm. ESMO recommends contrast-enhanced CT study of the chest, abdomen and pelvis for renal mass staging. Bone scan and brain CT or MRI can be considered if indicated by clinical or laboratory investigations. For characterization of renal masses, a multiphase CT or MRI study is recommended. CT study constitutes an unenhanced study followed by contrast injection and acquisition of corticomedullary phase at 40 seconds, nephrographic phase at around 100 seconds and delayed phase at around 5 minutes. MRI protocol includes T2-weighted single-shot fast spin-echo 2D sequences, axial T1-weighted 2D sequence with in-phase and opposed-phase gradient echo imaging, precontrast and postcontrast imaging with a 3D T1-weighted spoiled gradient recalled sequences in corticomedullary phase at 30 seconds, nephrographic phase at 100 seconds, 180–210 seconds and delayed phase imaging at 5 minutes and diffusion-weighted imaging with multiple b-values 0–50, 400–500 and 800–1000 s/mm2. The ACR White Paper on CT imaging of incidental renal mass recommends using the following descriptors for characterizing renal masses: Given the prognostic implications, it is worthwhile for radiologists to know the imaging features that may help discriminate between the common histological subtypes of RCC on various imaging modalities. This is the most common histological type of RCC, accounting for about 70% of cases. These are exophytic tumours with a heterogeneous appearance due to the presence of necrosis, intratumoural haemorrhage, cystic components with septations and calcific foci and hence have a more heterogeneous appearance on cross-sectional imaging than the other subtypes (Fig. 11.25.1.1). Necrosis is seen more often in larger masses and higher tumour grades (Fig. 11.25.1.2). On CT, depending on the tumour composition, these are seen as heterogeneous lesions, show marked contrast enhancement in the corticomedullary phase with washout on nephrogenic phase (Fig. 11.25.1.2). On MRI, these characteristically show high signal intensity on T2W sequences (Fig. 11.25.1.3). The presence of intracytoplasmic fat in the clear cells of the tumour is reflected in the loss of signal in opposed phase images on chemical shift imaging, which is seen in nearly 60% of these tumours. A pseudocapsule may be seen, which is best appreciated on T2-weighted imaging on MRI; the presence of a pseudocapsule has a high negative predictive value for perinephric extension (Figs. 11.25.1.3 and 11.25.1.4). Conversely, larger tumours with higher grades often have interrupted pseudocapsule and hence tend to be irregular, spread into the perinephric fat with renal vein and inferior vena cava (IVC) invasion (Figs. 11.25.1.5 and 11.25.1.6). On diffusion-weighted imaging, clear cell RCCs have been seen to have higher ADC values than nonclear cell RCCs, and lower-grade tumours have been seen to have higher ADC values than higher-grade tumours. The clear cell variant has worse prognosis than the papillary and chromophobe subtypes, presenting at a more advanced stage and being more likely to recur or metastasize (Fig. 11.25.1.7), with lower 5-year survival rates at 44%–69% as compared to 78%–92% for the other two. Papillary RCC comprises about 10%–15% of RCCs. These are slow-growing tumours and hence are well-marginated in contrast to clear cell RCC. As they are hypovascular, their enhancement on CT is significantly less than clear cell RCC (Fig. 11.25.1.8). These show characteristically low signal on T2W images on MRI (Fig. 11.25.1.9). Intracytoplasmic or macroscopic fat is less often seen on MRI imaging as compared to the clear cell variants. Larger tumours tend to be more heterogeneous (Fig. 11.25.1.10). These tumours may sometimes show cystic appearances, mural projections or blood degradation products. Calcifications are more common in papillary variants than clear cell RCC. Multifocality and bilaterality are also more common in these tumours than clear cell variants. These account for about 5% of RCCs. These are less aggressive, more homogeneous and hypovascular lesions than clear cell variants, with intensity of contrast enhancement on cross-sectional imaging being midway between clear cell and papillary variants. They show low to intermediate T2 signals on MRI. A characteristic feature is the presence of a central scar and spoke-wheel enhancement, the latter being a histopathological and imaging similarity between these tumours and oncocytoma (Fig. 11.25.1.11). Other subtypes of RCC are much less common but some may show distinct imaging findings. Multilocular cystic RCCs have excellent prognosis and lack mural nodules within the cystic components, unlike clear cell RCCs with cystic degeneration which show mural nodules. Collecting duct carcinomas, on the other hand, are aggressive tumours with poor prognosis and have medullary origin, and therefore appear similar to transitional cell carcinomas on imaging. Medullary RCCs are associated with sickle cell disease and sickle cell trait and are seen as infiltrating intracalyceal obstructive lesions with associated nodal disease. The imaging work-up of a suspected RCC is aimed at: Localized renal cancer is defined as a disease confined to the renal capsule and refers mainly to stage I and II disease. Nearly 70% of RCCs, especially the lower-stage lesions, are incidentally diagnosed on cross-sectional imaging. Also, amongst incidentally diagnosed renal lesions less than 4 cm in size, about 20% turn out to be benign on histopathology. Asymptomatic incidentally diagnosed small renal masses have an indolent course and better prognosis. Nephron-sparing surgery (NSS) has gathered momentum in recent years due to promising results and prognoses in small lesions. The 2017 AUA guidelines for localized renal masses describe restricted and well-defined indications for radical nephrectomy, bigger role of nephron-sparing procedures such as partial nephrectomy, tumour enucleation and thermal ablation, as well as increasing role for biopsy as well as active surveillance of such lesions. Hence, imaging findings in these lesions become critical in charting management of these patients. The imaging features of common histopathological subtypes of RCCs have already been discussed above. Signal intensity on T2W images and corticomedullary phase enhancement have been seen to be independent predictors of clear cell and papillary RCCs. Further, T2 signal homogeneity can be a predictor for slower growth rate. Hence, in general, multiparametric MRI studies have been shown to be effective in small renal mass characterization and can subsequently guide decisions regarding biopsy, surgery or surveillance. CT is a good alternative in patients with contraindication to MRI. For cystic renal lesions, the Bosniak classification, which stratifies the risk of neoplasia in cystic renal lesions based on the complexity of their appearance (wall thickness, septations, solid component), can be used effectively to decide further course of management. The Bosniak classification originally applies to CT findings but can logically be extrapolated to MRI, USG and Contrast Enhanced Ultrasound (CEUS) as well. Bosniak I and II cysts are benign while Bosniak IIF, III and IV cysts show progressively increasing risk of neoplasia. Given the more indolent course of cystic RCCs as compared to solid lesions and possible complications of interventions, lately there has been a case for even the type III and IV cysts, which previously would be operated, to be followed up, especially if patient has existing comorbidities or if the solid component is minimal. Initial follow-up would be at 6 months, followed by annual imaging. AUA 2017 guidelines recommend considering renal mass biopsy if haematologic, metastatic, inflammatory or infectious aetiology is suspected. Once the need for surgery is established in a localized disease, NSS may be considered for stage Ia and Ib disease. To predict perioperative outcomes in NSS, various scoring systems have been proposed for preoperative renal mass evaluation, such as R.E.N.A.L. nephrometry score, PADUA score (Preoperative aspects and dimensions used for anatomical classification), C-index method and mathematical tumour contact surface area (CSA). The popular R.E.N.A.L. nephrometry score takes into account various tumour descriptors that help decide the technical feasibility of NSS and predict surgical outcomes. These include tumour radius, exo/endophytic location, nearness to collecting system or renal sinus, anterior/posterior location and location with reference to polar lines. These descriptors need to be commented upon diligently while reporting renal masses (Table 11.25.1.4). Higher scores are seen to correlate with ischaemia time, postoperative urologic complications, higher grade and mortality. These include organ-confined tumours more than 7 cm in size, or tumours of any size which show regional nodal involvement and/or invasion of perinephric tissues but confined to Gerota’s fascia. These include renal vein and IVC invasion (luminal and mural). Generally, radical nephrectomy is recommended by the National Comprehensive Cancer Network (NCCN) in stage II and III RCCs. Both CT and MRI are effective in diagnosing locally advanced disease in RCC. The loss or interruption of pseudocapsule is seen in more infiltrative and aggressive disease and is best seen on MRI. Local infiltration may be in the form of contiguous spread to perinephric tissues or discrete deposits in perinephric fat (Fig. 11.25.1.15). IVC or renal vein invasion could be in the form of intraluminal thrombosis or invasion of the vessel wall and have implications on surgical approach and outcomes. Tumour thrombus can be distinguished from bland thrombus by confirming vascularity within the thrombus on imaging. Doppler evaluation, CEUS, CT and MRI can all be helpful for the same. MRI is better than other modalities for venous evaluation, especially for mural invasion and IVC invasion (Figs. 11.25.1.16 and 11.25.1.17). Right-sided tumours, anteroposterior IVC diameter of 2.4 cm or more at the level of renal hilum and complete IVC occlusion at this level are associated with higher risk of IVC resection. About 16% of patients with RCC have metastases at diagnosis and about 20%–30% of patients operated for local disease develop recurrence or metastases at a later date. The recurrent disease occurs most commonly within 3 years of diagnosis and uncommonly even later in young patients or large tumours. The most common sites of metastases from RCC are lungs, liver, bones, nodes, adrenals and brain. CT scan is the preferred modality for primary staging and surveillance for metastases because of its widespread availability and versatility for diagnosing bone and soft tissue lesions. MRI is preferred for looking for recurrence in postablative lesions, wherein these are seen as new enhancing lesions or show increase in the size of preexisting enhancing components. 18FFluorodeoxyglucose – Positron Emission Tomography (PET)/CT and PET/MRI may be helpful to look for metastatic disease as well. 18FSodium fluoride – PET/CT has been found to be better than CT and bone scan in detecting osseous metastases. RCC is a relatively radiation-resistant tumour, and treatment options tilt in favour of surgical procedures for localized and locally advanced diseases. For metastatic disease, post-risk assignment, the treatment has to be planned. Treatment can be offered as per size and the clinical stage.
11.25: Urogenital malignancies
Renal cell carcinoma
Introduction
Epidemiology
Histopathological subtypes
Renal cell tumours
Metanephric tumours
Nephroblastic and cystic tumours occurring mainly in children
Mesenchymal tumours
Mesenchymal tumours occurring mainly in children
Mesenchymal tumours occurring mainly in adults
Mixed epithelial and stromal tumour family
Neuroendocrine tumours
Miscellaneous tumours
Metastatic tumours
Imaging of renal masses
Guidelines
Scan protocols
Descriptors that help characterize renal masses
Imaging features of common histological subtypes of RCCs
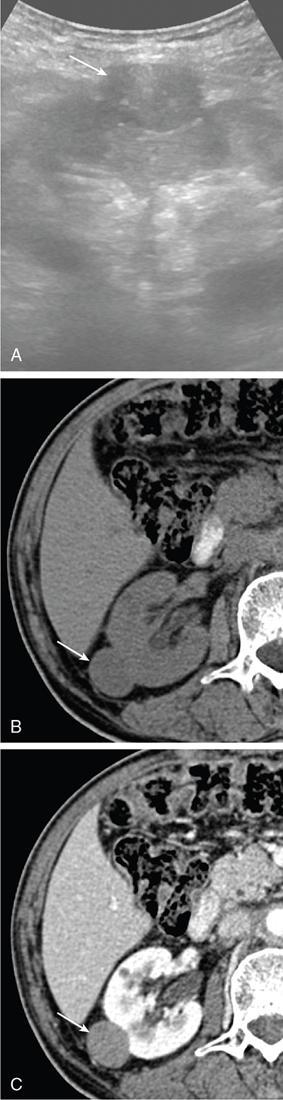
Clear cell RCC.
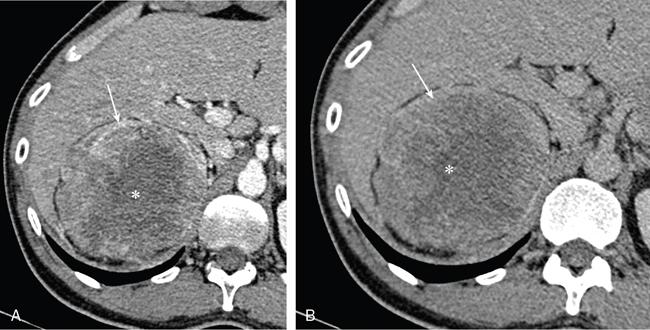
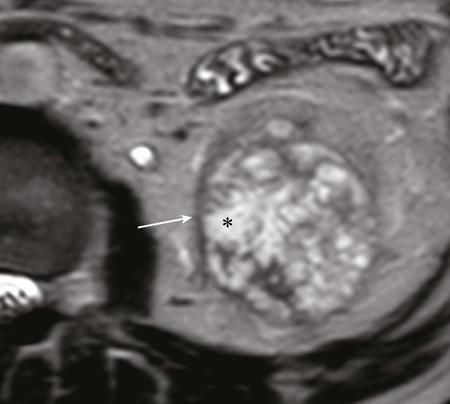
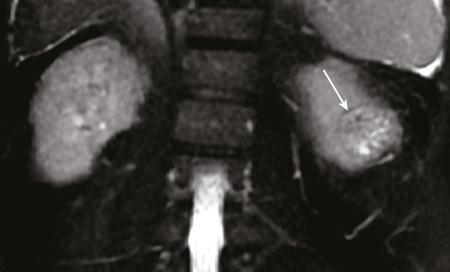
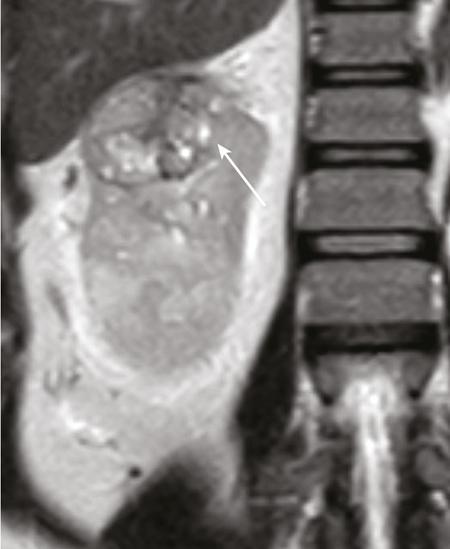
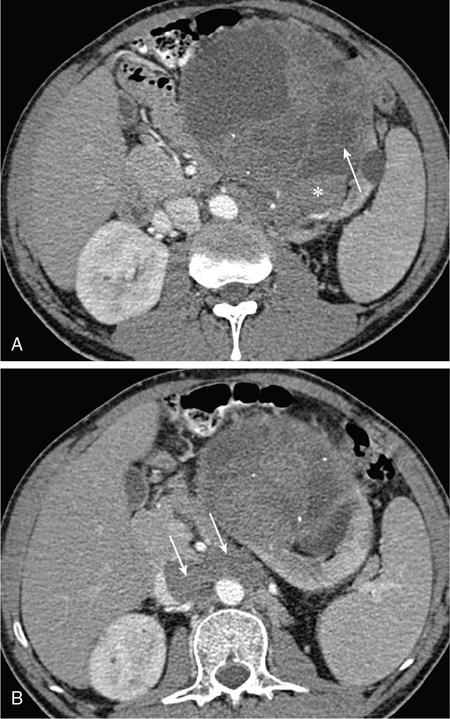
Papillary RCC.
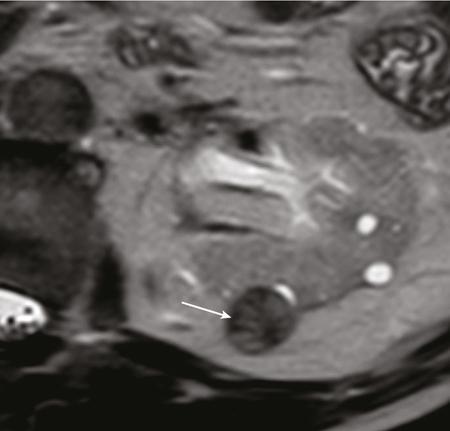
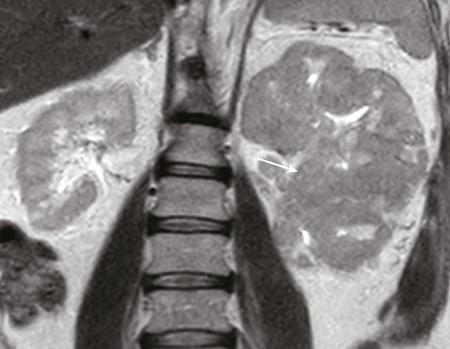
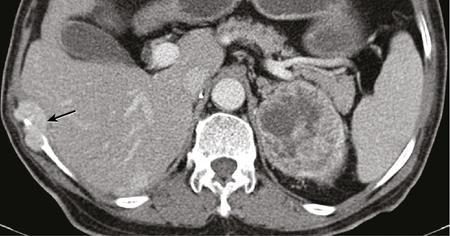
Chromophobe RCC.
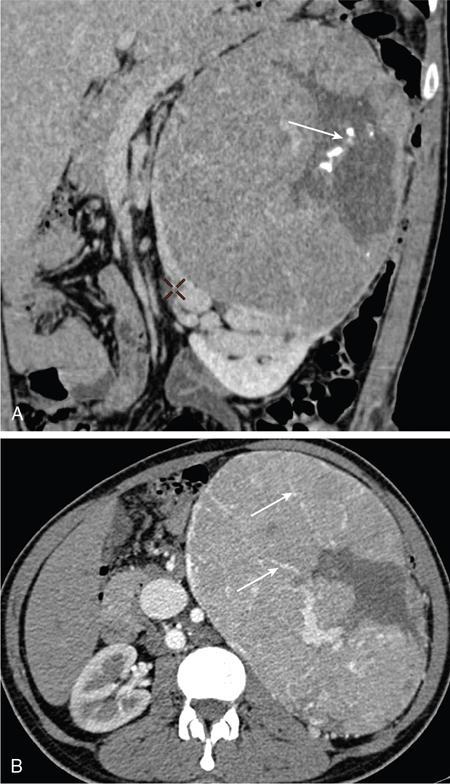
Less common subtypes.
Imaging work-up of suspected RCC – what the treating physicians expect from radiologists
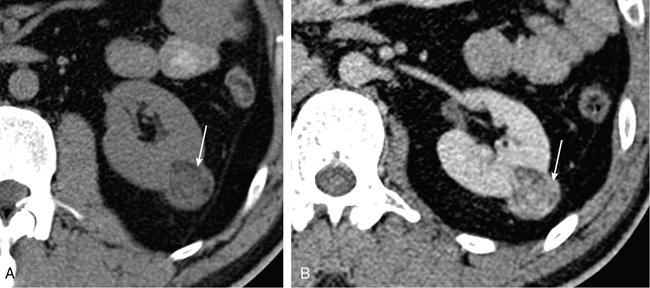
PRIMARY TUMOURS (T)
TX
Primary tumour cannot be assessed
T0
No evidence of primary tumour
T1
Tumour ≤7 cm in greatest dimension, limited to the kidney
T1a
Tumour ≤4 cm in greatest dimension, limited to the kidney
T1b
Tumour >4 cm but ≤7 cm in greatest dimension, limited to the kidney
T2
Tumour >7 cm in greatest dimension, limited to the kidney
T2a
Tumour >7 cm but ≤10 cm in greatest dimension, limited to the kidney
T2b
Tumour >10 cm, limited to the kidney
T3
Tumour extends into major veins or perinephric tissues but not into the ipsilateral adrenal gland and not beyond the Gerota’s fascia
T3a
Tumour grossly extends into the renal vein or its segmental (muscle-containing) branches, or tumour invades perirenal and/or renal sinus fat but not beyond the Gerota’s fascia
T3b
Tumour grossly extends into the vena cava below the diaphragm
T3c
Tumour grossly extends into the vena cava above the diaphragm or invades the wall of the vena cava
T4
Tumour invades beyond the Gerota’s fascia (including contiguous extension into the ipsilateral adrenal gland)
REGIONAL LYMPH NODE (N)
NX
Regional lymph nodes cannot be assessed
N0
No regional lymph node metastasis
N1
Metastasis in regional lymph node(s)
DISTANT METASTASIS (M)
M0
No distant metastasis
M1
Distant metastasis
Stage
T
N
M
I
T1
N0
M0
II
T2
N0
M0
III
T1–2
N1
M0
T3
NX, N0 or N1
M0
IV
T4
Any N
M0
Any T
Any N
M1
Localized renal cancer
SCORE
Component
1 Point
2 Points
3 Points
R (radius, maximal diameter) (cm)
≤4
>4 but <7
≥7
E (exophytic/endophytic)
≥50% exophytic
<50% exophytic
Completely endophytic
N (nearness to collecting system/renal sinus) (mm)
≥7
>4 but <7
≤4
A (anterior/posterior locator)
No points given. Descriptor of ‘a’, ‘p’ or ‘X’ assigned to describe mass location.
L (location relative to polar lines)
Entirely below lower polar or above upper polar line
Mass crosses polar line
50% of mass is across polar line or mass is entirely between polar lines or mass crosses axial midline
Larger organ-confined disease and locally advanced RCC
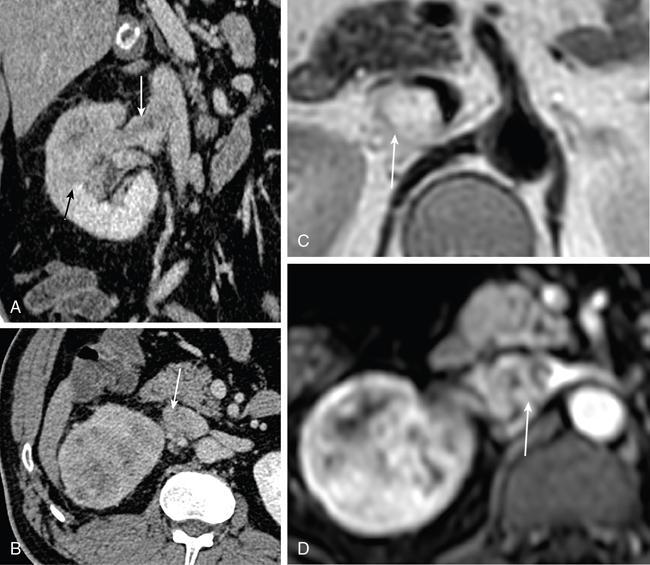
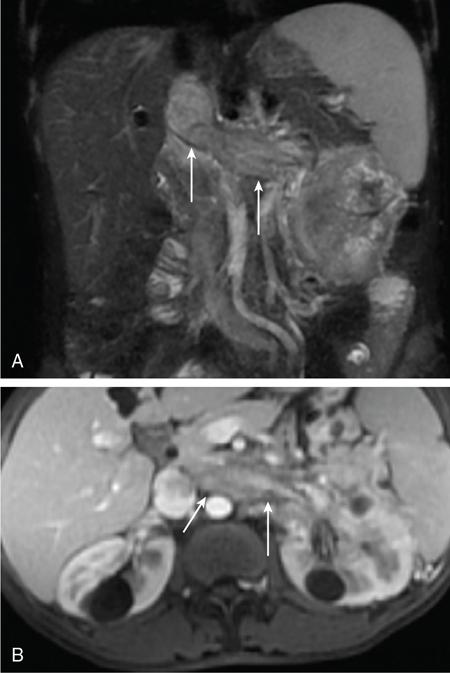
Recurrent/metastatic RCC
Sample reporting template for renal mass
Treatment of RCC
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








