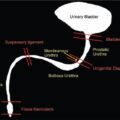
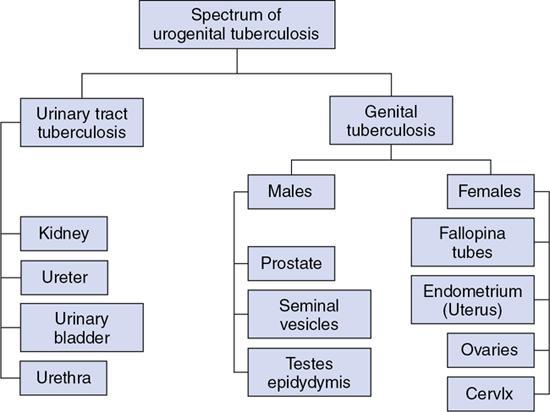
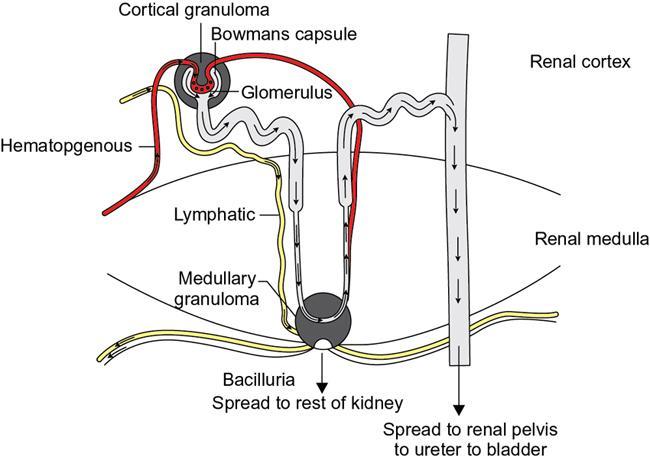
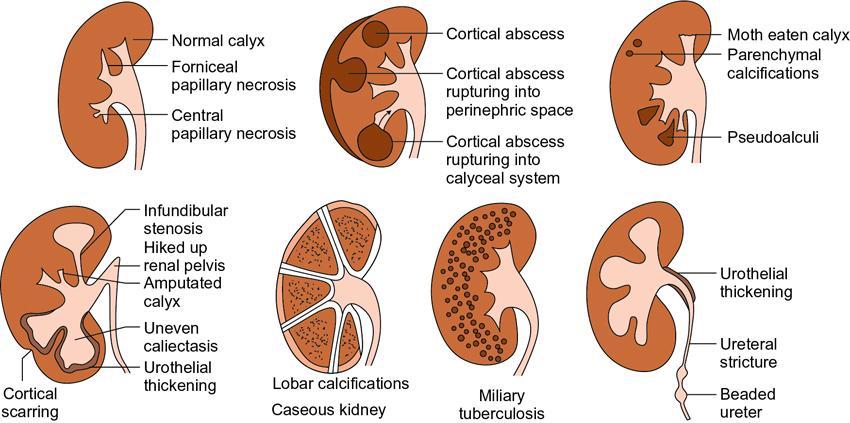
TB is a major cause of ill health and the leading cause of death from a single infectious agent. According to the WHO Global TB report 2019, there was an estimated annual incidence of 10 million cases, with close to 1.5 million having succumbed to the disease annually. India accounts for the highest burden of TB disease in the world, about 27%. Extrapulmonary TB represents 15% of the annual incidence of TB. Genitourinary TB forms about 15%–20% of EPTB. Genitourinary TB is a term coined by Wildbolz in 1937 and encompasses TB of the kidneys, ureters, bladder, prostate, urethra, penis, scrotum, testicles, epididymis, vas deferens, ovaries, fallopian tubes, uterus, cervix and vulva. Involvement of the kidneys is the most common form of TB of the genitourinary tract. Up to 25.8% cases have been reported to have simultaneous involvement of the kidney, ureter and bladder. Urinary tract TB with concurrent active pulmonary TB has been reported in 10%–25.8% cases. Currently, the term urogenital TB (UG-TB) is thought to be more appropriate as urinary tract TB occurs more often than genital TB. Fig. 10.12.2.11.1 depicts the spectrum of involvement in UG-TB. This chapter aims at detailing the imaging findings of urogenital TB and their relevance in the present scenario. Familiarity with the imaging findings helps in early diagnosis, initiation of therapy, monitoring of patients on treatment and prevention of renal failure. Risk factors for developing TB include malnutrition, HIV infection, diabetes, chronic renal and liver disease, alcohol and substance abuse, immunosuppressive drugs, renal transplantation, chronic renal disease, dialysis and end-stage renal failure. MTB is the causative organism of TB. Mycobacterium avium intracellulare (MAC) has been implicated as the causative pathogen more commonly in immunosuppressed individuals. Mycobacterium bovis has also been implicated as a causative organism occasionally. The genitourinary tract is almost always involved via haematogenous dissemination of MTB from a primary TB focus in the lungs. The latent period varies from 15 to 40 years. Intravesical BCG, used as a treatment for bladder cancer, has also been postulated as a rare mode of infection. In the kidneys, the bacilli lodge in the periglomerular capillaries where they form microscopic granulomas. This occurs bilaterally, however, the macroscopic progression of the disease is mostly unilateral. The initial lesions tend to heal either spontaneously in those with intact innate immunity or secondary to ATT given for primary lung TB. These lesions can remain dormant for long periods, usually decades. The breakdown of host defence mechanisms results in reactivation of these tubercular foci. Following reactivation, the cortical granulomas enlarge, undergo caseation, subsequently coalesce and result in local tissue destruction. These lesions extend into the medullary pyramids, to involve the renal papillae and may cause papillary necrosis. Sloughing off of these necrotic areas produce cavities within kidneys. The necrosed granuloma can rupture into the renal pelvicalyceal system leading to the spill of the bacilli into the renal pelvis, ureters, urinary bladder and accessory genital organs. These bacilli then infect the wall of the calyces thereby causing fibrosis and resultant stricture formation, which causes hydronephrosis. The host’s defence and healing mechanism causes fibrosis, stricture formation and calcium deposition, which may contribute significantly to obstruction and progressive renal dysfunction. Thus, the pathogenesis of renal TB involves two competing processes – the destructive effects of the bacilli and the defence mechanism of the host which together ultimately lead to a nonfunctioning, calcified kidney of any size – autonephrectomy. The pathogenesis is depicted and summarized in Figs. 10.12.2.11.2 and 10.12.2.11.3. Occasionally, TB can affect the kidney more insidiously when it spreads to the interstitium to cause tubular interstitial nephritis (TIN). UG-TB is often suspected in the setting of conventional bacterial UTI when repeated cultures are negative or there is a failure of response to antibiotic therapy. It requires a high degree of clinical suspicion as it rarely symptomatic, especially early on, may present with proteinuria, pyuria and microscopic haematuria. Voiding symptoms such as dysuria, nocturia, frequency and urgency of micturition are seen when ureter and lower urinary tract is involved. Flank pain, back pain, renal colic are seen in less than 10% of the cases. The hallmark of renal TB is sterile pyuria. Constitutional symptoms such as fever, malaise, weight loss and night sweats are uncommonly seen.
11. Urogenital tuberculosis
Introduction
Pathogenesis
Clinical features
Laboratory diagnosis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree