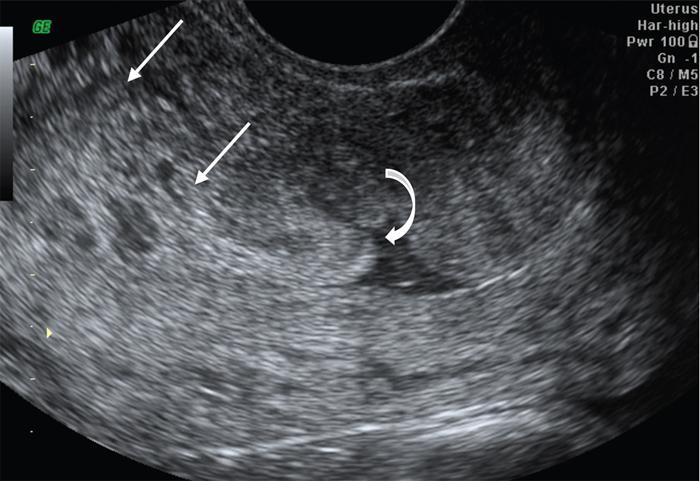
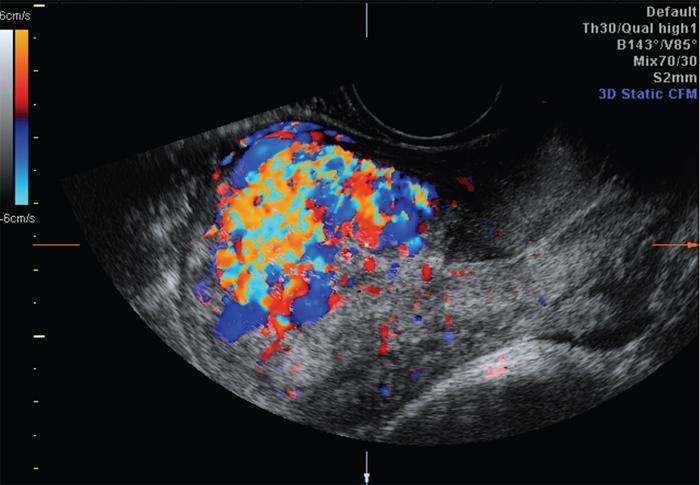
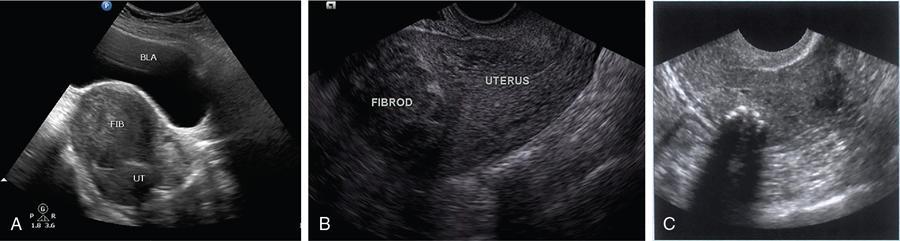
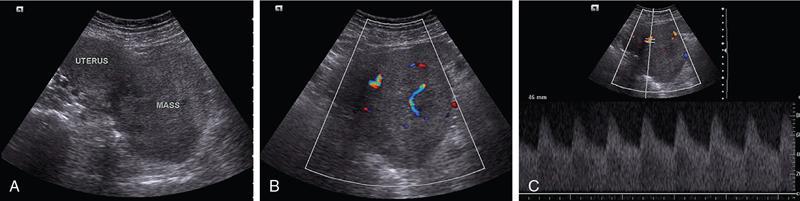
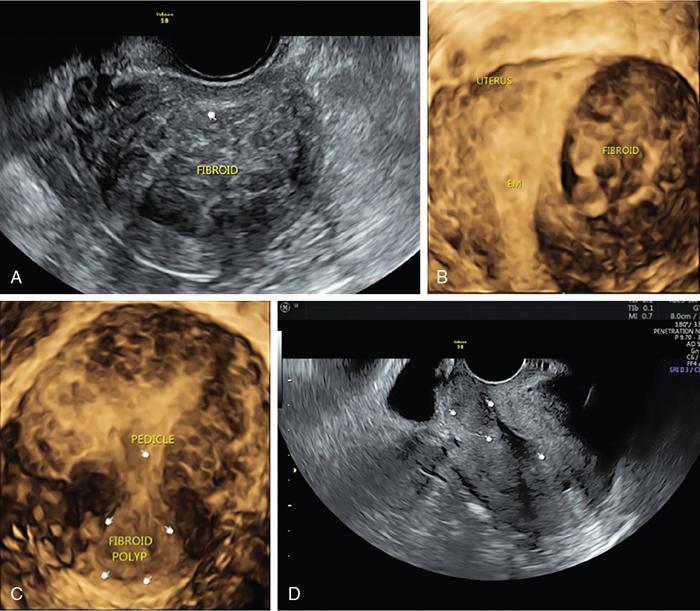
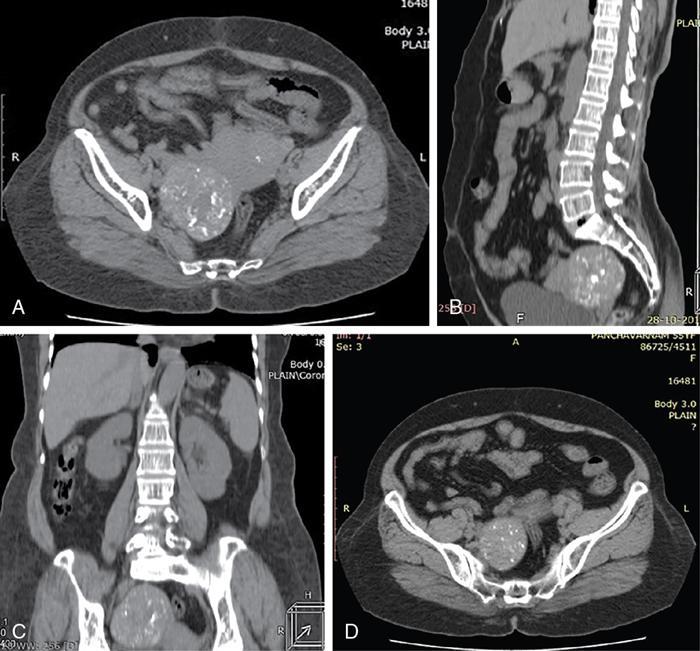
John De Lindsay, Sumathy, Vasumathy, Usha nandhini Ganeshan, N. Sundari ENDOMETRITIS Endometritis is defined as the inflammation of the uterine endometrial layer. Endometritis is frequently observed in the pregnant and postpartum population. Pelvic inflammatory disease (PID) and invasive gynaecological procedures are the most common causes of acute endometritis in nonobstetric population. Endometritis is the most complex asymptomatic and often presents with subclinical form of PID. Ultrasound may reveal minimal particulate endometrial fluid with mildly thickened endometrium showing increased vascularity. Computed tomography (CT) findings include a diffusely bulky uterus due by inflammation, mild fluid distension of endometrial cavity and striking endometrial enhancement in comparison to the adjacent myometrium due to mucosal hyperaemia. ‘Indistinct uterine border’ sign is observed in patients with endometritis, which is defined as the loss of distinction between the uterus and the adnexa. Since it is also observed in the cases of endometriosis or malignancy, it is a nonspecific sign. The age of the patient, clinical presentation, and the biopsy of endometrium helps in differentiating endometritis from other pathologies. UTERINE ARTERIOVENOUS MALFORMATION Uterine arteriovenous malformation (UAVM) is an abnormal connection between the arteries and the veins of the uterine myometrium without an intervening capillary bed. They were first reported by Dubreil and Loubat in 1926. They can be congenital or acquired. Even though UAVMs have varied presentation, uterine bleeding is the most common one. They often follow a pregnancy event. Episodic or occasionally torrential vaginal bleeding can occur. Congestive heart failure secondary to vascular steal can be a rare presentation due to a large UAVM. UAVM is most commonly an acquired condition. Pregnancy has a significant role in their pathogenesis. It is most commonly observed in reproductive age group. UAVMs are frequently observed in the postpartum period, few months after spontaneous miscarriage, surgical evacuation of uterus for miscarriage or medical termination of pregnancy. Infection, inflammation, retained products of conception (RPOC), gestational trophoblastic disease (GTD), gynaecologic malignancies, pelvic trauma and exposure to diethylstilboestrol can also lead to UAVMs. Few cases are also reported in young adolescent and postmenopausal females. UAVMs rarely are congenital and may be also associated with AVMs in other locations. Congenital AVMs Congenital AVMs have a central nidus with multiple feeding arteries and draining veins. In this condition, there is extension of the lesion beyond that uterus and multiple pelvic vessels other than uterine vessels draining into them. That cause of congenital AVMs are thought to be due to failure of embryological differentiation. With the progression of pregnancy, these vessels have the propensity to invade surrounding structures. Iatrogenic Acquired AVMs When the venous sinuses are incorporated into the scars of myometrium after the necrosis of chorionic villi, acquired malformations may arise. In contradiction to congenital AVMs, acquired AVMs multiple fistulous communication between the intramural arteries and the venous plexus. These AVMs may have either dual blood supply or a single supply from the uterine arteries and presents without nidus. Vaginal bleeding caused by pregnancy-related causes must be differentiated from UAVMs. Surgery/evacuation is the appropriate management for the former and the same is contraindicated in the UAVMs and hence an accurate definitive diagnosis is important. Grey-scale ultrasound findings are nonspecific and they are subtle heterogeneity of myometrium with multiple tubular or ‘spongy’ anechoic or hypoechoic areas. In spite of varied presentations such as an intramural uterine, endometrial or cervical mass-like lesions or prominent parametrial vessels, it has minimal mass effect. On colour Doppler, within the myometrium, a region of increased vascularity and most commonly with aliasing is seen. A group of vessels traversing the myometrium running perpendicular to and into the endometrium, from the arcuate vessels, can be seen. It is not specific, as they are also observed in RPOC, GTD, placental polyp and vascular endometrial neoplasm. In pulsed Doppler, multidirectional turbulent flow with intense vascularity can be seen with high peak velocity (mean peak systolic velocity [PSV] = 136 cm/s) and low resistance (mean resistance index = 0.3) flow. The arteries have low pulsatility while the draining veins are pulsatile and show high velocity flow. Magnetic resonance imaging (MRI) offers noninvasive confirmation of the diagnosis of UAVM. T1-weighted and T2-weighted images show multiple serpentine signal voids along the uterine wall, endometrial cavity and parametrium. Contrast-enhanced MR angiography shows intensely enhancing complex serpentine abnormal vessels with early venous return. Catheter angiography can be reserved only for women who require embolization of the AVM. The consent for both diagnostic angiography and therapeutic embolization should be obtained simultaneously to avoid repeat therapeutic procedures. Management of UAVM depends on: UAVM can be treated with medical therapy with hormones, uterine artery embolization or with definitive hysterectomy. Once the diagnosis of a UAVM is confirmed, the treatment option depends on the clinical condition of the patient. The size of the AVM bears no correlation to the need for embolization. Timmerman et al. found that AVMs having PSV of at or above 0.83 m/s, most often requires embolization. Also, the vascular malformation with PSV value below 0.39 m/s does not require embolization. Placental bed subinvolution, enhanced myometrial vascularity (EMV), molar pregnancy or RPOC are the other causes of uterine mass vascular malformations which usually has a mean PSV of 0.52 m/s and does not require embolization. Transcatheter targeted uterine artery embolization is indicated in selected cases, if bleeding persists to a degree that blood transfusions are required to maintain haemodynamic stability or multiple repeated acute hospitalizations. This is preferred over surgical management owing to its immediate minimally invasive and fertility sparing benefits. Gelfoam had been suggested earlier as the material of choice for embolization. Various other embolic materials like polyvinyl alcohol, Histoacryl glue, stainless steel coils, detachable balloons, and haemostatic gelatin are also used nowadays. Normal intrauterine pregnancies after embolization have been reported, proving that an adequate collateral blood supply can develop to support a full-term pregnancy. Gestational Trophoblastic Disease RPOC: Presence of endometrial pathology rather than myometrial origin. FIBROIDS Leiomyomas, also known as fibroids or myomas, are the most common gynaecologic neoplasms, occurring in 20%–30% of women of reproductive age. Symptomatic uterine fibroid accounts for about 30% of hysterectomies performed for women older than 18 years of age and 41% of hysterectomies performed for women from 44 to 64 years of age. Leiomyomas are the benign neoplasms made up of whorled fascicles of smooth muscle and fibrous connective attached to the uterine muscular wall. Even in the absence of true capsule, leiomyomas are well circumscribed and contains a pseudocapsule. Its size is variable, ranging from tiny microscopic to large tumours filling the abdomen. Most commonly, the uterine leiomyomas are seen in the myometrium of uterine corpus. However, it is also seen in the cervix (<5% of cases). On the basis of the location, uterine fibroids are classified as submucosal, intramural or subserosal. This classification is of clinical significance because the symptoms and treatment vary among these subtypes of leiomyomas. Submucosal myomas are further subclassified as 0, I and II depending on the percentage within the endometrial cavity. The first two categories can be removed hysteroscopically (Table 11.15.3.1). Pedunculated leiomyomas are attached to the uterus by a stalk and may be either intracavitary or exophytic (narrower than 50% of diameter of myoma). Rarely, a submucosal fibroid may grow pedunculated and extends into the cervical canal or vaginal canal and its prevalence is about 2.5%. Pedunculated subserosal leiomyomas may undergo torsion, resulting in infarction accompanied by pain. Lateral growth into broad ligament leads to intraligamentous leiomyoma (Table 11.15.3.1). Rarely, a pedunculated leiomyoma may become detached from the uterus (parasitic leiomyoma). Depending on the location, size and number of uterine fibroids, it has varied presentation. Symptoms caused by leiomyomas are classified into three different categories such as abnormal uterine bleeding, pelvic pressure and pain and reproductive dysfunction. Submucosal leiomyomas and intramural leiomyomas are often associated with menorrhagia (Table 11.15.3.1). Because of leiomyoma, uterus may be irregularly shaped and causes symptoms based on their locations. Anterior leiomyomas cause urinary symptoms, whereas the posterior leiomyomas cause constipation. Ureter may be compressed by broad ligament leiomyomas and presents as hydroureter or hydronephrosis. In case of degeneration or torsion of a pedunculated leiomyoma, acute pain may be the presentation. Leiomyomas are an infrequent primary cause of infertility. Malpresentation, pregnancy failures and dystocia are reported. Ultrasound is the first-line imaging modality because it is a cost-effective portable real-time examination that provides good anatomic detail. Leiomyomas appear as a well-defined round or oval hypoechoic solid mass. It is mostly associated with posterior shadowing possibly due to calcifications or interface of the margins of the leiomyoma with the normal myometrium. Degeneration or lipoleiomyoma can be suggested when there is internal echo-poor or hyperechoic foci, respectively. The presence of hypervascularity in a large solitary uterine mass can identify suspicious masses, such as a leiomyosarcoma (LMS). Another study found that there is an increased detection rate of uterine sarcomas while using a cut-off value of 41 cm/s. In the assessment of a uterus enlarged with multiple leiomyomas, ultrasound has FOV limitations. Likewise, it has limited role in the assessment of retroverted uteri, pedunculated subserosal myomas and concomitant adnexal processes. Little has been reported regarding sonographic changes after MR HIFU and uterine fibroid embolization (UFE). Differential diagnosis has been discussed in Table 11.15.3.2. Hyperechoic unlike a myomatous polyp which appears hypoechoic Heterogeneous endometrium with little mass effect Enlarged uterus and a deformed contour are the most common computed tomography (CT) findings. Even though CT is the primary modality for the staging of cancers, it has limitation of reduced contrast resolution for the assessment of focal myometrial masses, and associated with risk of ionizing radiation. Also, in delineating the zonal anatomy for accurate localization of leiomyomas and LMSs, CT is inferior to magnetic resonance imaging (MRI). More often dense or amorphous calcification is seen. Almost exclusively, on plain radiography or CT, these patterns of calcification favours the diagnosis of leiomyoma. Ring-like calcification at the margins of a leiomyoma is a rarely observed pattern, which represents the thrombosed veins. Positron emission tomography (PET)/CT is an ideal modality for the confirmation of malignancy and also helps in detection of metastasis and recurrence. In a small study of patients with histopathologically confirmed uterine sarcoma found that on comparing with the findings of MRI and ultrasound with power Doppler imaging, the results of fluorodeoxyglucose (FDG) PET examinations were 100% positive. However, it has limitations such as increased false-positive rate and reduced widespread availability. MRI appearance of leiomyomas: MRI is considered as the most accurate imaging technique for the detection and localization of leiomyomas. In determining the presence and location of leiomyomas in infertile women before myomectomy, MRI has been shown to be more accurate and sensitive than US or hysterosalpingography. Nondegenerated uterine leiomyomas usually has a typical MRI appearance of (Table 11.15.3.4) well-circumscribed masses with homogeneously decreased T2-weighted signal intensity. TABLE 11.15.3.3 Note: SI, signal intensity; ADC, apparent diffusion coefficient. Compared with that of normal smooth muscle myometrium. Degenerated leiomyomas have varied imaging appearances on T1-weighted, T2-weighted and contrast-enhanced images as described in Tables 11.15.3.4 and 11.15.3.5. Hyaline Oedema *Common *50% cases *Usually peripheral Cystic Myxoid Carneous, red, or UAE associated Calcific Leiomyoma variants (Tables 11.15.3.4 and 11.15.3.6): There are several, such as mitotically active, cellular and atypical leiomyomas, as well as smooth muscle tumours of uncertain malignant potential (STUMP). On histopathological examination, the nondegenerated fibroids are made up of goals of uniform smooth muscles with varied amounts of collagen. Cellular leiomyomas, which are composed of compact smooth muscles cells only with no collagen, will demonstrate increased T2-weighted signal intensity and homogeneous enhancement. There is a greater risk of recurrence in case of atypical leiomyomas and uterine STUMP. The patients who had undergone myomectomy and were found to have atypical leiomyoma, must be under surveillance with an annual pelvic ultrasound or MRI. Since the recurrences may presents as pelvic or abdominal masses or as pulmonary metastases, for all the patients with STUMP lesions, routine physical examinations, including pelvic examinations and the baseline CT of the chest, abdomen and the pelvis are recommended for every 6 months for 5 years and then annually thereafter. In addition, prompt re-exploration and staging is recommended for patients with STUMP who have undergone a morcellation myomectomy. An aggressive imaging surveillance is considered, when the disease is present. Atypical leiomyomas and STUMPs routinely express progestin receptors; however, because of the low recurrence rates, there is no role for adjuvant hormonal therapy. Atypical uterine masses can be treated surgically with hysterectomy or myomectomy. Histologic subtype Cellular leiomyoma Round or oval Single large lesion with absence of coexistent adenomyosis Responds to GnRH analogues Cured with surgery No need for follow-up Lipoleiomyoma Well defined: round or oval STUMP Round or oval Intense surveillance Note: SI, signal intensity; ADC, apparent diffusion coefficient; UAE, uterine artery embolization; STUMP, smooth muscle tumours of uncertain malignant potential. Being a benign neoplasm, fibroids usually have a pushing border and rounded edges. But, specific types of fibroids represent growth pattern variations, such as intravenous leiomyomatosis, metastasizing leiomyoma, diffuse leiomyomatosis and peritoneal disseminated leiomyomatosis. Retroperitoneal growth and parasitic growth are the other atypical growth patterns. Even cervical and vaginal leiomyomas may show growth pattern. It is important to note that adenomyosis and focal myometrial contraction can coexist and distinction is important at preprocedure imaging (Table 11.15.3.7).
11.15: Uterus
Introduction
Clinical presentation
Causes and risk factors
Pathophysiology
Radiological features
Ultrasound
Colour doppler
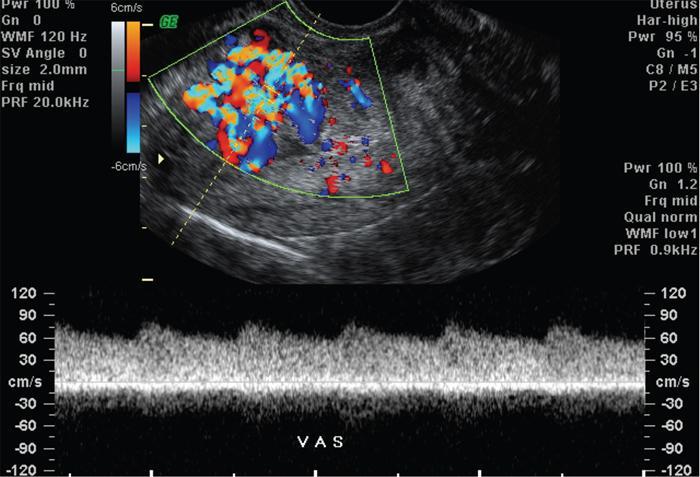
Spectral doppler analysis
Pelvic MRI
Angiography
Management
Treatment
Differential diagnosis
With elevated serum beta-HCG
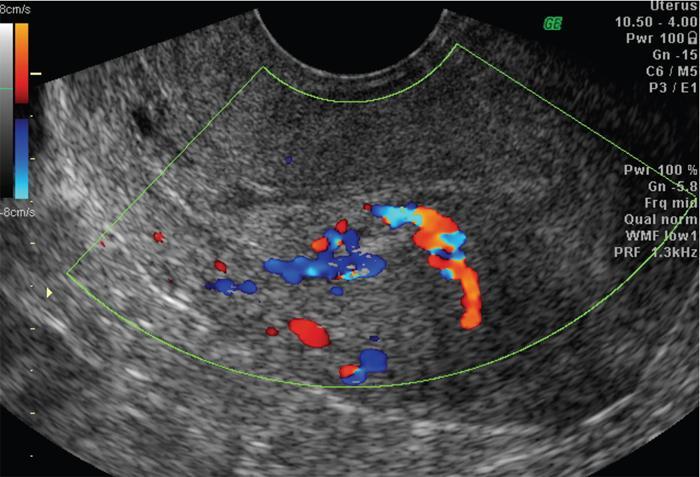
Introduction
Prevalence and incidence
Histopathologic features
Classification
Symptoms
Bleeding
Pressure on adjacent organs and pain
Reproductive dysfunction
Imaging
Differential Diagnosis on Ultrasound
Lesion
Features on Ultrasound
Submucosal
Endometrial polyp
Retained products of conception
Endometrial carcinoma
Endometritis/fluid collection
Subserosal and pedunculated myomas
Discussed under MRI
CT
Characteristics
Findings
Margins
Well defined
Shape
Round or oval
SI on T1-weighted images
Homogeneously isointense
SI on T2-weighted images
Homogeneously hypointense
Enhancement on gadolinium contrast-enhanced images
Yes (can be variable); enhancement indicates viability
SI on DW images (high b value)
Hypointense compared with normal smooth muscle (blackout phenomenon)
SI on ADC map
Hypointense compared with normal smooth muscle (blackout phenomenon)
Leiomyoma Degeneration Type
Overall Shape
SI on T1-Weighted Images
SI on T2-Weighted Images
Early Enhancement
Restriction on DW Images and ADC Maps
Round or oval
Hypointense or isointense
Hypointense
Minimal or non
No
May show speckled pattern
Hypointense
Hyperintense
Marked enhancement
—
Round or oval
Hypointense
Hyperintense
None
No
Round or oval
Hypointense
Markedly hyperintense
Progressive
No
Round or oval
Peripheral or diffuse hyperintensity
Peripheral or diffuse hyperintensity
None
No
Amorphous, central or peripheral
Hypointense
Hypointense
None
No
Leiomyoma Degeneration Type or Histologic Subtype
Overall Shape
SI on T1-Weighted Images
SI on T2-Weighted Images
Early Enhancement
Restriction on DW Images and ADC Maps
Follow-Up
Variable
Diffusely hyperintense
Marked
Yes
Heterogeneous (hyperintense with loss of SI on fat-saturated sequences) with amorphous bands of hypointensity
Heterogeneous, amorphous bands of hypointensity coursing through fat components
Marked enhancement of amorphous bands
Unknown
Heterogeneous with hyperintense areas
Heterogeneous with hyperintense areas
Variable
Unknown/yes at high b values
Unusual growth patterns
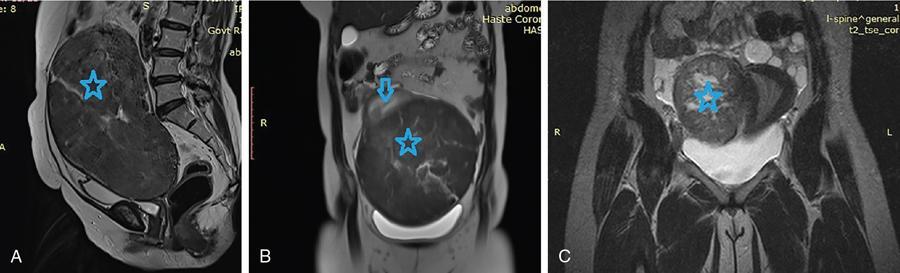
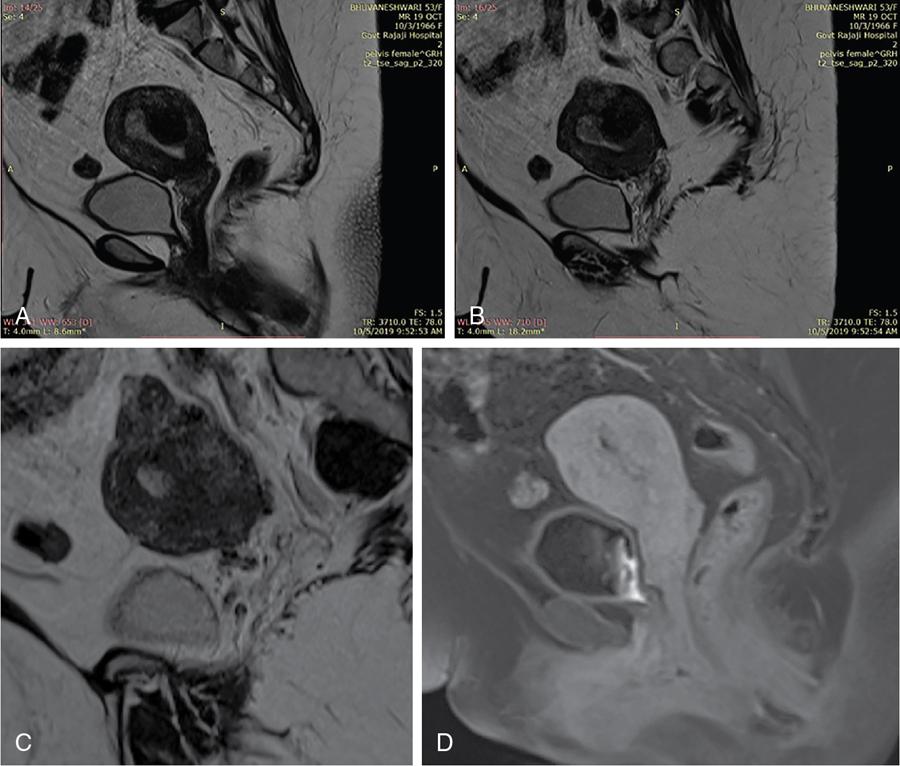
Differential diagnosis
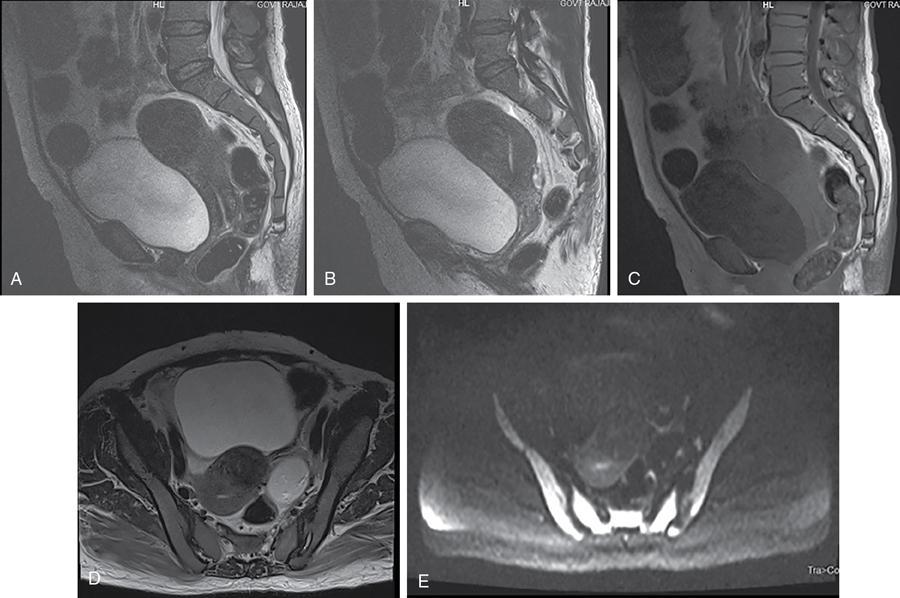
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








