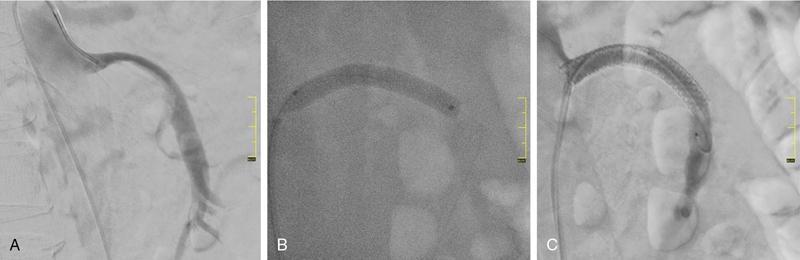
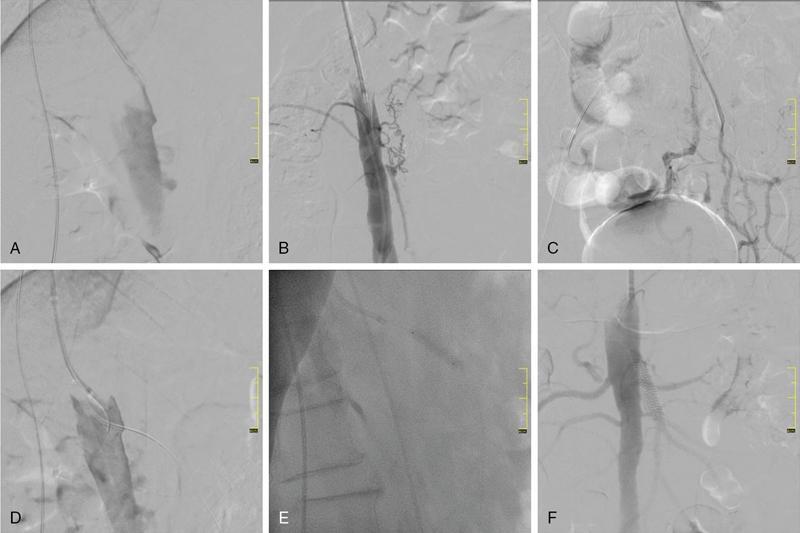
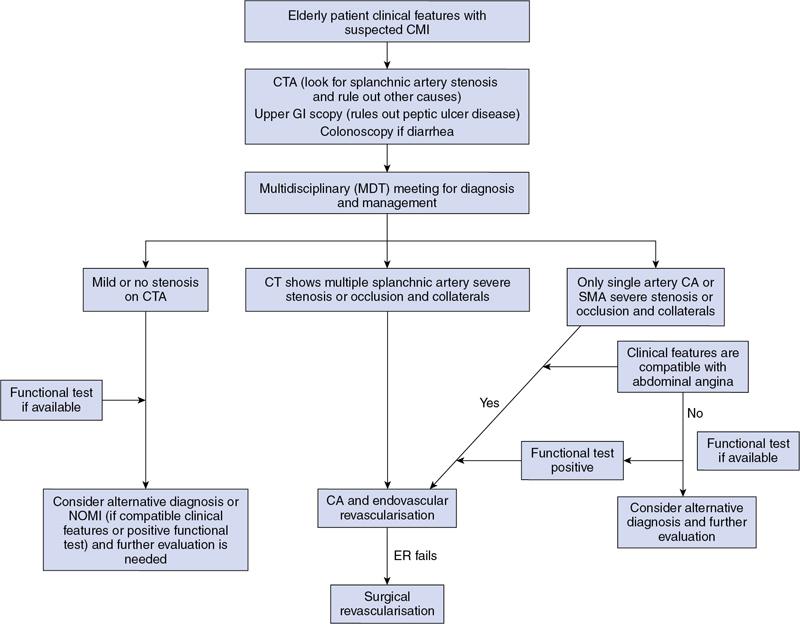
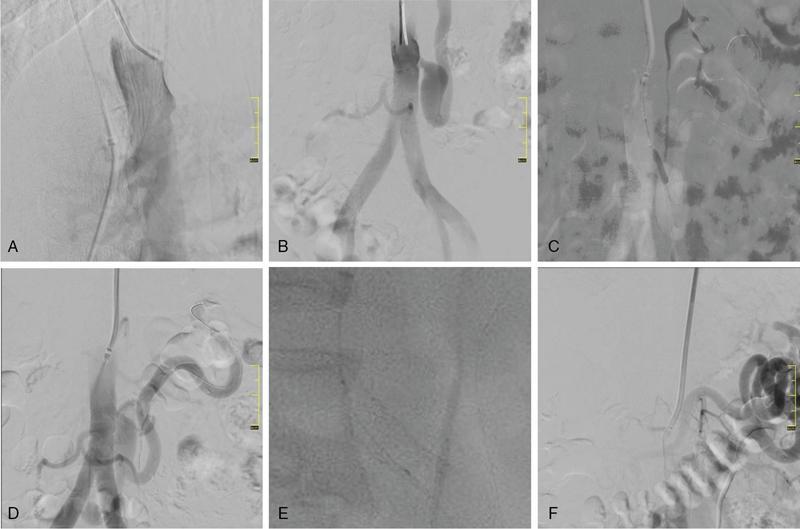
Munawwar Ahmed, Vikash Jain, Santhosh Babu K.B.,Shyamkumar N. Keshava Vascular interventions of gastrointestinal tract include recanalization and occlusive procedures. These are minimally invasive techniques, which play a role both in elective and emergency situations. Traditionally interventional radiological options have been considered as an alternative to the established surgical treatment options; however, currently many of these procedures have become the primary therapeutic option. In this chapter, we have discussed a few common conditions with case illustrations. Mesenteric ischaemia (MI) is an uncommon clinical condition caused by inadequate splanchnic blood flow to intestine (Table 7.17.1). MI can be acute or chronic as follows: SMA supplies largest part of the intestine including both small and large intestines and receives approximately 15%–20% of cardiac output in normal condition which increases to approximately 35% within 20 minutes of meal because of the increased oxygen demand. Small intestine can tolerate 75% reduction in blood supply for up to 12 hours (due to extensive mesenteric collaterals and auto-regulation) and bowel infarction or irreversible damage can develop in a minimum 6 hours of complete occlusion. Mucosa of the bowel is the most susceptible layer and affected first. Ischaemic bowel injury ranges from reversible mucosal injury to transmural infarction and necrosis. With intestinal necrosis there is bacterial translocation, robust inflammatory response resulting in cytokine storm which may cause profound hypotension, multiorgan failure, septic shock and even death. Reperfusion injury may occur following therapy secondary to formation of reactive oxygen species from interaction between oxygen rich blood and free radicals locally produced during inflammatory response. It may further lead to cell death through direct injury to cell membrane and proteins. Patients with CMI typically present with insidious onset of postprandial abdominal pain with associated significant weight loss. Pain typically starts 20–30 minutes after meal and lasts for 1–2 hours. Given the association of pain with food intake often leads to food aversion. Nonspecific symptoms are nausea, vomiting, diarrhoea and constipation. It is commonly seen in elderly (>60 years) smokers and unlike other atherosclerotic diseases, females are three times more likely to be affected. This is likely secondary to differences in the orientation of the mesenteric vessels to the aorta, with a more acute angle to the aorta in females when compared to males. The likely result is altered flow dynamics and increased susceptibility to atherosclerotic disease. Patients with mesenteric arterial stenosis may be asymptomatic due to presence of good collateral supply between mesenteric arteries (Fig. 7.17.1) and often require severe stenosis or occlusion of two or more proximal mesenteric arteries, more specifically SMA for clinical manifestation. Prevalence of mesenteric artery stenosis is not uncommon and progressively increases with increasing age, from 6% at 40 year and 14% at 60 year to 18%–67% at 75 year and above. Atherosclerosis is the commonest aetiology and most of the patients will have diffuse atherosclerosis in other territories (carotid, coronary and peripheral arteries) also. Other less common aetiologies are vasculitis, fibromuscular dysplasia, dissection, radiation therapy for cancer, postsurgical intimal hyperplasia and collaterals secondary to median arcuate ligament (MAL) compressing CA. The diagnosis is based on combination of clinical symptoms and radiological evaluation of the mesenteric vasculature. The value of symptoms alone is limited for the prediction of the diagnosis of CMI. Moreover, identification of occlusive disease in the mesenteric arteries is not suggestive of mesenteric ischaemia without associated clinical symptoms. Classical triad of symptoms of CMI include postprandial abdominal pain, weight loss and abdominal bruit on clinical examination; however, it is seen in only 22% of cases and has low predictive value (60% probability). The presenting symptoms are abdominal pain (94%), weight loss (80%), food aversion (52%), nausea, vomiting (26%) and diarrhoea (24%). Intermittent abdominal pain can progress to more constant and prolonged pain. If left untreated, CMI has the potential to develop AMI. Clinical examination may reveal abdominal bruit in 17%–87% of cases and cachexia. Laboratory findings are nonspecific and may include anaemia, hypoalbuminemia. Objective of the imaging is to confirm the diagnosis, and exclude the other causes of abdominal pain which can mimic CMI such as chronic pancreatitis, gastro-duodenal ulcer, gastric or pancreatic malignancy, inflammatory bowel disease, irritable bowel syndrome, MAL compressing CA. Imaging helps in treatment planning and assessment of the arterial lesion in terms of length of stenosis/occlusion, number of arteries involved, collateral flow and possible aetiology (atherosclerosis, arteritis, dissection, etc.) (Fig. 7.17.2). Duplex ultrasonography (DUS) is often the initial imaging modality and used to assess proximal stenosis or occlusion of the CA and SMA. DUS provides physiologic flow data and can be performed in the fasting and postprandial states to detect haemodynamically significant stenosis. In the fasting state, the SMA possesses a high-resistance waveform characteristic of the splanchnic circulation while the CA demonstrates low resistance wave form characteristic of flow throughout the cardiac cycle within hepatic and splenic parenchyma. However, in the postprandial state, the SMA will show a marked increase in EDV with a less dramatic increase seen in the coeliac circulation. A significant stenosis (>70%) will be characterized by an increase in the PSV and EDV during duplex US interrogation. Retrograde flow in the hepatic artery may be seen with a severe stenosis or occlusion of the coeliac artery while loss of diastolic flow or flow reversal may be seen with a significant stenosis of the SMA. Increased PSV of 275 cm/sec or more and EDV of 45 cm/sec or more is suggestive of critical stenosis (>70%). However, DUS has the limitations of operator dependency and poor window due to bowel gas and patient body habitus. Computed tomography angiography (CTA) with neutral oral contrast or water in both arterial and portal venous phase has become the imaging modality of choice as it efficiently demonstrates diseased mesenteric arteries including IMA with their distal branches, calcifications, collaterals, secondary changes in bowel wall and rules out other causes of abdominal pain. Multiplanar reformation (MPR), maximum intensity projection (MIP) and volume rendering (VR) have been used to optimally delineate significant lesions and aid greatly in preprocedure planning like presence of stump. In CMI, arterial findings are more prominent than bowel findings and bowel changes are rarely seen (Table 7.17.2). Sensitivity and specificity of CT angiography in the diagnosis of CMI is 95% and 100%, respectively. MRA is reserved for patient with poor renal function and severe allergy to iodine contrast. Catheter angiogram is a gold standard for assessing the severity and length of stenosis, size, angle at origin of the involved artery and for dynamic assessment of collaterals. Catheter angiogram is almost always combined with endovascular revascularization (ER). In the presence of single artery disease, 70% or more stenosis is considered significant and in presence of multiple arterial diseases, 50% or more stenosis is considered significant. Preprocedural workup includes complete blood count, coagulation parameters, renal function test and upper GI endoscopy and lower GI endoscopy in patients with lower GI symptoms such as diarrhoea and altered bowel habits. Functional tests include tonometry and visible light spectroscopy (VLS) which can be performed in selective cases if available such as single artery involvement to support the diagnosis of MI. In tonometry, partial pressure of CO2 carbon dioxide (PCO2) is measured in the antrum and duodenum between two balloons in the stomach and duodenum, placed through nasogastric and nasojejunal tubes. PCO2 reflects local arterial PCO2 and indicates mucosal ischaemia. Sensitivity and specificity of 24-hour tonometry is 76%–92% and 72%–94%, respectively. VLS is performed by using endoscopy and measures mucosal oxygen saturation in antrum and duodenum. It has sensitivity and specificity of 90% and 60%, respectively. Diagnosis and treatment decision should ideally be made in a multidisciplinary meeting involving gastro-enterologist, interventional radiologist and vascular surgeon. Please refer to management algorithm for CMI (Flowchart 1, Fig. 7.17.3). Frequent small low-fat meals are advised to reduce metabolic demand of mesenteric vasculature. Aggressive medical atherosclerotic risk factor reduction plays a pivot role. Smoking cessation and appropriate management of comorbidities such as diabetes mellitus, hypertension and hyperlipidaemia are equally important. The primary goal of revascularization is to relieve symptoms, restore normal weight and thereby improve quality of life. The challenge is to select the patient with mesenteric stenosis who will benefit from revascularization. Revascularization is accepted in case of symptomatic multivessel stenosis. In case of single-vessel stenosis, those with typical symptoms and by excluding other causes of pain should be offered treatment. Most patients with asymptomatic stenosis, however, do not require intervention. However, revascularization may be considered in asymptomatic patients with multiple severe mesenteric arterial stenoses before major abdominal surgery where disruption of collateral supply is contemplated. ER and open surgical revascularization (SR) are the two treatment options and former is often the first choice. ER includes balloon angioplasty and primary stenting. In case of multiple artery stenosis or occlusion, often revascularization of single artery is sufficient to relieve symptoms and prevent bowel ischeamia. SMA is the most preferred artery for revascularization as it has higher postprandial blood flow and supply larger area of GI tract compared to CA. CA is the second preferred artery for revascularization. Revascularization should be considered in presence of typical symptoms of CMI and imaging findings of multiple arterial (two or more) severe stenosis or occlusion after excluding other causes of pain. In presence of typical symptoms and severe stenosis of single artery (CA or SMA), revascularization can be considered after carefully ruling out the other aetiology. Functional testing (VLS or tonometry) if available can be a helpful guide. Revascularization in asymptomatic patient is debatable and may be considered for prevention of development of AMI, in patient with severe stenosis or occlusion of CA and SMA and positive functional test or nonavailability of functional test and in patients with multiple severe arterial stenoses before major abdominal surgery where disruption of collateral supply is contemplated. Revascularization may be considered in atypical abdominal pain with severe stenosis or occlusion of CA and SMA after ruling out other causes of abdominal pain. Revascularization is not considered in nonsevere stenosis of CA and SMA, single artery stenosis without typical symptoms or other causes of symptoms are not ruled out in multidisciplinary meeting. ER is relatively contraindicated in heavily calcified arteries, long segment stenosis or occlusion of mesenteric arteries and also contraindicated in MAL compression of CA. MAL compressing the CA is better managed with surgical decompression since it also alleviates compression of the coeliac ganglion and the neural plexus. Endovascular management will frequently result in stent fracture by extrinsic forces associated with diaphragmatic compression. Endovascular management has taken on an increasing role in the treatment of chronic mesenteric ischaemia caused by atherosclerotic occlusive lesions. In last 20 years there has been several folds increase in endovascular intervention for CMI that has largely eclipsed the number of surgical procedures. Endovascular approaches, when feasible, are associated with lower morbidity, in-hospital mortality, costs and shorter length of stay. Also, patients treated initially with an endovascular approach who require repeat intervention for in-stent stenosis seem to have better outcomes with lower morbidity and mortality. Guidelines from various societies now recommend ER as the preferred initial treatment approach due to high initial technical and clinical success rate, better perioperative outcomes and comparable long-term survival compared with SR. The patient may be admitted day before the procedure or on the same day. Patient is given dual antiplatelet agents like aspirin 75 mg and clopidogrel 75 mg. Patient should be fasting for 6 hours before the procedure. Groin area should be shaved for femoral arterial access. Patient should be shifted to angiosuite with patent IV line connected to IV fluid (normal saline or 5% dextrose). Preprocedural sedatives include injections Morphine 5 mg IM and Phenergan 25 mg IM half to one hour before the procedure. Low profile system (compatible with 0.014 inch and 0.018 inch guidewire) and balloon expandable stent are preferred over high-profile system and self-expandable stent.Some operators prefer 0.035-inch platform, especially for long segment and heavily calcified coeliac and SMA lesions. Access can be through femoral artery or brachial or radial (radial artery is preferred over brachial) artery. Though brachial or radial artery access provides better angle for cannulation and navigation of SMA and CA, authors prefer femoral artery because of our experience and less complications at access site. After placing the introducer sheath in the femoral artery, 5000 IU of heparin bolus is injected. For brachial/radial access 6 or 7F 90 cm Destination sheath (Terumo Medical Corp., Somerset, NJ) may be used. For femoral access, 6 or 7F 45/65 cm femoral Ansel guiding sheath (Cook medical, Bloomington, IN) or Morph deflectable guiding sheath (Biocardia, San Carlos, California) is preferred. Morph deflectable guiding sheath provides considerable support to catheters buckling into the supracoeliac aorta. Diagnostic angiography of the aorta and mesenteric vessels is done initially in AP and lateral projection. AP projection allows for assessment of overall mesenteric flow dynamics and lateral projection is used to evaluate ostial stenosis. A Cobra 2 or Simmons 1 catheter can be used to cannulate mesenteric artery. The lesion may be crossed with a hydrophilic guidewire (V18; Boston Scientific, Natick, Massachusetts) or a 0.014-inch guidewire, with matching support catheter. Predilatation can be done with a 4- or 5-mm balloon. The guidewire can then be exchanged for a stiff exchange length guidewire. For ostial and calcified lesions, balloon expandable stents are preferred for ease of precise placement and high radial force. Self-expanding stents are preferred for trunk lesions and intervention-related dissection. In addition, proximal end of the stent in aorta can be flared to maximize flow within the stent. Retrograde recanalization of SMA and coeliac arteries can also be performed through gastro-duodenal and pancreatico-duodenal arcade. Postprocedure patient is put on dual antiplatelet (clopidogrel 75 mg and aspirin 75 mg) for 3 months and then single antiplatelet (aspirin 75 mg daily) is continued lifelong. DUS is performed at 48 hours to see patency of stent and thereafter patient is followed up clinically at 3 month, 6 month and at 1 year and yearly after 1 year. DUS is a good imaging modality for checking the patency of the stent and CECT is reserved for cases with difficult and equivocal DUS findings and suspected cases of recurrence (reappearance of symptoms). Procedure-related complications depend on many factors such as operator’s experience, individual case, device selection and access site selection. Access site complications are the most common and include haematoma, pseudoaneurysm, dissection and thrombosis. It is more with brachial artery access than femoral artery access. Complications are more with use of large profile system (0.035 inch) than low profile system (0.014 inch or 0.018 inch). Incidence of mesenteric artery complications of ER was 7% with 2.5% mortality in a retrospective study involving 156 patients. Complications were higher in IMA compared to SMA (20% vs 10%) and lowest in CA (2%). Other complications include dissection of the diseased artery, acute mesenteric ischaemia due to stent thrombosis and distal embolization, stent migration and perforation of the branch artery with mesenteric haematoma. Cause of mortality was bowel ischaemia resulting in multiorgan system failure due to distal embolization, stent thrombosis and dissection of mesenteric artery. Distal protection device can be used to prevent distal embolization especially in stenosis with high risk features (long stenosis >30 mm, severe calcifications and occlusion). In-stent stenosis can be seen in 28%–36% of endovascular treated patients within 2 years. Female gender, diabetes mellitus and small mesenteric arteries (diameter <6 mm) are among those associated with high risk of restenosis. Repeat intervention is often successful. Technical success rate of endovascular treatment is defined as successful angioplasty and stenting with no residual stenosis or residual stenosis <30% and is high (95%–96%) in various studies. Presence of angiographic stump allows for engagement of the occluded artery for traversing the lesion and is an important predictor of technical success. Lower technical success rate and/or increased procedural complications are seen in long-segment occlusion, small-diameter vessels, absence of angiographic stump and heavily calcified stenosis. Clinical success rate is defined as relief of symptoms and varies from 90%–100% in cases with stenosis or occlusion of two or three arteries and 73%–76% in cases with stenosis or occlusion of only one artery either CA or SMA. Compared to open surgery, endovascular treatment is associated with significantly lower perioperative complications and mortality, comparable long-term survival rate and higher recurrence rate. Perioperative mortality in open surgery was 8% compared to 2% in ER (RR 1.57; 95% CI 0.84–2.93) in a recent meta-analysis. This is because open surgery carries a higher risk in very elderly and patients with preexisting comorbidities. Long-term patency of revascularization and survival during follow-up are longer after open SR compared with ER. One year and five-year primary patency of open SR and ER were 91%–94% versus 69%–74% and 80%–81% versus 51%–52%. Five-year secondary patency was also longer in open SR (96%–98%) compared to ER (79%–88%). Despite better long-term results in open SR, minimally invasive ER is still the first choice of revascularization because of lower procedural complications and mortality, lower cost and shorter hospital stay. Open surgery is reserved for younger patient with good surgical risk and failed or difficult cases (long-segment stenosis, heavy calcification) of ER. CMI is an uncommon type of MI and should be considered in an elderly patient presenting with insidious onset of abdominal pain associated with significant weight loss or change in eating habit or food fear. CTA is the imaging modality of choice. The patient should also be evaluated with endoscopy to rule out other aetiology. ER has become the first choice of treatment for mesenteric artery revascularization because of lower morbidity and mortality. SR should be considered in failed cases or technically difficult cases of ER in patients otherwise fit for surgery. Long-term survival is comparable in both surgical and ER. Acute mesenteric ischaemia (AMI) is a life-threatening condition that occurs due to sudden reduction in blood flow to the intestines thereby unable to maintain its functional demand, subsequently leading to bowel infarction. Classically it is divided into four groups by eatiology: arterial embolism, arterial thrombosis, NOMI and venous thrombosis. The mortality rate is 50%–69% and increases further up to 80%–90% with delay in diagnosis and long-segment bowel infarction. Establishing early diagnosis is crucial as it provides opportunity of aggressive treatment before bowel infarction. Absence of specific symptoms and laboratory data makes early detection difficult. Classical clinical presentation is sudden onset of severe abdominal pain that is out of proportion to physical examination. Physical examination may show tachycardia, hypotension. Laboratory evaluation shows neutrophilic leukocytosis, haemoconcentration, raised C reactive protein, elevated D-dimer, metabolic acidosis and raised arterial lactate. None of them can be used to rule in the possibility of AMI, moreover arterial lactate is often negative in early phase of AMI. Considering nonspecific symptoms and laboratory results, imaging plays an important role in the diagnosis of AMI. Multidetector computed tomographic angiography (CTA) is the most important imaging test for early diagnosis of AMI and also allows for exclusion of other causes of acute abdomen It can demonstrate critical stenosis or occlusion of mesenteric arteries, its underlying causes and its severity while also providing information concerning bowel viability. It has been shown that early diagnosis with contrast-enhanced computed tomography and revascularization can reduce the overall mortality in AMI by up to 50%. Treatment has conventionally been surgical restoration of mesenteric blood flow and resection of necrotic bowel. In stable patients without advanced peritoneal signs endovascular therapy (EVT) has increasingly played important role. It has shown to significantly reduce the segment of resected bowel and improvement in mortality rates. Arterial embolism is responsible for 40%–50% of cases of AMI. It is frequently caused by emboli of cardiac origin; frequent causes are atrial fibrillation, prior myocardial infarction, valvular heart disease and embolic from aortic atheromatous plaques. One-third of these patients also have history of prior embolic event. Superior mesenteric artery is particularly vulnerable visceral artery to emboli owing to its large diameter and acute branching angle from aorta. Patients generally present with sudden onset of severe abdominal pain which may be associated with haematochezia. CT scan shows filling defect typically located 6–8 cm beyond the origin of SMA around the origin of middle colic artery with poor distal flow. Associated embolism may involve multiple organs like brain, spleen, kidneys or extremities. Arterial thrombosis accounts for 25% of cases of AMI and is the most common cause in patients more than 70 years. It is caused by an underlying atherosclerotic lesion and is associated with history of abdominal angina and weight loss in many of the patients. Most patients have atherosclerotic risk factors like dyslipidemia, diabetes, hypertension or smoking. Atherosclerotic disease at other sites like cardiac, cerebrovascular, renal or peripheral vascular is also noted. It has a relative insidious onset as compared to embolic occlusion because of the development of additional collateral circulation, which suggests an acute on chronic process. Imaging reveals stenoocclusive lesion 1–2 cm beyond the origin of SMA. Findings of bowel ischaemia may be seen with complete arterial occlusion (Table 7.17.3).
7.17: Vascular interventions in gastrointestinal tract
Introduction
Mesenteric ischaemia
Chronic mesenteric ischaemia and angina
Diagnosis
Imaging
Other diagnostic workup
Management
Medical management
Revascularization
Indication of revascularization
Contraindication of revascularization
Endovascular treatment
Patient preparation
Technique of ER
Postprocedure care and follow-up
Complications of ER
Outcome of ER
Summary
Acute mesenteric ischaemia
Overview
Acute arterial occlusion
Radiology Key
Fastest Radiology Insight Engine