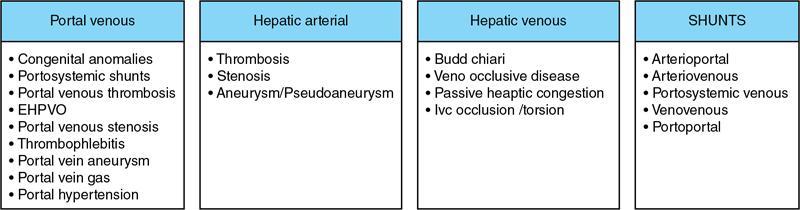
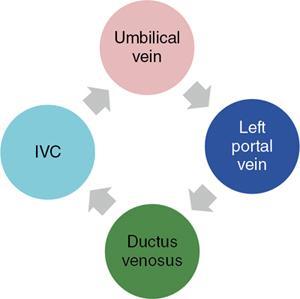
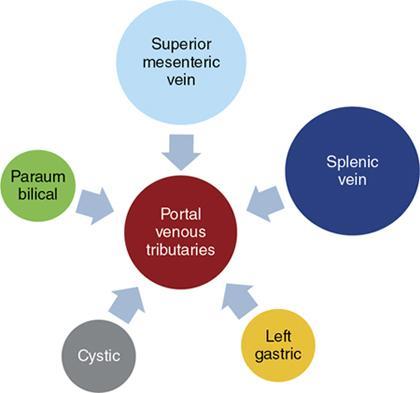
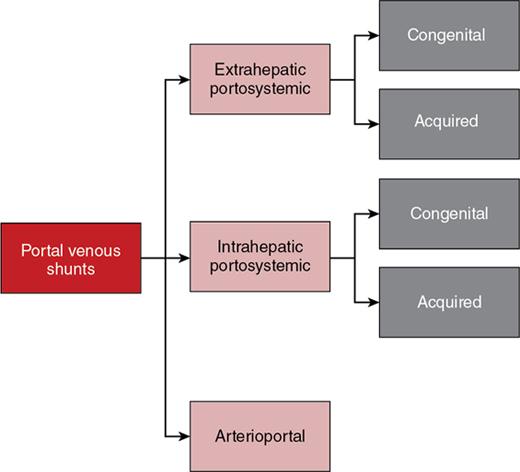
Ritu K. Kashikar, Shrinivas B. Desai, Payal Agrawal A spectrum of vascular variants can involve the liver. Both congenital and acquired anomalies can be seen. The abnormalities can involve the portal venous system, hepatic artery, hepatic veins or maybe the result of shunting between any of these vessels. Though not commonly encountered, familiarity with the pathogenesis and imaging features of these vascular entities can guide appropriate patient management. This chapter aims at classification of vascular pathologies of the liver (Table 9.12.1) with description of important entities. The umbilical vein accounts for approximately 80% of the afferent flow into the liver in utero. The umbilical vein joins the left portal vein, which in turn drains into the ductus venosus and carries blood to the inferior vena cava. The umbilical vein and ductus venosus start involuting at birth and typically close in the first week of life in term neonates (Fig. 9.12.1). Detailed embryology of the portal venous system has been discussed in previous chapters. The portal vein accounts for two-thirds of the hepatic blood supply. The distinctive feature of the portal vein is that it begins and ends in capillaries. The blood from the entire gastrointenstinal tract (except lower rectum), gallbladder, pancreas and spleen is carried via the portal vein to the liver. The hepatic arterial branches divide along the distribution of portal venous radicals and supply oxygenated blood to the sinusoids (Fig. 9.12.2). These include The dorsal and ventral limbs of the vitelline vein form the normal portal vein. Atrophy of the ventral limb occurs during embryogenesis. This maybe an incidental finding, though association with duodenal obstruction is seen in 50% cases. The aetiology of duodenal obstruction is often due to secondary causes such as duodenal stenosis, atresia, Ladd bands due to malrotation or annular pancreas. Associations include heterotaxy (polysplenia) and biliary atresia. Aberrant vessel may give an extrinsic impression on the high second portion of the duodenum on upper GI series. USG may show aberrant vessel in patients being scanned for other anomalies. The aberrant vessel is usually incidentally discovered intraoperatively during surgery for duodenal obstruction or malrotation. Hypoplasia or atresia of the portal vein may involve all or a portion of the portal vein. Atresia of a major branch is associated with absence of the corresponding hepatic lobe. Main portal vein hypoplasia or stenosis results in prehepatic portal hypertension and gastrointestinal hypertension (Flowchart 9.12.1). A portal vein smaller in calibre or as small as the adjacent hepatic artery is termed hypoplastic. The venous diameter in cases of hypoplasia is of 3 mm. Twenty-six per cent cases with biliary atresia have hypoplastic portal vein. TAPVR is a congenital cardiac malformation in which pulmonary veins instead of draining into left atrium during cardiac development, drain into the right atrium via an anomalous vein, resulting in right to left shunts. Types: In type 3 TAPVR an anomalous infradiaphragmatic vertical vein is formed by the confluence of pulmonary veins, typically through the oesophageal hiatus. Venous obstruction at the level of the diaphragm and accounts for the clinical presentation of pulmonary venous congestion and oedema. Early presentation (within 24–36 hours of life) included respiratory distress with cyanosis, tachypnea, and tachycardia. Features on X-ray include normal–sized heart with pulmonary interstitial oedema. One-third of patients have an additional complex cardiac anomaly and heterotaxy with right atrial isomerism. May show abnormal draining vein draining into the IVC or pulmonary vein. CT/MR venography allows accurate depiction of the anomaly. The pulmonary venous confluence drains to systemic veins below the diaphragm. The confluence is usually posterior to the left atrium and vertically oriented. From here, a descending vein passes through the oesophageal hiatus and most frequently drains into the connection in to ductus venosus, hepatic veins or inferior vena cava is not as frequent. Portosystemic shunts are classified into intrahepatic and extrahepatic. In the intrahepatic shunts, the shunt is at the level of PV branches after its division whereas in the extrahepatic shunts the anastomoses are between the tributaries of the portal or mesenteric system or main PV and a systemic vein (Flowchart 9.12.2). Abernethy malformation, also known as congenital extrahepatic portosystemic shunts (CEPS) is a rare clinical entity, in which portal blood is shunted partially or completely into the systemic circulation via an abnormal communication of the portal system with the systemic circulation. It results from persistence of embryonic vessels. The entity was named by John Abernethy in 1793. The embryological development of the portal vein occurs between the 4th and 10th weeks. The umbilical veins of chorionic origin, vitelline veins and the cardinal veins from the body of the embryo are the three paired venous systems seen in the embryo in the 4th week. Communications develop between the vitelline veins at 4 weeks, which anastomose to form a figure-of-eight configuration around the developing duodenum. Selective involution of these veins leads to the final configuration of the portal vein. Because the vena cava also has a complex development and is derived from several venous channels including the sinus venosus and a portion of the anastomosis between the right and left vitelline veins in the cranial part of the liver, it has been suggested that this may be the embryological basis of development of congenital extrahepatic portosystemic shunts. Various systems have been proposed for classification of Abernathy Syndrome: In this classification portal flow is classified as Patients with type A drainage show associated cardiac anomalies, while gastrointestinal bleeding is common in type C. In this system CEPS was classified into three types depending on the degree of severity of intrahepatic portal system hypoplasia in to mild, moderate and severe types. This system has therapeutic implications in providing information regarding acceptance of portal blood flow by the portal vasculature after shunt occlusion. CEPS may be asymptomatic well into adulthood and many authors have reported seeing adult patients presenting for the first time with clinical manifestations. Clinical features of portosystemic shunts may broadly be divided into: These include hepatopulmonary syndrome, metabolic dysfunction and hepatic encephalopathy. Patients with type 1 shunt have a female preponderance and often have concomitant congenital anomalies. These anomalies are less common in patients in type 2 shunts. Other anomalies have also been reported in patients with Abernethy malformation which include chromosomal anomalies such as Downs syndrome and structural anomalies of the heart, gastrointestinal, genitourinary, skeletal and vascular systems. Approximately half the patients of CEPS have nodular lesions of some sort in the liver (Flowchart 9.12.3). Nodular hepatic lesions in patients with congenital portosystemic shunts may be single or multiple. In order to establish a diagnosis of CEPS a communication between the portal and the systemic venous system, either before or after the formation of the PV by union of the SMV and SV has to demonstrated. Secondary causes of nonvisualization of the PV such as portal cavernoma or PV thrombus should be excluded. Patients with congenital portosystemic shunts do not have features of portal hypertension, such as splenomegaly, varices and collaterals. USG may demonstrate the shunt and show haemodynamics involved such as the magnitude and direction of flow. It may pick up congenital shunts preoperatively; however, it may not detect associated anomalies particularly of lung and heart. Smaller shunts, particularly type 1a may not be well picked up. Ultrasound may not fully characterize liver lesions seen in these patients. It is the modality of choice and can accurately diagnose the shunt and type and aids in management. Shunt is visualized as an abnormal communication between the portal venous and systemic circulation and is best depicted in the portal venous phase. CT also shows presence/absence of intrahepatic branches of portal vein. Evaluation of associated anomalies particularly in patients with congenital heart disease who require evaluation of pulmonary vasculature, or patients with suspected hepatopulmonary syndrome who require evaluation of the lungs can also be done with CT (Fig. 9.12.4 and Table 9.12.3). Another major advantage of CT is that it helps to detect and characterize hepatic lesions in these patients. It displays the arterial and venous anatomy, and provides an angiographic road map for surgical resection. Regenerative nodules are usually homogeneous with enhancement during arterial phase, on both CT, however without washout (Fig. 9.12.5). Rest of lesions show classic enhancement pattern as described in previous chapters. The imaging findings in patients with Abernethy malformation with hepatocellular carcinoma do not appear to be typical, that is hypervascularity on the arterial phase images with washout on delayed phase. Thus, patients who do not have typical findings of a benign lesion, that is lack of arterial enhancement, should be closely followed up or biopsied. Disadvantages of an MRI include longer periods of sedation which is a disadvantage in patients with CEPS who may be very young, very hypoxemic or encephalopathic. MRI also has lower spatial resolution than CT and may not show small intrahepatic portal venous radicles in type 2 patients. MRI can otherwise show all abnormalities seen on CT. Advantages of MRI using hepatobiliary contrast agents are in the characterization of hepatic nodules and for follow-up. MRI does not expose the patients to ionizing radiation. Patients with no symptoms, those with mild metabolic abnormalities and with liver nodules should be followed up with ultrasound and biochemistry. Patients with portosystemic encephalopathy, liver dysfunction or those with shunt ratio >60% should be treated according to type of shunt. Patients with type 1 shunts should be transplanted. Patients with type 2 shunts should be treated with shunt closure – either via embolization or surgical. Liver transplantation is considered when medical and surgical methods fail especially in patients with complications. Intrahepatic portosystemic shunts are rare. They may be congenital or result from trauma or portal hypertension. They develop due to persistent communications between vitelline and umbilical systems. These are communications between the branches of the PV and inferior vena cava (IVC). Park et al. classified these 1990 in four types. Type 5 was added later, these are classified in Table 9.12.4. Type 2 shunt with or without a focal varix is the most common type reported. Another classification system is proposed by Kanasawa et al. based on correlation with severity of portal hypoplasia (mild, moderate and severe) with portal venous pressure, histopathological findings, postoperative portal venous flow and hepatic regeneration. Associated anomalies such as cardiovascular, hepatobiliary, urogenital and gastrointenstinal can be seen. Complications such as portopulmonary hypertension are seen in 13%–66% children. As a consequence of long-term shunting, hepatic encephalopathy, and hepatopulmonary syndrome are the most common symptoms. Tumours such as FNH and regenerating nodular hyperplasia can be seen. These shunts may close spontaneously within the first 2 years of life or may remain asymptomatic and undetected for several years. When chronic shunting persists into adulthood, patients most often present with encephalopathy. The feeding (afferent) and draining (efferent) vessels of the shunt appear as enlarged, tubular, anechoic structures that are contiguous with the portal and hepatic veins. Antegrade flow is seen on colour Doppler images. Focal varix if present appears as an abnormal, rounded cystic structure with turbulent flow. Doppler study can also calculate the shunt ratio (total blood flow volume in the shunt divided by the blood flow in the portal vein). Shunt ratios greater than 60% should be corrected to prevent complications. Loss of normal undulating waveform of afferent portal vein branch with increased flow velocity and phasic waveforms owing to transmitted cardiac pulsations can be seen. The efferent hepatic vein branch of the shunt can show continuous flow with flattening of the Doppler waveform due to increased portal venous inflow. Communication between intrahepatic portal venous and peripheral hepatic venous radicals can be demonstrated easily on both contrast-enhanced CT and MRI. Similar to extrahepatic shunts CT is preferred over MRI in documentation of shunts. The afferent portal vein branch and the efferent hepatic vein branch are enlarged. Venous varices can be seen. The draining hepatic vein branch opacifies earlier than other hepatic veins (Fig. 9.12.6 and Table 9.12.5). The liver may show fatty degeneration and atrophy, but when the anomaly is corrected, fatty replacement disappears and liver size increases. Conservative medical therapy including restriction of protein and ingestion of lactulose. Symptomatic intrahepatic portosystemic shunts can be managed conservatively or with transcatheter embolization, surgical ligation or partial hepatectomy. The connection between the left umbilical vein and right hepatocardiac vein (future IVC) in the foetal circulation is called ductus venosus. This vessel is responsible for carrying nutrient-rich blood from placenta to the right atrium directly by bypassing the sinusoidal plexus of the liver. The umbilical vein and ductus venosus close at birth and form the ligamentum teres and ligamentum venous, respectively. The time interval following birth for closure of ductus venosus is variable ranging from few minutes after birth to 18 days in term neonates and as late as 37 days in premature infants. Patent ductus venosus is an intrahepatic portocaval shunt causing partial or complete diversion of portal blood to the systemic circulation and may present with hyperammonemia. Patent ductus venosus is seen on Doppler sonography as a vascular tubular structure in the left lobe of the liver, continuing from the umbilical vein and connecting the portal vein to the inferior vena cava. The foetal ductus venosus show waveforms similar to IVC corresponding to the cardiac cycle with a systolic and diastolic component. This diphasic waveform is seen in preterm and term infants and becomes monophasic as ductus closes. CT and MRI also accurately detect the shunt and patency. Associated hepatic lesions seen in patients with portosystemic shunts can be diagnosed and characterized better. Treatment (closure) is recommended in cases with complications or to prevent complications if the shunts persisted beyond 2 years of age. Shunt closure can be performed surgically or endoscopically. The complex nature of the shunt can pose problems during surgical closure. Transvenous and balloon occlusion have been done successfully. Occlusion of portal vein can occur due to a variety of conditions. The aetiologies of portal venous thrombosis in the neonatal age group include umbilical vein catheterization, omphalitis, dehydration or neonatal sepsis. Older children develop occlusion secondary to intraabdominal infections and portal hypertension. Other aetiological factors include prothrombotic states such as hereditary deficiency of protein C or protein S and factor V Leiden deficiency, vascular injury, trauma, stasis and congenital anomalies such as webs. Acute portal vein thrombosis can be asymptomatic, or the patient may present with abdominal pain, ascites or fever. Chronic portal vein thrombosis presents as ascites, encephalopathy, varices and upper gastrointestinal bleeding. Acute thrombus appears hypoechoic filling defect on USG with absent flow on Doppler. There is distension of the thrombosed vein (Fig. 9.12.7). CT and MRI with contrast will detect filling defect in the vein with distension. T2W1 images may show absence of flow void. Acute thrombus may appear hyperintense on T1W1 images. Tumour thrombus reveals signal similar to tumour on all sequences with diffusion restriction (Fig. 9.12.8). Chronic thrombus may present as eccentric filling defect, attenuation of vein or less commonly calcification of vessel wall. Collaterals are often seen in chronic portal vein thrombosis. Treatment usually involves combination of anticoagulation and intervention depending on age of thrombus. An acute portal venous thrombus may undergo partial or complete spontaneous resolution. The commonest cause of paediatric portal hypertension in the developing world is extrahepatic portal vein obstruction (EHPVO). It is also the second most common cause of portal hypertension in the western world. EHPVO is a condition characterized by obstruction of the extrahepatic portal vein (as the name suggests) with or without associated involvement of the intrahepatic branches, splenic vein (SV) or superior mesenteric vein (SMV). The hallmark of this chronic longstanding condition is carvernomatous transformation of the portal vein. Acute and chronic portal vein thromboses occurring in the setting of liver cirrhosis or HCC are not included in this disorder. EHPVO is an important cause of noncirrhotic portal hypertension with preserved liver structure and function till late in course of the disease. Proposed aetiologies include infection or prothrombotic event occurring early in life (in genetically predisposed individuals), leading to portal venous occlusion (Table 9.12.6).
9.12: Vascular pathologies of liver
Introduction
Portal venous pathologies
Embryology
Postneonatal
Congenital anomalies of portal vein
1. Preduodenal portal vein
Imaging
2. Portal vein hypoplasia or atresia

3. Infradiaphragmatic total anomalous pulmonary venous return (TAPVR)
Clinical presentation
Imaging
X-ray.
USG.
CT/MRI.
Portal venous shunts
Congenital extrahepatic portosystemic shunt (abernathy malformation)
Embryological basis of congenital extrahepatic portosystemic shunts
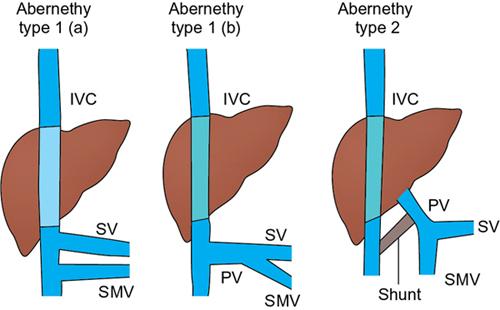
Clinical presentation
1. Symptoms due to shunt
2. Symptoms due to congenital anomalies
3. Symptoms due to hepatic lesions
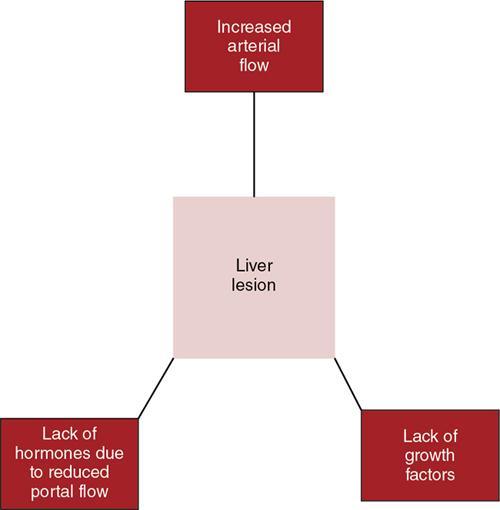
Types of liver lesions
Imaging features
USG
CT
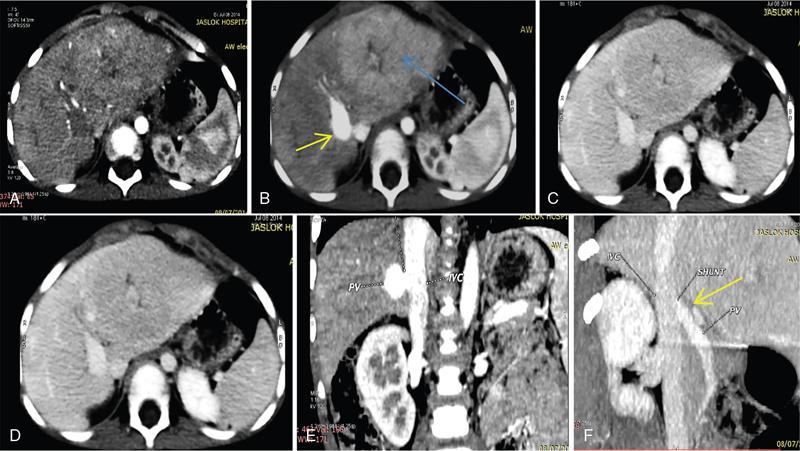
MRI
Treatment
Congenital intrahepatic portosystemic shunts
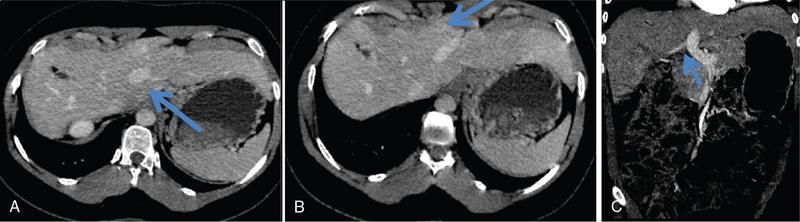
Intrahepatic shunts
Associations and clinical course
Imaging
USG and colour doppler
CT/MRI
Treatment
Patent ductus venosus
Imaging
Treatment
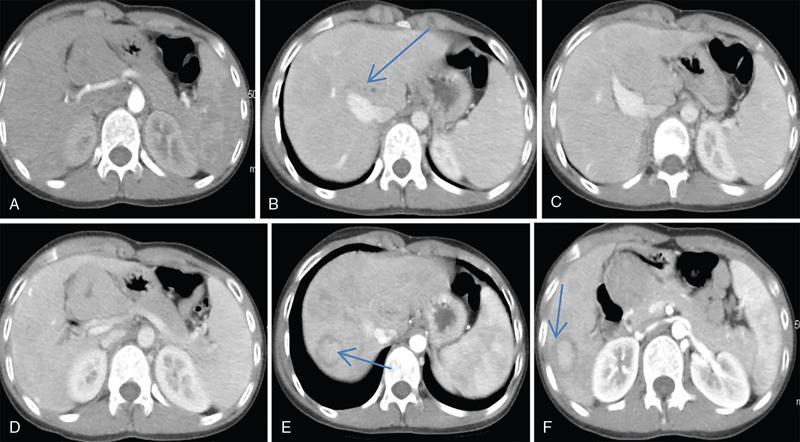
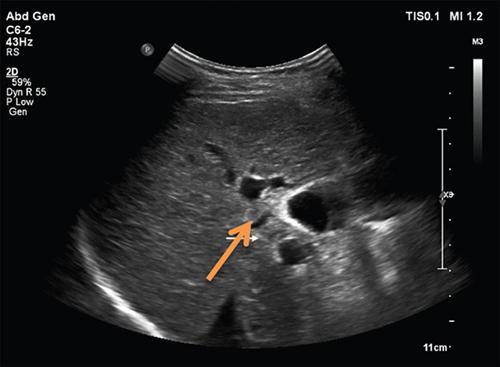
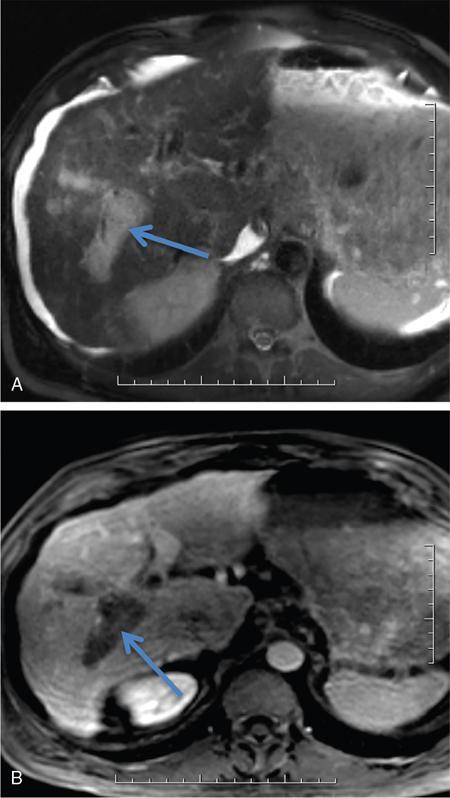
Portal venous thrombosis
Clinical presentation
Imaging
4. EHPVO – extrahepatic portal venous obstruction
Radiology Key
Fastest Radiology Insight Engine