Figure 8-1. Schematic of pancreas anatomy. P = pancreas; D = duodenum; K = kidneys; S = spleen, and the asterisk (*) marks the superior mesenteric vein. (Reprinted with permission from Flay NW, Gorelick FS. Pancreas, anatomy. In: Johnson L, ed. Encyclopedia of Gastroenterology. New York;2004:25–29.)
The arterial supply of the pancreas derives from the celiac trunk and the superior mesenteric artery. A main branch of the celiac trunk, the splenic artery takes its well-recognized tortuous course toward the splenic hilum. As it transverses along the superior edge of the pancreas, the splenic artery gives off the dorsal pancreatic artery, the greater pancreatic artery, and caudal pancreatic artery, which supply the neck, body, and tail. The celiac trunk also gives off the hepatic artery, which becomes the proper hepatic artery after the take-off of the gastroduodenal artery. The gastroduodenal artery courses inferiorly and branches into the anterior and posterior superior pancreaticoduodenal arteries to supply the duodenum and head of the pancreas. The superior mesenteric artery also supplies the head of the pancreas by giving off the two anterior and posterior inferior pancreaticoduodenal arteries, which travel cephalad to converge with their superior counterparts.
The venous drainage of the pancreas parallels its arterial supply. The splenic vein drains the tail and body regions, as it travels along the posterior and superior surfaces of the pancreas toward its confluence with the superior mesenteric vein. The head of the pancreas is drained by the two anterior and posterior inferior pancreaticoduodenal veins, which empty into the superior mesenteric vein. The anterior and posterior superior pancreaticoduodenal veins drain directly into the portal vein.
The pancreas has a simple ductal system for secretion of pancreatic enzymes. The main duct of Wirsung originates in the tail and takes a cephalad and posterior course as it traverses the pancreas. In the head it takes a caudad course, traveling in parallel with the common bile duct, and the two come together before the papilla of Vater, to empty into the duodenum. The accessory or minor pancreatic duct of Santorini joins the main duct where it takes its caudad course, and empties separately in the duodenum at the minor papilla.
NORMAL APPEARANCE OF ULTRASOUND
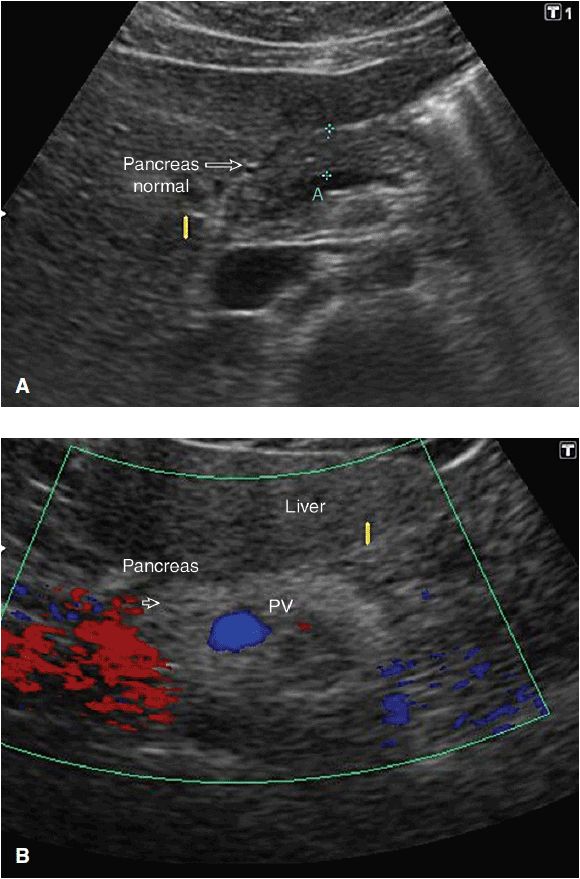
Familiarity with the normal anatomy makes interpretation of the image straightforward. In transabdominal ultrasound the abdominal wall and left lobe of the liver are traversed prior to imaging the pancreas; relevant and consistent sonograghic landmarks are shown in Figure 8-2. Healthy pancreatic tissue is uniformly echogenic and is similar in echotexture to the liver (Figure 8-3A), but appears coarser. Its echogenicity is increased with age and obesity (Figure 8-3B), and is decreased in thinner patients. Commonly, the pancreas undergoes fatty changes with age, and in 35% of patients it becomes difficult to distinguish its echogenicity from the surrounding retroperitoneal fat (Martinez-Noguera et al, 2007).
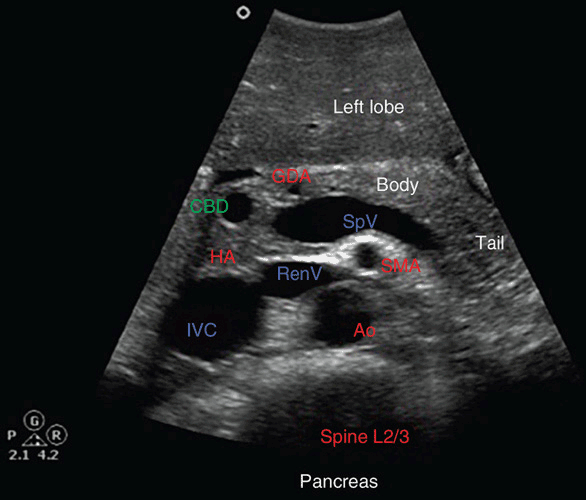
Figure 8-2. Transverse transabdominal ultrasound view of the pancreas. CBD = common bile duct; HA = hepatic artery; GDA = gastroduodenal artery; SpV = splenic vein; RenV = renal vein; SMA = superior mesenteric artery; Ao = aorta; IVC = inferior vena cava.
Figure 8-3. (A) Sonographic appearance of healthy pancreatic tissue. Note the uniformity of the gland, and its similarity in echotexture to the left lobe of the liver overlying it. (B) A fatty pancreas appears brighter than liver.
The normal pancreatic duct measures 2–3 mm in diameter and is seen as an anechoic tubular structure with two echogenic borders. Because it is thinner in the tail, it is often not appreciable in this location, particularly with transabdominal approaches. The common bile duct can usually be visualized on transverse imaging in cross section in the head of the pancreas, where it normally measures below 4 mm. The hepatic artery is generally anterome-dial to the bile duct, depending on the region imaged. The gastroduodenal artery can often be seen in cross section along the right anterior aspect of the pancreatic head. The neck of the pancreas is defined as it courses anterior to the superior mesenteric-splenic-portal venous confluence. The superior mesenteric artery lies medial to that in cross section, and often the left renal vein can be seen coursing between the superior mesenteric artery and aorta as it empties into the inferior vena cava.
SCANNING TECHNIQUE FOR OBTAINING THESE IMAGES
The pancreas is distinguished on ultrasound by its uniquely glandular parenchyma; however, since its echogenicity varies with age and disease process, it is most useful to rely on the adjacent anatomic structures as landmarks when tracing the gland in the transverse and longitudinal orientations. Of particular help in its identification is its relation to the aorta and splenic vein, which are both easily and consistently identified. In the transverse scan the splenic vein serves as the most useful landmark, whereas in the longitudinal/sagittal scan the aorta and its main branches are more helpful.
Using the left lobe of the liver as a starting point, in the transverse scan the pancreas is identified anterior to the splenic vein, which in turn lies anterior to the superior mesenteric artery and aorta. From there the transducer is moved upward and toward the left to scan the tail, which takes a posterior direction and often must be seen through the spleen. Next, to visualize the head of the pancreas in the transverse orientation, the transducer is returned to the midline starting position and then moved to the right and slightly downward. Helpful landmarks here are the superior mesenteric and splenic vein confluence and the left renal vein as it enters the inferior vena cava.
To view the pancreas longitudinally, it is helpful to assume the transverse starting position as mentioned above, and locate the aorta. Then, while keeping the gland in view, rotate the transducer toward a longitudinal orientation, maintaining the aorta in the center of the field, so that it lies in parallel to the aorta. Structures that serve as useful longitudinal landmarks are the aorta, celiac trunk, superior mesenteric artery, and the inferior vena cava.
Intraoperative imaging has the advantage of removing the abdominal wall from the ultrasound field; thus, higher frequencies may be used for greater resolution. Contemporary laparoscopic probes articulate in two directions, so that the pancreas may be comfortably and completely imaged from anywhere in the upper abdomen through an appropriately sized transducer. The pancreas may be imaged through the stomach, duodenum, or left lobe of the liver, although for target localization purposes, the lesser sac is opened, exposing the surface of the pancreas, so resection or biopsy may rely on surface landmarks. The uncinate and head of the gland may also be imaged through the duodenum.
As the pancreas abuts the stomach and duodenum, upper endoscopic ultrasound is an ideal modality for precise investigation of the gland. Expertise with this technique requires exquisite familiarity of the anatomy and transducer position, and further guidance on these techniques can be found elsewhere in this text.
COMMON FINDINGS/ABNORMALITIES
Ultrasound is often utilized to characterize cysts or tumors in the pancreas. There are several prognostic features definable on ultrasound, including size, borders, echotexture, or presence and type of internal architecture in cysts. Fluid aspirate and tissue biopsies are easily obtained via EUS-guided fine needle aspiration.
Widespread use of cross-sectional imaging has led to an increased reported incidence of asymptomatic cystic lesions of the pancreas, both benign and malignant. Many of these incidental findings, if not already neoplastic, have the potential for malignant transformation, and it is important to characterize the lesions as fully as possible upon initial diagnosis. Imaging characteristics such as the size of the cyst, complexity of its borders, and presence or absence of internal architecture such as nodules or papillary projections all contribute to risk stratification.
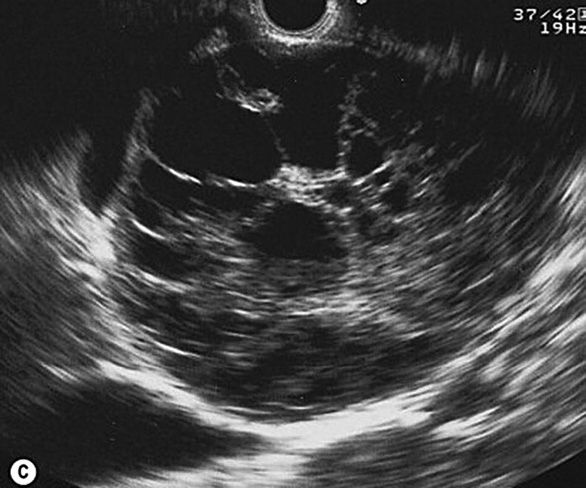
International consensus guidelines (“Sendai criteria”) suggest that for cysts with diameter greater than 30 mm surgical resection be considered (Tanaka et al, 2006); others (Jang, 2008, Rodriguez et al, 2007) have argued for a lower size criterion. A simple, anechoic cyst with one chamber usually predicts a benign neoplasm. Internal echoes, papillary projections, septae or mural nodules within the cyst can raise concern for occult or impending malignancy (Figure 8-4). Serous cystadenoma, a generally benign condition, has a characteristic microcystic or honeycomb appearance on imaging (Figure 8-5). Intraductal papillary mucinous neoplasm (IPMN) is a disorder of mucin secretion, leading to pancreatic ductal dilation. Considered a premalignant condition, up to 60% already harbor malignancy at the time of discovery. The thick mucinous secretions can also lead to acute or chronic pancreatitis. When malignancy is identified, or only a segment of the gland is affected, partial pancreatectomy is performed; in this scenario, though, it is imperative to maintain close, regular surveillance of the remainder of the gland. Endoscopic ultrasound has proven invaluable in the characterization of cystic lesions, with highly sensitive architecture description as well as the capability for sampling of cyst fluid, and potential for reliable, repeatable follow-up examinations.
Figure 8-4. Mucinous cystic neoplasm in the head of the pancreas with a mural nodule. (Reprinted with permission from Schaberg FJ, Doyle MBM, Chapman WC, Vollmer CM, Zalieckas JM, Birkett DH, Miner TJ, Mazzaglia PJ. Incidental findings at surgery—Part 1. Curr Prob Surg. 2008 May;45(5): 325–374.)
Figure 8-5. Serous cystadenoma on endoscopic ultrasound image with honeycomb appearance. (Reprinted with permission from Amin Z. Pancreas. In: Clinical Ultrasound. 3rd ed. Edinburgh;2011:285–323.)
Solid tumors of the pancreas can be primary neoplasms of the pancreas (adenocarcinoma, neuroendocrine tumors), metastases (renal cell carcinoma), or rarer entities such as pancreatic lymphoma or sarcoma. Similar to cystic lesions, ultrasound is invaluable in the characterization, biopsy, and treatment plan of solid pancreatic lesions. Most if not all solid masses will need a biopsy prior to surgery. Those with irregular borders are more concerning for adenocarcinoma (Figure 8-6), whereas lesions with a smooth, regular border are more typical of a neuroendocrine tumor (Figure 8-7). Extremely relevant to the treatment plan is the assessment of the tumor for vascular involvement, as the retroperito-neal pancreas is essentially draped over numerous major abdominal blood vessels. Figure 8-8 shows a hypoechoic tumor abutting the superior mesenteric vein, with loss of the interface between the two, suggesting invasion. Doppler ultrasound can help delineate blood vessels and identify narrowing or the vessel or thrombosis, consistent with tumor invasion (Figure 8-9). As with cystic lesions, endoscopic ultrasound is invaluable for image-guided biopsy of solid pancreatic masses for tissue diagnosis (Figure 8-10).
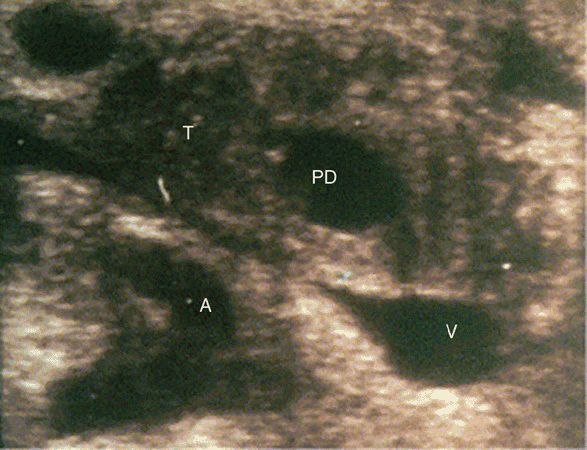
Figure 8-6. Intraoperative ultrasound scan of the head of the pancreas, showing a hypoechoic tumor (T) adjacent to a dilated pancreatic duct (PD). The splenic vein (V) and celiac trunk (A) are visible and clearly free of tumor. (Reprinted with permission from Long EE, Van Dam J, Weinstein S, Jeffrey B, Desser T, Norton JA. Computed tomography, endoscopic, laparoscopic, and intra-operative sonography for assessing resectability of pancreatic cancer. Surg Oncol. 2005 Aug;14(2):105–113.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree