3 Evolution of Lung Cancer Screening
Summary
This chapter discusses the evolution of initial unsuccessful early-detection lung cancer–screening programs with sputum cytology and chest radiography and the reasons for their failure to the current state-of-the-art early detection with LDCT (low-dose computed tomography). The natural history of lung cancer, the National Lung Screening Trials (NLST) results, and the endorsement of lung cancer screening for eligible high-risk persons by the U.S. Preventive Services Task Forces (USPSTF) and the Centers for Medicare & Medicaid Services (CMS) are also addressed.
Keywords: lung cancer, screening, cancer growth, chest radiography, sputum cytology, PLCO, NLST, USPSTF, CMS
3.1 What Is Screening?
Screening means testing for diseases, such as lung cancer, while the individual is still healthy, that is, when there are no symptoms or clinical signs of disease. The purpose of the screen is to find disease early on, when treatment options may be more successful, and individuals may have a much better chance of cure. The intent of lung cancer screening is to detect the disease at its earliest possible stage, while it is small, confined to the thorax and before it invades adjacent tissues, extends beyond the lung, and/or causes symptoms, at which point the chance for cure dramatically diminishes and treatment is less successful. The early detection of lung cancer when it is still small and localized is of paramount importance in mortality reduction. Individuals not infrequently undergo other forms of screening for similar purposes (e.g., blood pressure or cholesterol checks; annual mammography; PAP smear; colonoscopy; PSA; etc.).
3.2 Natural History of Lung Cancer
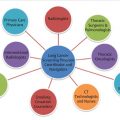
Unfortunately, lung cancer is a “sneaky” disease. It may harbor in the body for years, growing in size and volume, before becoming large enough to be detectable on a conventional radiograph or before it causes symptoms such as cough, hemoptysis, or weight loss (▶ Fig. 3.1). In the absence of screening, most patients already have locally advanced or metastatic disease when they first see their primary care doctor with these or similar symptoms. Only 15 to 25% of patients have potentially resectable, early-stage disease confined to the chest at that time. Over the years, screening for the early detection of lung cancer has included miniature chest photofluorography, conventional chest radiography, sputum cytology, and more recently low-dose helical computed tomography (CT).

3.3 Chest Radiography
The possibility of tobacco as a primary cause for the increasing incidence of lung cancer was introduced in the early 1900s. The first case control study to highlight this association was from a German scholar, Franz Hermann Müller, in 1939. By the 1950s, several epidemiologic studies further supported this link, concluding smoking 35 cigarettes or more per day increased the odds of death by lung cancer by a factor of 40. Concurrently, in the 1940s, mass screening for tuberculosis began using miniature chest photofluorography or abreugraphy. These early X-ray images were a miniature 50- to 100-mm photograph of the screen of an X-ray fluoroscopy of the thorax and were typically viewed on a projector. Screening with chest radiography was instrumental in the early detection of tuberculosis, leading to early appropriate antimicrobial treatment and improved prognosis. As most thoracic tumors were visible on photofluorography, there was a potential for reducing mortality through screening and early treatment with this modality. Early efforts at screening in the 1950s with several large uncontrolled studies showed mixed results. The earliest U.S.-led studies conducted in the 1950s were the Philadelphia Pulmonary Neoplasm Research Project (PNRP) and a cooperative pilot study between the American Cancer Society and the Veterans Administration (VA). Combined, these two studies enrolled 20,743 males older than 45 years to undergo screening with chest radiography every 6 months with a questionnaire regarding smoking habits, occupation, and respiratory symptoms. A total of 194 screen-detected
Conversely, two large uncontrolled screening studies in Japan and the United Kingdom suggested some survival benefit. The Tokyo Metropolitan Government study (1,871,374 men and women, all ages) and the South London Lung Cancer Study (67,400 men older than 45 years) showed higher rates of resectability (56%) with improved long-term survival against retrospective comparison groups (44 vs. 20% at 5 years and 18 vs. 9% at 4 years, respectively). Nearly a decade later, two nonrandomized controlled studies conducted in the United Kingdom and Germany were the first to report disease-specific mortality rates. The North London Lung Cancer Study and the Erfurt County Germany Study compared semiannual chest X-ray screening against a control group who received less frequent imaging. While 5-year survival in both studies was superior to the control groups (15 vs. 6% in the UK and 28 vs. 19% in Germany, respectively), there were no differences in mortality rates (0.6–0.8/1,000 persons screened).
By the 1970s, advancements in X-ray technology and imaging processing, sputum cytology techniques, and the flexible fiberoptic bronchoscope generated renewed interest in lung cancer screening using these new modalities. The National Cancer Institute (NCI)—the Mayo Lung Project (MLP), the Johns Hopkins Lung Project (JHLP), and the Memorial Sloan Kettering Lung Project (MSKLP), sponsored three large randomized-control trials (RCT). A fourth RCT was conducted in Czechoslovakia. The MLP and Czechoslovakian studies attempted to assess the efficacy of chest radiography. The JHLP and MSKLP were designed to assess the utility of adding sputum cytology to annual chest X-ray screening. The MLP consisted of men older than 45 years with a history of heavy smoking. After an initial prevalence screen, 9,211 men were subsequently randomized to chest X-ray and sputum screening every 4 months with regular reminders or a control group where annual screening was recommended but without routine reminders. At the end of the study, frequent screening detected a 29% higher incidence of lung cancers compared to the controls (206 vs. 160, respectively). The screened group had a favorable shift toward early-stage tumors (38 vs. 25%, respectively) and higher resectability rates (46 vs. 32%, respectively). Despite an apparent survival advantage in the screened group (33 vs. 15%, respectively), there was no difference in mortality (3.2/1,000 vs. 3.0/1,000 person-years, p = 0.72). Notably, there may have been a substantial degree of statistical contamination within the control group—nearly half had obtained annual chest X-rays, with 73% receiving X-rays in the final 2 years.
The Czechoslovakian study screened 6,346 men between the ages of 40 and 64 years. They were randomized to chest X-ray and sputum cytology screening every 6 months for 3 years and a control group who had no testing for 3 years following the prevalence screen. There were 36 screen-detected lung cancers in the study group compared to 19 in the control group with resectability rates of 25 and 16%, respectively. Similar to the MLP, there was an apparent stage shift with favorable 5-year survival in the screened group compared to controls (24 vs. 0%, p < 0.01) and no difference in mortality (1.7 vs. 1.5/1,000 person-years).
Despite the proportion of earlier screen-detected lung cancers and apparent survival advantage, these early RCTs were flawed and ultimately incapable of determining whether screening for lung cancer is better than no screening at all. They did not have a proper control group, excluded women, lacked power, and did not consistently report operative morbidity and mortality. The lack of mortality benefit in the MLP and Czechoslovakian studies was likely influenced by the above factors in addition to biases such as lead time and overdiagnosis. Furthermore, a Cochrane meta-analysis found a relative mortality increase of 11% attributed to frequent lung cancer screening and determined, based on available data, that there was insufficient evidence to recommend lung cancer screening with chest radiography or sputum cytology.
The most recent RCT concerning chest X-ray screening called the Prostate, Lung, Colorectal, and Ovarian (PLCO) trial was conducted to address the statistical flaws in the NCI-sponsored RCTs. Across 10 clinical sites, 154,901 men and women between the ages of 55 and 74 years were randomized to annual screening with posteroanterior (PA) chest X-rays versus usual care for 4 years. Over half were either active or former smokers. Patients were followed for a maximum duration of 13 years. Compliance with screening throughout the study was fair (79–87%) with little contamination in the control arm (11% had imaging). There was no significant difference in lung cancer incidence between groups; however, there were more stage I cancers in the screened versus usual care group (462 vs. 374, respectively). Comparable to the MLP and Czechoslovakian studies, disease-specific lung cancer mortality was similar between groups (relative risk [RR], 0.99; 95% confidence interval [CI], 0.87–1.22; p = 0.48).
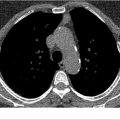
Thus, screening for the early detection of lung cancer with conventional radiography has not been widely accepted. Screening with conventional radiography is low-yield and has not been proven to reduce patient-specific mortality. In part, this is likely related to difficulty in perceiving upper lobe lesions often silhouetted by overlapping osseous structures such as the anterior and posterior ribs and clavicles, differentiating central hilar lesions from the normal hilar anatomy, and difficulty in perceiving nodules below a threshold of 1 to 2 cm, at which point more than 1 billion cells are already present (▶ Fig. 3.1; ▶ Fig. 3.2).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree