Abstract
Implementation of an abbreviated (AB-MR) technique as a method for breast screening for women at moderate (15-20%) risk and/or mammographically dense breasts is an innovative and challenging endeavor. In this chapter, a practical review of acquisition protocols, technical requirements, recruitment and clinical workflow for AB-MR will be discussed. In order to provide optimal screening, breast imaging facilities need to reconsider and modify their MRI workflow to allow for efficient throughput of women to be screened, while also maintaining technical and interpretive excellence. Approximately 50% of women will fall into the dense breast category [BI-RADS categories C & D], where the sensitivity of mammography is diminished and overlapping dense tissue may increase the false-positive call-back rate. Screening with AB-MR has been shown to be a more sensitive screening method for these patients.
If AB-MR breast screening is successfully implemented, providing lower costs and efficient workflow, women would greatly benefit by the detection of a significantly larger number of high-grade cancers than by other imaging methods while also reducing interval cancers and node positivity. The underlying principles of screening MR interpretation are similar, whether an abbreviated protocol or a full protocol is utilized. Descriptions using the BI-RADS lexicon categories ofl background parenchymal enhancement (BPE) and selected MRI-detected lesions are shown. Ten case examples of BPE and 21 case examples of MR screen-detected benign and malignant lesions are shown.
4 Screening MRI: Clinical Implementation
4.1 Introduction
Breast magnetic resonance imaging (MRI) has become the mainstay method for screening of high-risk women due to its high sensitivity for breast cancer compared to mammography and ultrasound. Although higher field strength magnets, increasing numbers of channels in breast coils, and advanced analytic methods have improved breast MRI greatly over the years, the principal technical acquisition methods used for breast screening have remained basically unchanged. The standard MRI protocol has consistently included a precontrast T2-weighted (T2w) acquisition followed by a dynamic contrast-enhanced MRI (DCE-MRI) sequence, encompassing a series of T1w acquisitions before and for approximately 5 to 7 minutes after the intravenous administration of a gadolinium-based contrast medium (GBCM). 1 Physician referrals for either a screening or a diagnostic indication will generally result in women receiving the same MRI acquisition protocol today. In almost all of the early high-risk screening trials (using a full diagnostic MRI technique), the sensitivity of MRI was higher than mammography, and the specificity was lower. As the technology has matured, the specificity of MRI screening has increased due to improved technical and radiological expertise. The current 6-month follow-up recommendation rates and positive predictive value (PPV3) of biopsy rates at MRI screening are now comparable to those of mammography.
The advent of breast density legislation has changed the landscape for screening women with dense breasts, and new methods are now sought for supplemental screening. 2 Despite its high sensitivity, MRI use has generally been restricted to the screening of high-risk women only for a number of reasons that primarily include time and costs. The length of time required to complete a standard MRI examination is often 30 minutes or more, the cost of the examination is much higher than mammography, and most clinical radiological practices are not geared toward efficient MRI screening. Large prospective screening studies of high-risk women employed annual mammography and annual MRI, yearly simultaneous screening allowing easy cross-referencing for interpretation and audit purposes.
4.2 Abbreviated MRI Technique
Abbreviated breast MRI (AB-MR) was developed to provide a low-cost, efficient, screening method, streamlined workflow, and short interpretation times being essential requirements for any image-based screening study. An abbreviated protocol usually includes only one or two postcontrast dynamic timepoints with the expectation that cancer enhancement will be most conspicuous at the first postcontrast timepoint, with the added benefit that background parenchymal enhancement will be less evident. The concept of abbreviated MRI was pioneered by Dr. Kuhl who published the results of a prospective clinical study on the use of abbreviated breast MRI in 2014. 3 In this study, use of the maximum-intensity projection (MIP) image to provide a quick overview to check for presence or absence of significant enhancement, significantly reducing radiologist reading times. Individual subtracted images were subsequently reviewed as necessary for further categorization of abnormal enhancement. Although a complete dynamic series was acquired, all rapidly enhancing lesions were identified on the first postcontrast subtracted images. For hundred and forty-three women at mildly to moderately increased risk underwent 606 MRI screening rounds. Of the 11 cancers identified in this study, four (36%) were in the intraductal stage, 7 (64%) invasive, with a median size of 8 mm, and all malignancies were node negative. All participants in the study had received a normal mammogram at time of recruitment and the supplementary cancer yield of MRI was 18.3 per 1000 women. Radiologist’s reading time was below 3 seconds for interpretation of a MIP image (is there significant enhancement or not?) and below 30 seconds for interpretation of the complete study when significant enhancement was identified on the MIP image. These short reading times are competitive with batch reading times of screening mammograms, and, for example, are substantially shorter than the time needed for review of tomosynthesis studies. Although the AB-MR protocol is likely not sufficient for a complete diagnostic examination, in the screening setting, this study showed a high detection rate without a corresponding high false-positive rate, an indication of an excellent high-quality screening test. These results have prompted other researchers to consider further development of shorter MRI protocols for screening.
Subsequent studies using AB-MR technique have further supported the concept of shortened acquisitions for screening. Various protocols acquiring a precontrast series, one or possibly two postcontrast series and in some cases a T2w series were employed, results showing that comparison performance between a shortened breast MRI protocol and a standard protocol were similar. 3 , 4 , 5 , 6 In the United States, AB-MR protocols have already been incorporated into some breast practices and modified according to the techniques commonly used in North America. Protocols for AB-MR screening are usually defined as an MRI study with a scan time of less than 10 minutes compared to a longer 20- to 40-minute full diagnostic breast MRI. Generally, a T2w scan followed by one T1w pre- and one postcontrast examination completes the protocol. It is important to note that standard kinetic assessment is not possible with AB-MR technique because delayed dynamic acquisitions(s) are not obtained. An ECOG/ACRIN trial, “EA1141: Comparison of Abbreviated Breast MRI and Digital Breast Tomosynthesis in Breast Cancer Screening in Women with Dense Breasts,” is now underway in the United States and Germany and will provide additional data on the efficacy of the AB-MR approach to screening. 7 The abbreviated technique has the potential to achieve not only sensitivities comparable to a full diagnostic protocol but also short imaging times and decreased costs. Introduction of this promising new application of breast MRI will be challenging, evoking memories of the early days of mammography screening when radiologists revamped their practices to improve efficiency and throughput. 8 Ultrafast (UF) (accelerated) MRI enables shorter acquisition times (3-7 seconds) and can be included into an AB-MR protocol. A variety of different UF techniques have been reported and discussed extensively in the previous chapter. Although most of the reported clinical studies are feasibility investigations, one study conducted a multicenter reader investigation of ultrafast screening MRI vs. a full diagnostic protocol. 9 No significant difference in the sensitivity of a full diagnostic protocol vs. an ultrafast protocol (0.86 vs. 0.84, P = 0.50) was found and specificity was significantly higher for the ultrafast protocol (0.76 full diagnostic vs. 0.82 ultrafast, P = 0.002), A significant decrease in reader time was also noted.
4.3 Recruitment for AB-MR Screening
The target population for adjunctive AB-MR annual screening will be asymptomatic women at mild-to-moderate increased risk (15–20%) and/or with dense breast tissue at mammography, beginning at age 40. It is possible that as the technology progresses, MRI screening of high-risk women with AB-MR may be effective as well; however more data are needed to support this hypothesis. As previously noted, approximately 50% of women will fall into the dense breast category (BIRADS categories C and D), where the sensitivity of mammography is diminished, and overlapping dense tissue may increase the false-positive callback rate. Several risk assessment models can be used to estimate lifetime breast cancer risk as discussed in Chapter 2. Radiologists have implemented their own risk assessment program in some practices, and in large breast centers, a dedicated genetic counselor may be on staff assisting the imaging team. In recent years, more than half of the states in the United States have passed mandatory breast density notification legislation, and several more have introduced a bill. 2 These laws generally recommend that women should consider adjunctive imaging screening methods if they are known to have dense breast tissue at mammography. By Mammography Quality Standards Act national statute, 10 letters in lay language are sent to every woman with dense breast tissue explaining that this finding is common, not abnormal, and that dense breast tissue can make it harder to find cancer on a mammogram and may increase her breast cancer risk. These lay reports inform women and recommend that the benefits of adjunctive screening methods should be discussed with their personal physician. 6
Education of the medical community is needed to provide information on the benefits of improved cancer detection directed toward women in both the high- and moderate-risk categories. Seminars and conferences emphasizing the benefits and efficiencies of AB-MR and the distinction between a screening and a diagnostic MRI examination could be held at community hospitals, physician practices, and local and national breast health organizations. Relevant publications could be circulated to physicians, genetic counselors, and allied health professionals involved in breast care. In local radiologic practices, informational brochures and printed literature could be distributed among referring physicians and placed in the breast imaging waiting room. Identification of those women within an existing mammography screening practice, who have been shown by a previous full field digital mammography (FFDM) examination to have dense breast tissue, could be directly informed about the value of additional AB-MR screening at the time of their routine mammography appointment.
4.3.1 Equipment
Magnet Strength
The majority of breast DCE-MRI studies are still performed at magnetic field strengths of 1.5 Tesla (T); however, recently there has been an increase in the use of higher field strength magnets, 3 T, and dedicated breast coils with an increasing number of channels. 7 A study by Elsamaloty et al reported increased sensitivity and specificity of DCE-MRI when performed at 3T, finding values of 100 and 93.9%, respectively. 8 Another study by Rahbar et al reported on breast MRI at 3 T and showed a higher correlation with the final pathologic size of ductal carcinoma in situ (DCIS) lesions, when compared to 1.5 T. The authors concluded that higher field strength might prove to be more accurate for assessment of disease extent in the preoperative setting. 9 Although excellent screening MRI technique is possible at both field strengths, these recent studies suggest that growing experience in scanning at higher field strength could lead to an even greater overall increase of both sensitivity and specificity at MRI.
Contrast Injection Devices
Power injectors are preferable for contrast injection because they provide consistent timing of the injection of a gadolinium-based contrast agent (GBCA), ensuring that the injection rate is the same for every patient, every time. Injection doses are calculated according to body weight, and a standard dose of 0.1 mmol/kg should be administered as a bolus, with an injection rate of 2 mL/s followed by a saline flush of at least 10 mL. 10 Power injectors can be triggered from outside the magnet room, facilitating image timing and minimizing patient motion. Standardized injections are especially important for women undergoing annual screening, because direct comparison of kinetic data from one examination to the next is necessary, and elimination of variation due to differences in the injection rate allows valid comparisons.
4.4 Preprocedure Scheduling and Preparation
Streamlined workflow is critical for well-organized screening, and an efficient breast MRI screening program requires well-trained scheduling and reception staff. FFDM and AB-MR studies can be scheduled contemporaneously, so that both imaging procedures are completed within a 1-hour time frame. It is important to note that current ACS guidelines for breast MRI screening of high-risk women do not address the timing of MRI in conjunction with FFDM. Screening protocols therefore include women who prefer to undergo both procedures together on an annual basis and those who prefer to alternate between AB-MR and FFDM every 6 months.
Preparation beforehand is essential in order to achieve effective streamlined workflow on the day of the AB-MR examination. Women should be prescreened, usually by telephone, for claustrophobia, allergic predisposition or history of prior reaction to contrast agents, presence of implantable devices, and history of renal disease. Some practices deliver a packet of information to the woman by mail or by e-mail before the appointment date, covering topics such as the length of time of the procedure, the need for contrast injection, the possibility of follow-up imaging, or biopsy. Other practices may rely on their website for providing this information. Consent for the MRI procedure and administrative details regarding payment and insurance preauthorization should be arranged by phone in advance of the appointment day. Some women may have further questions about breast MRI screening before consenting for the procedure for the first time; a discussion in the radiology department with either a qualified technologist or radiologist before the examination appointment is confirmed can be helpful.
Scheduling of premenopausal women for screening MRI requires special attention. Although premenopausal women undergoing a diagnostic MRI study are not always scheduled according to their menstrual cycle, preferential scheduling is useful for screening. Although it is often recommended that screening breast MRIs should be performed during the 7 to 10 days of the menstrual cycle in order to minimize the degree of background parenchymal enhancement (BPE), some recent studies have suggested that the degree of BPE does not impact MRI sensitivity or specificity. Investigations have shown that BPE typically increases during the fourth week of the menstrual cycle, thus, if at all possible, imaging should be avoided in the premenstrual phase of the cycle. Decreased BPE is found in postmenopausal women and those treated with radiation therapy and antiestrogen therapy (tamoxifen and aromatase inhibitors). The goal is to image when the background enhancement of normal fibroglandular breast tissue is lower, abnormal findings are more conspicuous, and false-positive findings are less frequent. 11 , 12 , 13 Scheduling of screening appointments for premenopausal women requires flexibility, and appointments may need to be adjusted on an individual basis. Women using oral contraceptives should observe the menstrual cycle imaging recommendations. Certain women may experience irregular menses, particularly in the perimenopausal years, thus encountering difficulty in selecting the appropriate timing for their examination. In these instances, blood sampling for estimation of serum progesterone levels is helpful for determination of the optimal time for breast MRI. This method may also prove to be particularly useful when earlier examinations have been deemed nondiagnostic because of marked BPE. 14 It is interesting to note that a recent report indicates that hormone replacement therapy has negligible effect on the BPE of postmenopausal women who undergo MRI. 15
When a woman is referred for MR examination and it is questionable as to whether she will fit into the magnet bore and the breast coil, the issue can be resolved by inviting her to view the magnet and breast coil and attempting to position her in the device. If positioning and placement prove unsuccessful, then alternate screening or diagnostic methods should be employed. All such questions and concerns should be managed, if at all possible, prior to the day of the screening appointment. Attempts must be made to obtain all prior breast image examinations in advance, 16 and these studies should be uploaded onto the picture archiving and communication system (PACS) before the patient arrives for her screening examination.
4.5 Imaging Procedure
Women presenting for screening MRI will often experience increased anxiety, fearing the possibility of a finding that might be malignant. A separate check-in line will minimize the anxiety of waiting with other patients and expedite workflow. Women should receive clear instructions and a complete explanation of the MR procedure by a radiology technologist or nurse once she enters the MRI suite. She will be asked to fill out a detailed questionnaire to confirm that she has no contraindications to the MR examination or to the contrast injection and will be asked to sign a specific informed consent. She will then be directed to the dressing area where she will change into a patient gown, having removed her brassiere, jewelry, and any clothing zippers or other metallic objects.
In the preparation room of the MRI suite, a technologist or nurse will provide venous access and inform the woman that she might experience a warm or tingling sensation in her arm, possibly extending throughout her body, both during and following the injection. She will also be instructed to avoid any movement during scanning, emphasizing the importance of remaining still and avoiding the need for repeat examination. Once the woman is positioned prone on the MRI table, the technologist will position her so that her breasts are symmetrically placed into the wells of the coil with nipple, palpable masses, and scar markers applied, if requested by the radiologist. Folds or overlapping of breast tissue at the edges of the coil should be avoided, and slight breast compression may be used to reduce motion artifacts. The scanning procedure is intermittently noisy, and the woman should be informed that she can be given earplugs or earphones with a choice of music, if she wishes to minimize the noise during image acquisition. It is important that a method of verbal communication between the woman and the technologist or radiologist is established before scanning takes place. An alarm system device should be provided to her, so that she is aware that when she activates the alarm, the procedure will be stopped, and she will immediately receive assistance.
Following completion of imaging, the intravenous line is removed, the puncture site is compressed, and the woman will then be asked to sit on the table for a moment to avoid any dizziness, and then exit from the magnet room. Once she is dressed in her own clothes, she may be asked to remain in the department for about 15 minutes to check for any possible delayed reaction to the GBCA before leaving the facility.
4.5.1 Precautions and Contraindications
Claustrophobia
Claustrophobia may be experienced by some women when they are asked to enter, and remain immobile, inside the narrow bore of the magnet, for a period of about 10 minutes. 17 Claustrophobia is usually mild and generally less severe when she is able to enter the scanner with her feet first rather than her head first. Moveable trolleys with built-in breast coils are designed to enter the magnet feet first and are especially useful in this regard (Fig. 4‑1). These trolleys also allow streamlined workflow when two are available on site; one can be used in the scanning room during image acquisition, and a second trolley can be used to position the patient in the coil ready for scanning. Women are usually able to tolerate the study without difficulty, and verbal reassurance from the technologist may help her to remain immobile in between acquisition sequences. Women who are extremely anxious may benefit from a viewing of the scanning room and magnet before the examination is scheduled. Sedation may be needed for women with a history of severe claustrophobia, and this is usually arranged by consultation with her referring physician. When sedation is provided, arrangements must be made for another individual to accompany the woman home after the procedure.

Allergic Predisposition
GBCAs are extremely well tolerated by the vast majority of patients who undergo DCE-MRI. Acute adverse reactions are rare, and reports indicate a lower frequency than is reported following administration of iodinated contrast media. Women with a history of multiple allergies, bronchial asthma, or previous allergic reactions to GBCAs have a higher risk for allergic reactions. These women should discuss their allergic history with their referring physicians. Precautionary methods can be taken in these cases, including preprocedure administration of antihistaminic and corticosteroid drugs. In women with serious allergic symptoms, a balance between the potential advantages of MRI and the potential risks of serious allergic reaction should be considered. It is important to note, however, that here is no cross-reactivity between GBCAs and iodinated contrast media.
Foreign Bodies
Absolute contraindications to MRI include the presence of ferromagnetic intracranial aneurysm clips and MR-incompatible implanted electronic devices, such as pacemakers, implantable cardioverter defibrillators, and neurostimulators. Women with intravascular stents or metal screws or plates for osteosynthesis can safely undergo breast MRI 6 weeks following surgical implantation. Questions regarding a history of prior eye injury from a metal object (metal slivers, metal shavings) are routine before any MRI examination is undertaken, and orbital radiographs may be necessary to document the presence or absence of metal foreign bodies in some cases. Enquires should also be made concerning body tattoos or permanent makeup, including tattooed eyeliner. If the tattoos are extensive or very dark, they may contain iron pigments, and the woman may experience radiofrequency (RF) heating of the tattooed tissue causing local burns. Burning may occur if the tissue in question is located within the volume in which the body coil is being used for RF transmission. Cold compresses or ice packs placed over the tattooed areas, locally applied throughout the MRI examination, may alleviate the heat effect. Additional information may be found in the ACR guidance document on MR safe practices. 18
Renal Function
Renal function test, obtaining a creatinine level and estimating the glomerular filtration rate (GFR), is recommended within 30 days of the scheduled examination for women over the age of 60. Testing should also be obtained for younger women with a history of bladder or kidney disease, diabetes mellitus, or cardiovascular disease. 19 , 20 AB-MR screening should be avoided in women with severely impaired renal function because injection of a GBCA may lead to a rare condition known as nephrogenic systemic fibrosis. 21 DCE-MRI is generally contraindicated in pregnant women and AB-MR should be avoided during pregnancy and lactation. 22
Costs
Although MRI may eventually become a routine method for breast cancer surveillance, the high cost of the examination is currently a major impediment to wider application of its use. Studies of the cost-effectiveness of annual screening with both MRI and mammography have only incorporated to date; those women deemed to be at the highest risk of developing breast cancer. The costs associated with screening MRI, as with other screening methods, include not only those associated with the procedure itself, but also include the downstream costs generated by repeat MRI examinations, targeted ultrasound studies, and image-guided biopsies of screen-detected breast lesions.
Plevritis and colleagues reported in 2006 that the addition of breast MRI to a screening regimen was cost-effective in both BRCA1 and BRCA2 mutation carriers. 23 They showed that although the cost–benefits were greatest in patients aged 40 to 49 years for both mutation types, the cost–benefits per quality-adjusted life year (QALY) for women aged 35 to 54 were greater in women with BRCA1 ($55,420) than for BRCA2 carriers ($130,695), and if women with dense breasts were considered, the costs decreased for BRCA1 ($41,183) and for BRCA2 carriers ($98,454).
Another report in 2014 by Ahern and colleagues studied various options for integrating mammography and MRI schedules into a screening protocol for women with a strong family history of breast cancer and a lifetime risk of greater than 25% or higher. Using current costs of MRI examinations and an incremental cost-effectiveness ratio of $100,000 per QALY, they found that the most cost-effective strategy was an alternating schedule of MRI and mammography examinations plus a clinical breast examination every year from age 30 to 74. For those women with a 50% lifetime risk, the recommended strategy was to follow the same screening protocol but to alternate these examinations every 6 months, provided there was a 70% reduction in MRI costs. At 75% risk, the recommended strategy became biennial MRI combined with mammography plus clinical breast examination every 6 months. 24
Health care reimbursement for breast MRI screening is variable among developed countries across the world. When considering costs of screening women at intermediate or average risk, it is certainly essential that the imaging time must be shorter and workflow efficient in order to decrease costs. Future cost–benefit analyses of AB-MR breast screening need to take into account the proven low-interval cancer rate and early detection rate of biologically aggressive cancers, which allow lower downstream costs when compared to treatment of larger tumors. Analyses must consider also that although ultrasound procedure costs are lower, the method is less sensitive and is associated with substantial costs associated with recalls and biopsies.
4.6 Screening Interpretation
Interpretation of screening MRI examinations is one of the most demanding in all of breast imaging and excellent technique, and interpretive experience is of the utmost importance. The high prevalence of benign enhancing lesions observed in young women at high risk is challenging for the radiologist and the “atypical’ appearance of many BRCA-associated breast cancers may be mistaken for benign lesions. There is a deep learning curve for interpretation of these studies, and double reading can be helpful for an observer new to screening interpretation. Joint reading of screening studies may benefit a new reader until confidence is established.
The primary difference in interpreting an AB-MR study compared to a full MR diagnostic study is that only one, or at the most two, postcontrast series is obtained. If ultrafast (UF) technique is used during the first postcontrast minute time point, then a second standard acquisition may be used in the second time point, allowing two postcontrast acquisitions but keeping to the 10-minute time limit. The delayed phase of enhancement is not acquired in an abbreviated study, thus standard kinetic analysis cannot be used as an interpretation tool. Small cancers are visible on AB-MR in the initial enhancement phase; however, there are no set criteria yet for defining the actual threshold percentage increase in enhancement. Invasive cancers usually exhibit early enhancement of 50% or more, and additional measures of the enhancement slope and quantitative measures derived from UF techniques are under development.
Although investigations have shown that there is considerable overlap in the kinetic patterns of benign and malignant lesions on standard diagnostic examinations, invasive cancers classically demonstrate early and intense enhancement in the initial phase. 25 , 26 , 27 Benign masses exhibiting rapid enhancement include lymph nodes, papillomata, and fibroadenomata in premenopausal women. Interpretation should therefore be based primarily on the morphology and internal composition of suspect lesions. Invasive cancers that present as masses are generally small in size (< 1 cm) when visible only on MRI, whereas noninvasive cancers presenting as nonmass enhancement (NME) may be small or large and still remain occult on mammography (MG) and ultrasound (US). Careful morphological analysis is critical for maintaining high sensitivity and specificity with AB-MR.
4.6.1 Assessment of Fibroglandular Tissue
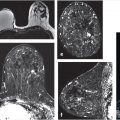
The second edition of the MRI Breast Imaging Reporting and Data System (BIRADS) Atlas requires that a description of the amount of breast fibroglandular tissue (FGT) is included in every MR report. 16 The amount of FGT is considered to be the amount of normal breast parenchyma, as it relates to the amount of normal fatty tissue within the breast and is similar to the mammographic assessment of breast density. FGT is best assessed on a non–fat-saturated T1w or T2w image, or alternatively, the fat-saturated T1w images in the dynamic sequence may be used. The amount of FGT is classified into four categories: (1) almost entirely fat, (2) scattered FGT, (3) heterogeneous FGT, and (4) extreme FGT, based on the ratio of fat and parenchyma (Fig. 4‑2).

4.6.2 Background Parenchymal Enhancement
The accuracy of MR interpretation may be affected by the variable enhancement of normal breast tissue, which usually enhances slowly initially, generates a persistent kinetic curve, and is referred to as BPE. The hormonal response of breast tissue in premenopausal women varies during the menstrual cycle. Marked BPE can limit the sensitivity of the examination because enhancing abnormalities maybe less conspicuous and present a diagnostic challenge. For this reason, it is recommended that premenopausal women should be scanned ideally during the second week of the menstrual cycle and week 4 should be avoided if at all possible. The BI-RADS Atlas recommends that each breast MRI report includes a description of the level of BPE. BPE is considered to be the amount and degree of enhancement of the normal breast parenchyma according to approximate quartiles, and is classified as (1) minimal (< 25% of glandular tissue demonstrating enhancement), (2) mild (25–50% enhancement), (3) moderate (50–75% enhancement), or (4) marked (> 75% enhancement), and symmetric or asymmetric enhancement (Fig. 4‑3).

It is important to note that the degree of BPE does not correlate with the amount of FGT. Glandular tissue consists of hormonally influenced terminal ductal lobular units (TDLUs), which enhance on MRI and can be distinguished from surrounding nonenhancing parenchymal fibrosis. A very dense breast exhibiting extreme FGT may exhibit minimal BPE, whereas a fatty breast composed of scattered FGT may show moderate or marked BPE. 28 Early reports studying the relationship between cancer risk and BPE suggest that BPE is a risk marker, the odds of breast cancer risk increasing with increasing levels of BPE. 29
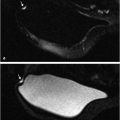
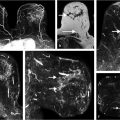
BPE usually exhibits bilateral scattered foci of enhancement (Fig. 4‑4) often with symmetric preferential enhancement located at the periphery of the breast parenchyma seen at the fibroglandular/fat junction (Fig. 4‑5). The arterial blood supply to the breast is primarily from the periphery of the breast tissue from perforating branches of the internal thoracic artery (internal mammary) and branches of the lateral thoracic artery and lateral cutaneous branches of the intercostal arteries. This pattern of vascular inflow accounts for the preferential visualization of BPE at the periphery of the breast tissue cone with later central and retroareolar enhancement. BPE usually demonstrates slow early initial enhancement and progressive delayed enhancement kinetics. 30 BPE may exhibit asymmetric, regional, or focal enhancement, and unilateral asymmetric focal BPE may prompt biopsy in some cases (Fig. 4‑6). Patients treated with BCS often exhibit diminished enhancement in the posttreated breast due to radiation change and fibrosis (Fig. 4‑7, Fig. 4‑8). A report has also shown similar findings in women treated with aromatase inhibitors. 31 Bilateral similar areas of enhancement, regardless of their distribution, are more characteristic of benign enhancement than of malignancy (Fig. 4‑9). BPE increases in postmenopausal women undergoing hormone replacement therapy. 32 , 33 Marked increase in BPE has been shown in lactating women; however, the findings usually resolve completely in the postlactation phase (Fig. 4‑10). Viewing of the source and subtraction MIP images generated from the pre- and first postcontrast series is a good way of providing an overview of enhancement for both breasts and for assessment of the level of BPE.







Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree