7 SPECT and SPECT/CT in Neoplastic Disease
7.1 Introduction
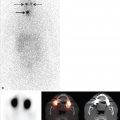
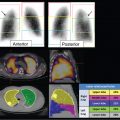
Planar imaging has been used for decades to detect sites of malignant disease, to plan therapy, and to assess response to treatment. More recently, multiplanar, multimodality imaging including singlephoton emission computed tomography (SPECT), computed tomography (CT), magnetic resonance imaging (MRI), and hybrid imaging, such as SPECT/ CT, have entered the realm of routine clinical care. SPECT improves the sensitivity and specificity of oncological planar scintigraphy studies and provides anatomical localization by generating multiplanar images. SPECT/CT can be even more helpful than SPECT alone since it combines the advantages of functional with anatomical imaging, improving anatomical localization of radiotracer uptake, and the characterization of physiological versus pathological sites of radiotracer uptake. The addition of SPECT and/or SPECT/CT to planar imaging tends to decrease the incidence of false-negative studies, increase reader confidence, and improve the concordance of intra- and interobserver interpretation of the findings. More recently, SPECT and SPECT/ CT are being performed for preoperative imaging to guide biopsy and treatment, and are considered tools for quantitative measurements of tumor burden at baseline and following therapy. ► Fig. 7.1 and ► Fig. 7.2 illustrate the advantages of SPECT and its correlation with CT in the evaluation of a man with prostate cancer and lower back pain.


The application of SPECT and SPECT/CT to neoplastic disease has been discussed throughout several chapters in this book. In this chapter, we focus on SPECT and SPECT/CT in the context of lymphoscintigraphy, neuroendocrine tumors, scintimammography, and oncological therapy.
7.2 Lymphoscintigraphy in Oncology Patients
Lymphoscintigraphy is commonly performed in cancer patients to localize sentinel lymph nodes (SLNs) at the time of staging. The concept of the SLN was first proposed by Cabanas in 1977, who suggested the existence of a specific lymph node that appeared to be the primary site of metastasis in 100 men with penile carcinoma. 1 Specifically, SLN is the term used to describe the first lymph node to drain a site of malignancy. Through the years, several methods have been used to identify the SLN, including, among others, contrast lymphangiography, injection of isosulfan blue dye, and lymphoscintigraphy.
Lymphoscintigraphy may also be helpful in the evaluation of postoperative complications in oncology patients, such as lymphedema, lymphoceles (► Fig. 11.10 in Chapter 11 illustrates the value of SPECT/CT for the evaluation of a lymphocele), and the presence of a chyle leak, as well as for the characterization of primary lymphedema. Today, lymphoscintigraphy is performed to inform therapy and reduce morbidity in patients with cancer. 2 , 3
7.2.1 Technique
Lymphoscintigraphy is used to identify lymphatic drainage pathways by planar and/or tomographic imaging following radiotracer administration.
Lymphoscintigraphy is easy to perform and is safe and inexpensive, and it may be helpful in staging several cancer types, most often breast cancer and melanoma, and less commonly cervical, vulvar, and endometrial cancers.
The radiotracers used include technetium-99m (99mTc)-sulfur colloid, 99mTc-antimony trisulphide, and 99mTc-rhenium sulphide, among others. 4 In North America, 99mTc-sulfur colloid is the most common.
The particle size of 99mTc-sulfur colloid has a range of 0.1 to 1 pm. 4 Since 0.1-pm particles show rapid clearance from the interstitial space into the lymphatic channels with low capillary penetration, the radiotracer is commonly passed through a 0.22-pm filter prior to administration.
No special patient preparation is necessary, and pregnancy is not a contraindication. It is generally recommended that breast-feeding should be suspended for 24 hours after radiopharmaceutical administration.
Sentinel Node Studies
The time needed to complete the procedure is generally less than 45 minutes and may be performed 30 minutes to several hours prior to surgery, although often it is done on the morning of or the afternoon prior to surgery. There is considerable variability in technique between centers and cancer types. In general, if the study is performed on the same day as surgery, a total amount of 15 to 30 MBq filtered 99mTc-sulfur colloid is divided into 0.5-mL volume injections. If the study is performed the day before surgery, up to 50 to 150 MBq may be used. Typically, one to four injections are administered symmetrically, intradermally about the areola in patients with breast cancer and adjacent to the tumor/excision site in patients with melanoma, penile, or vulvar cancer. Early dynamic images and delayed static images are obtained with imaging tailored to the indication/tumor type. A study by Uren et al of 209 patients with cutaneous melanoma of the trunk showed that more than 10% of patients had drainage to several lymph node groups, frequently on both sides of the midline. 5 Thus, in patients with trunk melanoma, imaging of both the right and left axillae and the groin is recommended. In patients with breast cancer, the breast that is injected and the ipsilateral axilla are imaged. During surgery, an intraoperative probe is used to identify the sentinel node as well as additional lymph nodes with radiotracer accumulation.
Other Indications (such as Lymphedema)
Two intradermal injections each of 0.05 mL of 18.5-MBq filtered 99mTc-sulfur colloid are given in the dorsum of the hands or feet. Static images of the extremities are obtained within 15 minutes of radiotracer administration, and delayed images are taken at multiple time intervals up to 4 hours following radiotracer injection. The imaging field of view should extend to include the liver.
7.2.2 Advantages of Including SPECT/CT in Sentinel Lymph Node Studies
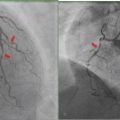
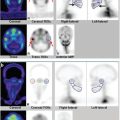
SPECT/CT improves the sensitivity and anatomical localization of planar lymphoscintigraphy for SLN detection. ► Fig. 7.3 shows a patient who underwent preoperative lymphoscintigraphy for a scalp melanoma. Planar images show radiotracer draining to the neck. SPECT/CT images show the anatomical location of several radioactive lymph nodes. A review of the literature by Hoogendam et al suggested that SPECT/CT improved SLN detection in patients with cervical cancer, 6 and a review by Vercellino et al indicated that SPECT/CT improved SLN detection in patients with breast cancer. 7 Mücke et al reported that SPECT/CT identified more lymph nodes than planar lymphoscintigraphy in patients with endometrial carcinoma. 8
The detection of multiple lymph nodes on SPECT/CT has been correlated with an increased likelihood of metastases. 9
The addition of preoperative SPECT/CT compared with SLN excision alone has been shown to be associated with a higher rate of disease-free survival. 10
Exact anatomical and three-dimensional localization on SPECT/CT may increase preoperative surgical confidence and may alter surgical approach and patient management (► Fig. 7.4). 11 , 12
Preoperative SPECT/CT in patients may be cost-effective compared with planar lymphoscintigraphy as a result of reduced operative time and hospital stay, among other things. 13 , 14 In addition, the use of SPECT/CT in patients with head and neck cutaneous malignancies resulted in better postoperative aesthetic results and lower morbidity. 14


Other Indications
Although SPECT/CT is most commonly performed for the localization of SLN in patients with malignant disease prior to therapy, sometimes it may be helpful for the evaluation of postoperative complications or for the characterization of suspected primary lymphedema.
SPECT/CT allows more accurate identification of a chyle leakage site following surgery 15 , 16 and provides additional information about the presence of dermal backflow, the anatomical extent of the lymphatic disorder, and the detection of lymphatic vessels. 17
7.2.3 Pearls
SPECT/CT is incorporated into lymphoscintigraphy studies for several reasons: (1) it improves the sensitivity and anatomical localization of SLNs; (2) it provides additional information about lymphatic vessels, lymphatic leakage sites, dermal backflow, and the extent of the lymphatic disorder; and (3) it is associated with higher preoperative surgical confidence, better postoperative aesthetic results, lower morbidity, and a higher rate of disease-free survival.
7.3 Neuroendocrine Neoplastic Disease
Pheochromocytoma, paraganglioma, and neuroblastoma are among the most common neuroendocrine tumors, and scintigraphy is frequently requested for their evaluation. However, the family of neuroendocrine tumors is large and includes entities such as pituitary adenoma, small cell lung cancer, carcinoid, pancreatic islet cell neoplasm, and medullary thyroid carcinoma, among others. Although symptoms of endocrine hyperfunction may be clinically apparent, often the disease is incidentally detected at the time of cross-sectional imaging for another indication. Functional imaging can be helpful to assess disease burden and plan therapy. It is commonly done using an 111In-labeled somatostatin analogue, such as 111In-pen-tetreotide and/or radioactive iodine (123Ior131I) labeled metaiodobenzylguanidine (MIBG).
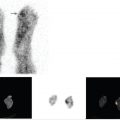
SPECT or SPECT/CT is usually performed in addition to planar imaging to improve the sensitivity and specificity of the technique and to provide anatomical correlation for the functional imaging findings. SPECT/CT is particularly helpful in detecting and localizing small lesions that may not be seen on planar imaging and in differentiating tumor uptake from physiological radiotracer activity. 18 In a study of 72 patients evaluated with 111In-pentetreotide for suspected neuroendocrine disease, the addition of SPECT/CT improved disease detection and localization, resulting in a change in image interpretation in 32% of cases and a change in management in 14% of patients. 19 In a study of 54 patients with neuroendocrine tumors, the addition of SPECT/CT improved the specificity of image interpretation and resulted in a change in management in 28% of patients. 20 ► Fig. 7.5 illustrates the benefit of SPECT/CT in the localization of a pancreatic neuroendocrine tumor.

7.3.1 Somatostatin Receptor Imaging
Somatostatin is a naturally occurring neuropolypeptide. Although endogenous somatostatin has a biological half-life of a few minutes, synthetic analogues have been designed with longer half-lives and improved imaging characteristics. Today, 111In-labeled OctreoScan (pentetreotide) is the most common radioactive somatostatin analogue probe used in clinical practice. It has greater stability than previously synthesized somatostatin analogues and has high renal excretion and limited gastrointestinal activity. 21
There are several somatostatin receptor subtypes, and 111In-pentetreotide preferentially binds to subtypes II and V.
Most neuroendocrine tumors express somatostatin receptors, and the administration of radiolabeled somatostatin analogues followed by planar imaging, SPECT, and/or SPECT/CT can be helpful in detecting the site of primary disease and determining overall disease burden.
111In-pentetreotide-positive disease suggests improved symptom control with cold octreotide. Radionuclide therapy using 131I- or 90Y-labeled octreotide has been used with limited success.
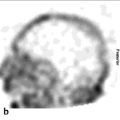
111In-pentetreotide uptake is not specific to neuroendocrine tumors, and the intensity of radiotracer uptake does not characterize tumor histology. Nonneuroendocrine neoplastic diseases can express somatostatin receptors as well. For example, meningiomas (► Fig. 7.6), gliomas, lymphoma, breast cancer, and renal cell carcinoma show variable radiolabeled somatostatin analogue uptake, depending on the concentration of somatostatin receptors present. Nonneoplastic processes may also express somatostatin receptors, such as infection (e.g., tuberculosis) or inflammation (e.g., sarcoidosis and inflammatory bowel disease).

Technique
Prior to imaging, patients should be hydrated and may be given a laxative to decrease bowel activity. Furthermore, patients are often on a cold somatostatin analogue since somatostatin receptor binding has an inhibitory effect on hormone secretion, which can result in improved symptom control. Cold somatostatin analogue should be discontinued prior to imaging, for which consultation with the referring clinician is suggested.
Intravenously, 111 to 222 MBq 111In-pentetreo-tide is administered. When SPECT or SPECT/CT is performed, generally a higher administered activity is preferred (~ 222 MBq) to improve image quality.
Planar whole-body images and SPECT (or SPECT/ CT) are acquired at 24 hours. Imaging at 4 hours is optional, and imaging at 48 hours may be obtained for clarification of sites of equivocal activity. 22
The normal biodistribution of radiotracer includes activity in the blood pool, thyroid tissue, kidneys, spleen, liver, gallbladder, and gastrointestinal and genitourinary tracts. Activity in the gastrointestinal and genitourinary tracts tends to change over time. Radiotracer uptake at sites of disease is variable and depends on the tumor somatostatin receptors subtype expressed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree