9 SPECT/CT for Infection and Inflammation
9.1 Introduction
The detection and localization of inflammation and infection with nuclear medicine techniques have been studied for nearly half a century. A major advantage of radionuclide, or functional, imaging tests compared to anatomical imaging tests is the ability to provide information early in the course of a disease process, that is, prior to the development of anatomical or structural changes. Functional imaging unfortunately does not always provide the anatomical detail necessary to differentiate physiological from pathological processes. This limitation has been largely overcome with the introduction of hybrid imaging, which is redefining the diagnostic workup of patients with suspected or known infection and inflammation. In addition to improving diagnostic accuracy, hybrid imaging also affects patient management. This chapter reviews the role of single-photon emission computed tomography/computed tomography (SPECT/CT) in inflammation and infection.
9.2 Procedures
9.2.1 Bone Scintigraphy
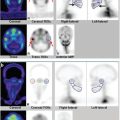
Bone scintigraphy is performed with technetium-99 m (99mTc)-labeled diphosphonates. Radiophar maceutical uptake depends on blood flow and the rate of new bone formation. The procedure is usually performed as a three-phase bone scan for suspected osteomyelitis: the flow, or perfusion, phase acquired immediately after radiopharmaceutical injection, followed immediately by the blood pool, or soft tissue, phase. The third, or bone, phase is performed 2 to 4 hours later. On three-phase bone scintigraphy, osteomyelitis typically presents as focal hyperperfusion, focal hyperemia, and focal bone uptake (► Fig. 9.1). It is important to remember that abnormalities on bone scintigraphy reflect the rate of new bone formation in general, not infection specifically. 1

Advantages
Widely available.
Relatively inexpensive.
Rapidly completed (2–4 hours).
Extremely sensitive: becomes positive within 2 days after onset of symptoms.
Accuracy exceeds 90% in the setting of unviolated bone.
Disadvantage
Decreased specificity in the presence of underlying bony conditions (fracture, orthopaedic hard ware, neuropathic joint, tumor, and so on).
9.2.2 Gallium-67 Imaging
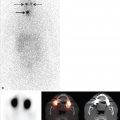
Gallium-67 (67Ga) has a half-life of 78 hours and emits gamma radiation with four energy peaks suitable for imaging: 93 keV (40%), 184 keV (24%), 296 keV (22%), and 388 keV (7%). By 24 hours after injection, 10 to 25% of the administered dose is excreted via the kidneys. Beyond 24 hours, the colon is the principal excretory pathway. At 48 to 72 hours after injection, when imaging is usually performed, about 75% of the injected dose remains in the body, distributed among soft tissues, liver, and bone/bone marrow (► Fig. 9.2). This “normal” distribution, however, is subject to considerable variation. Nasopharyngeal and lacrimal gland activity can be prominent. Breast uptake can be intense in pregnant and lactating women as well as in other hyperprolactinemic states. Normally, healing surgical incisions concentrate 67Ga for variable time periods. The biodistribution can be altered by blood transfusions and the magnetic resonance imaging (MRI) contrast agent gadolinium, with increased skeletal and urinary tract and decreased hepatic and colonic activity. 2

67Ga uptake in inflammation and infection likely depends on several factors. Approximately 99% of circulating gallium is in the plasma, nearly all transferrin bound. The increased blood flow and vascular membrane permeability associated with inflammation result in increased 67Ga delivery and accumulation at inflammatory sites. Lactoferrin, another plasma protein, is present in high concentrations in inflammatory exudates. 67Ga is presumably transported via transferrin to inflammatory foci, where it dissociates from transferrin and complexes with lactoferrin. Bacteria are also involved in 67Ga uptake in infection, probably through nonspecific binding and facilitated diffusion. Small-molecule metal chelates known as siderophores, which are produced by bac teria, are 67Ga-avid. The siderophore–67Ga complex is transported into the bacterium, from which it can not be released without destruction of the entire molecule. Some 67Ga may be bound to leukocytes and transported by leukocytes to inflammatory foci, but this is probably a relatively unimportant mechanism. There are ample data confirming 67Ga accumulation in infection, even in the absence of circulating white cells.2
Advantages
Detects inflammation and infection.
Sensitive in immunocompromised individuals.
Disadvantages
Nonspecific accumulation in tumor, at sites of recent surgery, and at sites of new bone formation.
A delay of 24 to 72 hours between injection and imaging.
Variable biodistribution.
Low-resolution images.
9.2.3 In Vitro Labeled Leukocyte Imaging
In vitro leukocyte (white blood cell [WBC]) labeling is most often performed with either 111In-oxyquinolone or 99mTc-exametazime (HMPAO). Labeled leukocyte uptake depends on intact chemotaxis, which is not an issue with routine in vitro labeling procedures, the number and types of cells labeled, and the cellular component of a particular inflammatory response. A total circulating WBC count of at least 2,000/u is needed to obtain satisfactory images. Usually, the majority of leukocytes labeled are neutrophils, and the procedure is most sensitive for identifying neutrophil-mediated inflammatory processes, such as bacterial infections. WBC imaging is less useful for those illnesses in which the predominant cellular response is not neutrophilic, that is, sarcoidosis. 3
Regardless of whether the white cells are labeled with 111In or 99mTc, images obtained shortly after injection are characterized by intense pulmonary activity. This activity, which clears rap idly and reaches background levels within 4 hours after injection, is probably due to leukocyte activation during labeling, which impedes cellular movement through the pulmonary vascular bed, slowing passage through the lungs.3
111In-WBC
The usual adult administered activity is 10 to 20 MBq (0.3–0.5 mCi). Imaging is typically performed 18 to 30 hours after injection. The advantages of using 111In as the radiolabel include label stability and, at 24 hours postinjection, a normal distribution of activity limited to the liver, spleen, and bone marrow (► Fig. 9.3). The 67-hour half-life of 111In allows for delayed imaging. Patients with musculoskeletal infection may need to undergo bone marrow scintigraphy, which can be performed while the patient’s cells are being labeled, as part of a simultaneous dual-isotope acquisition with 111In-WBC imaging, or after completion of the 111In-WBC study. Disadvantages include suboptimal photon energies, low-resolution images, and the 18- to 30-hour interval between injection and imaging.3

99mTc-WBC
The usual administered activity is 185 to 370 MBq (5–10 mCi). The normal biodistribution of 99mTc-WBC is more variable than that of 111In-WBC. In addition to the reticuloendothelial system, activity is normally present in the urinary tract, large bow el (within 4 hours after injection), blood pool, and occasionally the gallbladder (► Fig. 9.4). The time interval between injection and imaging varies with the indication; imaging is usually performed within a few hours after injection.3

Advantages of using 99mTc as the radiolabel include a photon energy that is optimal for imaging using current instrumentation, a high photon flux, and the ability to detect abnormalities within a few hours after injection. This is especially important when performing SPECT/CT. Disadvantages include urinary tract activity, which appears shortly after injection, and bowel activity, which appears by 4 hours after injection. The instability of the label and short half-life of 99mTc are disadvantages when delayed 24-hour imaging is needed. Simultaneous dual-isotope imaging, of course, is not possible, and when bone or bone marrow imaging is necessary, an interval of 48 to 72 hours is required between the two tests.3
White Blood Cell/Marrow Imaging
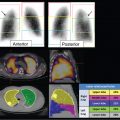
WBC imaging, which is the radionuclide test of choice for diagnosing most cases of complicating osteomyelitis, must often be performed in conjunction with bone marrow imaging to maximize accuracy. This is because leukocytes accumulate in both infection and in the bone marrow. The normal distribution of hematopoietically active bone marrow in adults is very variable. Systemic diseases cause generalized alterations in marrow distribution, while fractures, orthopaedic hardware, neuropathic joints, trauma, and heterotopic bone cause localized alterations. In children, the normal distribution of hematopoietically active marrow varies with age. It is not always possible to determine if activity on a WBC image indicates infection or bone marrow. This difficulty can be overcome by performing 99mTc-sulfur colloid bone marrow (marrow) imaging. Leukocytes and sulfur colloid both accumulate in bone marrow; leukocytes also accumulate in infection, while sulfur colloid does not. WBC/marrow imaging is positive for infection when activity is present on the WBC image without corresponding activity on the marrow image (► Fig. 9.5, ► Fig. 9.6). The overall accuracy of WBC/marrow imaging is approximately 90%. 4


Advantages of White Blood Cell Imaging
Specific for neutrophil-mediated processes, such as bacterial infections.
Accurately diagnoses complicating osteomyelitis.
Disadvantages of White Blood Cell Imaging
Labor-intensive labeling process.
Not always available.
Usually needs to be combined with bone marrow imaging for musculoskeletal infection.
Difficult to perform in leukopenic patients and children.
9.2.4 In Vivo Labeled Leukocytes
In vivo techniques for WBC labeling using antigranulocyte antibodies and antibody fragments have been explored as alternatives to the in vitro labeling procedure. Besilesomab is a murine monoclonal G1 immunoglobulin that binds to the normal cross-reactive antigen (NCA)-95 antigen on leukocytes. About 10% of the injected activity is neutrophil bound at 45 minutes after injection; 20% of the activity circulates freely in the blood. Studies usually become positive by 6 hours after injection, although delayed imaging may increase lesion detection. Up to 40% of the injected dose accumulates in the bone marrow, which can obscure small foci of infection.1 Besilesomab incites a human antimurine antibody (HAMA) response in up to 30% of individuals receiving this agent. Patients should be prescreened for HAMA. A positive result is a contraindication to the procedure. Because of immunogenicity concerns, patients should not undergo repeat studies with this agent. 5
Antibody fragments do not induce a HAMA response and are a potential alternative to whole antibodies. Sulesomab is a 50-kD fragment antigen binding (Fab′) portion of a murine monoclonal IgG1 class antibody that binds to NCA-90 present on leukocytes. NCA-90 is also present on the macrophage–monocyte cell lineage, in normal colonic mucosa, and in colonic adenocarcinoma. The exact uptake mechanisms of sulesomab are somewhat controversial. Some data suggest that sulesomab not only binds to circulating neutrophils, which then migrate to foci of infection, but also crosses permeable capillary membranes and binds to leukocytes already present at sites of infection. Other data, however, suggest that sulesomab does not bind to circulating leukocytes; rather, it accumulates nonspecifically through increased capillary membrane permeability.5
9.2.5 Other Radiopharmaceuticals
Radiolabeled synthetic fragments of ubiquicidin, a human antimicrobial peptide that targets bacteria, possess the ability to differentiate infection from sterile inflammation and may be useful for monitoring the efficacy of antibacterial agents in certain infections.5
Biotin is used as a growth factor by certain bacteria. 111In-labeled biotin has been used in the evaluation of patients with suspected spinal infections.5
9.3 Specific Indications
Adding SPECT/CT to conventional scintigraphic imaging of infection and inflammation increases reader confidence and improves accuracy by increasing the specificity of the test and better defining the location and extent of the infection (► Fig. 9.7). SPECT/CT has proven to be especially useful in the evaluation of cardiovascular and musculoskeletal infections.

9.3.1 Cardiovascular Infections
Infective Endocarditis
Imaging plays a key role in the diagnosis of infective endocarditis (IE). Echocardiography is useful for diagnosis, risk stratification, prognosis, guiding treatment (medical vs. surgical), and monitoring re sponse to treatment. The major echocardiographic criteria for IE are vegetation and abscess. The sensitivity for diagnosis of vegetations is 75% for transthoracic echocardiography (TTE) and 85 to 90% for transesophageal echocardiography (TEE). Abscess is the second most typical finding associated with IE. The sensitivities of TTE and TEE for detecting abscesses are about 50 and 90%, respectively. Although echocardiography is the primary imaging modality used to diagnose IE, there are limitations to the test. Atypical findings are frequent, and the test may be false-negative in up to 15% of patients in whom there are preexisting conditions, such as mitral valve prolapse, and in the presence of prosthetic valves. 6
Until recently, the use of radionuclide imaging in suspected IE was limited. The development of hybrid imaging has stimulated renewed interest in radionuclide imaging for diagnosing IE and its complications. Although most investigations have focused on fluorine-18 fluorodeoxyglucose (18F-FDG) positron emission tomography/CT (PET/CT), labeled leukocyte SPECT/CT shows considerable promise. Erba et al 7 assessed the value of 99mTc-WBC scintigraphy including SPECT/CT in 131 consecutive patients with suspected IE. Results were correlated with TTE or TEE, blood cultures, and the Duke criteria. These investigators reported that 99mTc-WBC SPECT/CT was 96% (46/51) sensitive and 100% specific (80/80) for IE. Extracardiac uptake of 99mTc-WBC was present in nearly half of the patients with IE. 99mTc-WBC SPECT/CT was useful in patients with possible IE by the Duke criteria. The scan was especially valuable in patients with negative or difficult-to-interpret echocardiograms. Three patients with marantic vegetations who had false-positive echocardiograms were true negative on 99mTc-WBC SPECT/CT. These investigators concluded that 99mTc-WBC SPECT/CT reduces the rate of misdiagnosis of IE when combined with standard diagnostic tests when clinical suspicion is high but echocardiography is inconclusive; when there is a need to differentiate between septic and sterile vegetations detected on echocardiography; when echocardiographic, laboratory, and clinical data are contradictory, and when valve involvement (especially prosthetic valves) needs to be excluded during febrile episodes, sepsis, or postoperative infections.
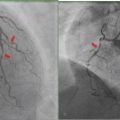
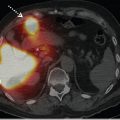
IE is a serious complication of valve replacement that occurs in up to 6% of patients with prosthetic valves. The diagnosis of IE by echocardiography is more difficult when a prosthetic valve is involved than when a native valve is involved. The Duke criteria are less helpful in prosthetic valve endocarditis (PVE) because of lower sensitivity. TEE is mandatory in the evaluation of suspected PVE because of its higher sensitivity and specificity for the detection of vegetations, abscesses, and peri-valvular lesions in patients with prosthetic valves.6 Hyafil et al 8 studied 42 patients with suspected PVE and reported that patient management was affected by the results of 99mTc-WBC SPECT/CT in 12 (29%) patients (► Fig. 9.8).

The development of severe prosthetic dehiscence requiring cardiac surgery was closely related to the extent of perivalvular infection, and in this investigation, 99mTc-WBC SPECT/CT was more accurate than echocardiography for delineating the extent of infection in perivalvular regions. These investigators also found that intensity of 99mTc-WBC uptake might be useful to distinguish between those patients needing surgery and those for whom medical therapy is sufficient. They concluded that 99mTc-WBC SPECT/CT is useful in patients with suspected PVE and inconclusive TEE.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree