Advances in the treatment of pediatric abdominopelvic malignancies have increased survival drastically. Imaging is critical in initial tumor characterization/staging, assessment of treatment response, and surveillance following therapy. Magnetic resonance imaging (MRI) is playing an increasing role in the care of these patients due to its lack of ionizing radiation, superior contrast resolution and the ability to characterize tumors based on tissue characteristics (e.g., T1 and T2 relaxation times). Modern MR techniques also allow for assessment of tumors based on functional characteristics. This article is focused on emerging MRI technologies and potential applications in the imaging of pediatric abdominopelvic malignancies.
Key points
- •
The use of magnetic resonance imaging (MRI) in the setting of pediatric abdominopelvic malignancy is increasing because of concerns regarding ionizing radiation exposure, superior contrast resolution compared with computed tomography (CT), and recent technical advances.
- •
Many ongoing oncologic clinical trials now incorporate MR imaging in their protocols as an alternative to CT imaging.
- •
Advanced MR imaging techniques, such as diffusion-weighted imaging, dynamic contrast–enhanced imaging (MR imaging perfusion), and 18 F-fluorodeoxyglucose positron emission tomography MR imaging, provide functional assessment of abdominopelvic tumors in addition to morphologic information.
- •
Whole-body (WB) MR imaging has been shown to be useful in staging and surveillance of pediatric malignancy without the use of ionizing radiation, as is required for other currently used WB imaging techniques. The major disadvantage of WB-MR imaging is its lack of specificity compared with more targeted imaging strategies, such as metaiodobenzylguanidine (MIBG) scanning in neuroblastoma.
- •
Major weaknesses of MR imaging in pediatric oncologic imaging include difficulty evaluating the lung parenchyma for metastatic disease and detection of calcification within tumors.
Introduction
Magnetic resonance imaging (MRI) is firmly established as a useful imaging technique in pediatric patients. However, MRI use has lagged behind the use of computed tomography (CT) in pediatric abdominopelvic oncology because of a variety of factors, including issues related to cancer research protocols, concerns about the sensitivity of MR imaging, and a general lack of awareness among clinical providers as to the feasibility and advantages of current MR imaging techniques. As abdominopelvic MRI has become more widely available, pulse sequences have become faster and more robust, and image quality has improved, the use of MRI in pediatric oncologic imaging has increased. This growth in MRI use in most instances has been as an alternative to CT imaging for morphologic evaluation, although recent technologic advances now allow functional assessment of abdominopelvic tumors as well.
This article focuses on advanced MRI techniques currently being clinically used or investigated in pediatric abdominopelvic oncologic imaging. A range of different techniques, including diffusion-weighted imaging (DWI), dynamic contrast-enhanced (DCE) MRI, whole-body (WB) MR imaging, and 18 F-fluorodeoxyglucose ( 18 F-FDG) positron emission tomography MR (PET-MRI) imaging, are discussed. In addition, the increasing acceptance of MRI in pediatric cancer research protocols and a brief discussion of specific issues related to performing MR imaging in children, such as sedation, are addressed.
Introduction
Magnetic resonance imaging (MRI) is firmly established as a useful imaging technique in pediatric patients. However, MRI use has lagged behind the use of computed tomography (CT) in pediatric abdominopelvic oncology because of a variety of factors, including issues related to cancer research protocols, concerns about the sensitivity of MR imaging, and a general lack of awareness among clinical providers as to the feasibility and advantages of current MR imaging techniques. As abdominopelvic MRI has become more widely available, pulse sequences have become faster and more robust, and image quality has improved, the use of MRI in pediatric oncologic imaging has increased. This growth in MRI use in most instances has been as an alternative to CT imaging for morphologic evaluation, although recent technologic advances now allow functional assessment of abdominopelvic tumors as well.
This article focuses on advanced MRI techniques currently being clinically used or investigated in pediatric abdominopelvic oncologic imaging. A range of different techniques, including diffusion-weighted imaging (DWI), dynamic contrast-enhanced (DCE) MRI, whole-body (WB) MR imaging, and 18 F-fluorodeoxyglucose ( 18 F-FDG) positron emission tomography MR (PET-MRI) imaging, are discussed. In addition, the increasing acceptance of MRI in pediatric cancer research protocols and a brief discussion of specific issues related to performing MR imaging in children, such as sedation, are addressed.
Protocol and patient issues
A standardized, but flexible, MRI protocol for abdominopelvic tumor imaging is important for success in pediatric oncology imaging. Standardization of the protocol, including standardized pulse sequence parameters and imaging planes, allows for signal intensity and size measurements that can be used for characterization of the lesion(s) at the time of initial diagnosis and for assessing treatment response on follow-up imaging. A large number of pediatric oncology patients, especially those with solid tumors, are formally treated and followed after therapy using specific protocols, often through the Children’s Oncology Group (COG). Having a standardized, reproducible protocol is important in these patients because imaging findings commonly determine patient eligibility for clinical trials and changes in lesions over time directly affect patient treatment and follow-up decisions. Fortunately, most COG protocols are straightforward and commonly used clinical pediatric abdominopelvic MR imaging protocols often fulfill necessary imaging requirements. Our institutional routine clinical abdominopelvic mass MRI protocol is presented in Table 1 .
| T1W TSE | T2W TSE | T2W SSFSE | T2W TSE | DWI EPI | T1W In-phase FFE | T1W 3D FFE Precontrast | T1W 3D FFE Postcontrast | |
|---|---|---|---|---|---|---|---|---|
| Plane | Coronal | Coronal | Coronal | Axial | Axial | Axial | Axial | 3 planes |
| Slice thickness | 5 | 5 | 5 | 5 | 6 | 5 | 2-3 | 2-3 |
| TR (ms) | 554 | Shortest | Shortest | Shortest | Shortest | Shortest | Shortest | Shortest |
| TE (ms) | 4.6 | 80 | 80 | 80 | Shortest | 4.6 | Shortest | Shortest |
| Respiratory compensation | None | Trigger | Trigger | Trigger | Trigger | Breath hold | Breath hold | Breath hold |
| NSA | 4 | 2 | 1 | 2 | 3 | 1 | 1 | 1 |
| Fat saturation | N | Y | N | Y | Y | N | Y | Y |
| b values | — | — | — | — | 0, 100, 750 | — | — | — |
MRI protocols can be altered in order to optimize evaluation depending on the clinical setting and suspected tumor type. For example, in the setting of a known or suspected primary liver neoplasm, we routinely perform dynamic postcontrast MR imaging through the liver, including arterial phase imaging. In this clinical situation we also routinely administer a hepatocyte-specific contrast agent (gadoxetate disodium; Eovist, Bayer HealthCare, Wayne, NJ) and acquire additional delayed hepatocyte phase imaging at 10 and 20 minutes following injection. Tailoring the MRI protocol to the tumor or organ of interest often provides added information, while maintaining the ability to perform routine assessments of signal intensity and size.
The use of sedation and general anesthesia is commonplace in young children undergoing abdominopelvic MRI for the assessment of suspected or known malignancy, because of several factors. Most young children are unable to remain sufficiently motionless for MRI, because imaging times are commonly in the 30 to 60 minute range. In addition, young children often have difficultly cooperating with breath-holding instructions. The physical appearance of the MRI scanner and the noises produced during imaging may also be frightening to some children, making cooperation even less likely. To this end, the use of ear plugs, noise-cancelling headphones, and distraction devices (eg, music headphones, movie goggles) may be helpful, especially in school-aged children. Preprocedure preparation with play therapy and other strategies have also been shown to be effective in reducing the need for general anesthesia. Imaging studies performed under general anesthesia have been shown to require longer recovery times and are more costly than those performed without general anesthesia. Some studies have shown that repeated exposure to general anesthetics may be associated with negative long-term cognitive effects, although others studies have not found such an association. When deciding between the use of nonsedated CT and MRI performed under sedation or general anesthesia, the advantages and disadvantages of both modalities should be carefully considered, including the potential harmful effects of both ionizing radiation and general anesthesia.
Diffusion-weighted imaging
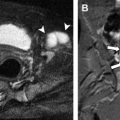
DWI uses the motion of water molecules to provide image contrast and to characterize tissues. The motion of water molecules in a glass of water is random (Brownian motion), whereas in biological tissues this random motion can be impeded (or restricted), primarily by cell membranes. The first widespread clinical use of DWI was in neuroimaging, specifically stroke imaging, because the cellular swelling that accompanies acute infarction (cytotoxic edema) results in restricted water motion and signal hyperintensity. DWI is now being evaluated as an oncologic imaging biomarker for distinguishing benign from malignant lesions and for evaluating response to therapy, including in the setting of pediatric abdominopelvic malignancy. The increased cellularity of many malignant tumors compared with normal tissues impedes the motion of water and presents as restricted diffusion (signal hyperintensity) on DWI. In current clinical practice, DWI sequences (with higher b values) are used for qualitative assessment, because tumors and abnormal lymph nodes are usually hyperintense relative to the background, allowing increased sensitivity ( Fig. 1 ). The degree of restricted diffusion can be quantified by performing DWI with multiple b values and calculating a map of apparent diffusion coefficient (ADC) values ( Fig. 2 ). The number of b values required (eg, 2, 3, 4 or more) as well as their absolute values (0 to greater than 1000 m/s 2 ) have yet to be established for accurate, reproducible ADC calculation in pediatric oncologic imaging.
Humphries and colleagues evaluated ADC values in 19 children with extracranial soft tissue masses. Although they found an inverse relationship between the ADC value and the cellularity of tissues on histopathology, there was no significant difference between benign and malignant lesions, and their final conclusion was that ADC value alone could not accurately distinguish between benign and malignant tumors. Contradictory results were observed in a study performed by Kocaoglu and colleagues, who found that significantly lower ADC values were observed with malignant lesions in a group of 31 pediatric abdominal masses (15 benign, 16 malignant), although there was some overlap between the two groups. Interestingly, these studies used different methods for determining the region of interest for calculation of the mean ADC values. More recently, Gahr and colleagues retrospectively evaluated 19 pediatric neurogenic tumors and found a significant difference between the ADC values of neuroblastoma and ganglioneuroblastoma/ganglioneuroma. Their conclusion was that neuroblastoma could be differentiated from the other less aggressive neurogenic tumors based on ADC values, and that the lower ADC values observed in neuroblastoma were caused by the greater cellularity of this neoplasm.
In a feasibility study involving 7 pediatric oncology patients with 9 tumors, McDonald and colleagues analyzed ADC values before and after completion of chemotherapy. The ADC values of all tumors changed following chemotherapy, with an increase in the median ADC values seen in most lesions. The increased ADC value (indicating less impeded diffusion and less hyperintense signal on DWI images) was thought to represent decreased cellularity of the tumor, thus representing treatment response. The largest increases in ADC value were observed in those tumors that showed the greatest response to therapy on histopathologic evaluation ( Fig. 3 ).