Various methods of cross-sectional imaging are used for visualization of the cranial nerves, relying heavily on MR imaging. The success of the MR imaging sequences for visualization of cranial nerves depends on their anatomic context at the point of evaluation. The heterogeneity of opinion regarding optimal evaluation of the cranial nerves is partly a function of the complexity of cranial nerve anatomy. A variety of approaches are advocated and variations in equipment and terminology cloud the field. This article proposes a segmental classification and corresponding nomenclature for imaging evaluation of the cranial nerves and reviews technical considerations and applicable literature.
Key points
- •
The cranial nerves (CNs) pursue a complex course through tissues with widely varying MR imaging signal characteristics as they extend from brainstem nuclei into the fluid-filled subarachnoid spaces and ultimately pass through the skull base to exit the cranium.
- •
In turn, the reported success of the variety of available MR imaging sequences for visualization of the CNs depends largely on their anatomic context at the point of evaluation.
- •
Consideration of the general segmental architecture of the CNs aids in evaluation of patients with pathologic conditions affecting or adjoining their course.
Introduction
The 12 pairs of cranial nerves (CNs) arise directly from the brain within the cranial vault (with the exception of spinal rootlets of CN XI, which arises from the rostral cervical spine). The CNs serve a variety of highly specialized functions, including those necessary for vision, movement of the eyes and face, and identification and consumption of food. The branching patterns and/or proximity of CNs to each other at points along their course may allow localization of pathology on clinical grounds. MR imaging plays an important role in the localization and identification of pathology as well as presurgical planning. A variety of modalities have been used in the imaging evaluation of CNs. Clinically, the first cross-sectional imaging study to directly demonstrate the CNs was pneumoencephalography. During pneumoencephalography, the introduction of subarachnoid air surrounding the CNs allowed for visualization of the optic, oculomotor, trigeminal, and hypoglossal nerves within the basal cisterns. The advent of CT enabled visualization of the region of the CNs with a greater degree of detail and with injection of intrathecal contrast; the CNs were visualized as linear filling defects within the subarachnoid space. In both pneumoencephalography and CT cisternography, visualization was principally limited to the cisternal/subarachnoid course of the CNs and pathology was implied by alterations in the adjacent osseous structures. With the advent of MR imaging, cross-sectional examination of the structures of the head and neck without ionizing radiation became possible, with the ability to acquire images in any arbitrary plane allowing for the examination to be tailored to the CN in question. MR imaging is now the standard mode of imaging of the CNs and is the focus of this article.
Introduction
The 12 pairs of cranial nerves (CNs) arise directly from the brain within the cranial vault (with the exception of spinal rootlets of CN XI, which arises from the rostral cervical spine). The CNs serve a variety of highly specialized functions, including those necessary for vision, movement of the eyes and face, and identification and consumption of food. The branching patterns and/or proximity of CNs to each other at points along their course may allow localization of pathology on clinical grounds. MR imaging plays an important role in the localization and identification of pathology as well as presurgical planning. A variety of modalities have been used in the imaging evaluation of CNs. Clinically, the first cross-sectional imaging study to directly demonstrate the CNs was pneumoencephalography. During pneumoencephalography, the introduction of subarachnoid air surrounding the CNs allowed for visualization of the optic, oculomotor, trigeminal, and hypoglossal nerves within the basal cisterns. The advent of CT enabled visualization of the region of the CNs with a greater degree of detail and with injection of intrathecal contrast; the CNs were visualized as linear filling defects within the subarachnoid space. In both pneumoencephalography and CT cisternography, visualization was principally limited to the cisternal/subarachnoid course of the CNs and pathology was implied by alterations in the adjacent osseous structures. With the advent of MR imaging, cross-sectional examination of the structures of the head and neck without ionizing radiation became possible, with the ability to acquire images in any arbitrary plane allowing for the examination to be tailored to the CN in question. MR imaging is now the standard mode of imaging of the CNs and is the focus of this article.
Technical considerations for MR imaging acquisition field strength
Fischbach and colleagues studied T2-weighted spin-echo imaging of the CNs at 1.5T and 3T, the two most commonly available field strengths of clinical MR imaging units, and found that images acquired at higher spatial resolution on the 3T scanner nonetheless also had higher clarity and signal-to-noise ratio. The detection of perineural spread of neoplastic disease in the face initially not detected on 1.5T evaluation was possible on repeat examination at 3T. Such results are not generally surprising because the tissue discrimination generally improved with higher field strengths. Although 3T evaluation is generally preferred over 1.5T evaluation, diagnostic images may be obtained at either field strength, in particular when 3T MR imaging is not available, of questionable safety, or otherwise deemed inappropriate.
Voxel size and coverage
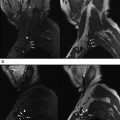
Thin-section imaging significantly improves detection of the CNs although visualization of the cisternal trochlear (CN IV), abducens (CN VI), and accessory (CN XI) nerves may remain challenging. Due to its small caliber and proximity to multiple vascular structures, visualization of CN IV is particularly dependent on the spatial resolution of the sequences acquired. Choi and colleagues compared conventional resolution (0.67 mm × 0.45 mm × 1.4 mm) to high-resolution (0.3 mm × 0.3 mm × 0.25 mm) imaging for detection of the cisternal trochlear nerve and found that the rate at which the nerve could probably or definitely be identified rose significantly from approximately 23% to 100%. Fig. 1 demonstrates visualization of the trochlear nerve on 0.4-mm, 0.5-mm, and 0.6-mm isotropic constructive interference in the steady-state (CISS) images. Although increasing spatial resolution may improve visualization of small structures, the trade-off with respect to length of acquisition, reduced coverage, and/or decreased signal-to-noise ratio renders optimal coverage in all cases difficult. In the authors’ practice, multiple 3-D sequences are typically used and include CISS imaging for the highest spatial resolution acquisition. The typical CISS acquisition includes 0.6-mm isotropic voxels with coverage of the entirety of the posterior fossa, skull base, and upper face. In select cases where CN IV palsy has been clinically diagnosed, a higher spatial resolution is often used.
2-D versus 3-D imaging
Initially, MR imaging tailored to the CNs required careful attention to 2-D slice angulation to best demonstrate the CN in question. Modern MR imaging equipment allows 3-D acquisition from which post hoc reconstruction in multiple planes can be created, often better demonstrating the CNs. One study of the cisternal components of the CNs in the cerebellopontine angle cistern with fast spin-echo technique found that 3-D imaging was superior to 2-D imaging due to suppression of flow artifacts and thinner sections and suggested that MR imaging evaluation of the cisterns be performed with 3-D technique. When isotropic 3-D images are acquired, post hoc reconstructions can be made in any arbitrary plane, which is often useful in evaluating the complex anatomy of the CNs and surrounding structures.
Injection of intravenous contrast agents
The central nervous system (CNS) components of the CNs (including the entirety of the ophthalmic (CN I) and optic (CN II) nerves, which are properly tracts of the CNS rather than nerves per se) are at least partly isolated from the contents of the bloodstream by the blood-brain barrier and do not normally demonstrate visible contrast enhancement. The components of the CNs in the peripheral nervous system (PNS) are likewise separated by the blood-nerve barrier. When there is disruption of the blood-nerve barrier or blood-brain barrier, it is detected by the presence of an increase in intensity on postcontrast MR images with T1 weighting. Perhaps owing to lack of a similar barrier mechanism or increased blood flow, enhancement of the ganglia of the CNs may be detected as a physiologic finding. Additionally, a circumneural arteriovenous plexus surrounds portions of the CNs in the interdural and foraminal regions of the skull base. Enhancement of the arteriovenous plexus may be seen, for instance, in the region of the tympanic and mastoid segments of the facial nerve (CN VII) in addition to the region of the geniculate ganglion. Enhancement in the other CN components is pathologic. When present pathologic enhancement aids in the detection/localization of pathologic conditions of the CNs such as neoplastic, infectious, or inflammatory diseases.
Nomenclature
International consensus on anatomic nomenclature has been established in the Terminologia Anatomica . The accepted terms for the CNs are olfactory nerve (CN I), optic nerve (CN II), oculomotor nerve (CN III), trochlear nerve (CN IV), trigeminal nerve (CN V), abducens or abducent nerve (CN VI), facial nerve (CN VII), vestibulocochlear nerve (CN VIII), glossopharyngeal nerve (CN IX), vagus nerve (CN X), accessory nerve (CN XI), and hypoglossal nerve (CN XII). The system defines multiple named subcomponents of the CNs by function. Structures with a single function and without branches, such as CN VI, receive no further subclassification. The international nomenclature incompletely serves the needs of a radiologist or clinician. For instance, lesions in various locations along the course of CN VI, although presenting with a similar clinical deficit of abduction of the globe, may have vastly differing differential diagnostic and clinical implications. Therefore, the authors propose a systematic method of segmental imaging evaluation of the CNs.
Anatomic segments
After emerging from the brain, each of the CNs courses through the cerebral spinal fluid (CSF) before it traverses the meninges, extending through an associated skull base foramen to emerge into the head and neck. Along this path, the nerves are surrounded by CSF in the subarachnoid space, venous blood in the interdural compartment, bone within the skull base foramina, and various soft tissues after exiting from the skull. Although the anatomic course of each of the CNs is different, some fundamental anatomic considerations are sufficiently similar to allow a systematic classification of different segments, which share similar imaging properties ( Fig. 2 ). General anatomic and imaging considerations for each segment follow. Selected information on pathology is included for illustrative purposes, although a comprehensive description of pathology affecting each segment is beyond the scope of this article.
a. Nuclear Segment
Anatomic considerations
The CN nuclei contain the cell bodies of neurons that either give rise to the efferent fibers, which exit the brainstem or receive afferent input. The CN nuclei extend from the midbrain (CN III) cranially into the rostral cervical spine caudally (CN XI). The afferent nuclei generally rest lateral and dorsal to their efferent counterparts. Nuclei are further separated by their functional and evolutionary relationships from the somatic, branchial, and visceral (autonomic) motor nuclei. The visceral, somatic, and special sensory nuclei are arranged in columns approximately medial to lateral.
Imaging approaches
In the fetal and neonatal brain, MR imaging allows direct identification of several CN nuclei, because the nucleus and proximal fibers myelinate earlier than the surrounding mesencephalic and rhombencephalic white matter tracts, best seen on T2-weighted imaging ( Fig. 3 ). Direct visualization of CN nuclei in the mature brain is difficult, and identification of pathology that has an impact on the nuclei requires knowledge of the location and function of the nuclei. The location of CN II.a, the lateral geniculate nuclei, can be directly seen as a focal protrusion on the posterolateral thalamus. The location of CN VI.a within the dorsal pons can be deduced when the facial colliculus is visualized (a name earned due to the traversing motor fibers of CN VII.b). The locations of components of CN V.a are reported as visible using diffusion tensor imaging (DTI) in the research setting.
Selected pathologic entities
Most acquired pathologic conditions that have an impact on CN nuclei are related to regional abnormalities as opposed to abnormalities specific to the nucleus itself; thus, any process that has an impact on the adjacent brain parenchyma can have an impact on the nucleus of the CNs. Stroke, inflammatory processes, infection, tumor, and metabolic disorders, for instance, can result in nuclear-based cranial neuropathies.
b. Parenchymal Fascicular Segment
Anatomic considerations
The parenchymal fascicular segment runs from the point fibers exit the nuclei through the parenchyma to the point of separation from the brainstem at the apparent origin (AO). With the exception of CN IV.b, which alone exits the brainstem along the dorsal surface (see Fig. 1 ), the parenchymal fascicles of the CNs (also known as the CN tracts) pursue an anterior and lateral course through the brainstem to exit its ventral surface. Before separating from the brainstem to enter the cisternal segment, the CN fascicles may course along the surface of the brainstem. The point at which a discrete fascicle emerges from the parenchyma is known as the root exit point and the fascicular component, which may then course on the surface of the brainstem, is known anatomically as the attached segment (AS). Studies in mammals demonstrate that these emergent fibers largely retain central myelination by oligodendrocytes before separating from the brainstem within the cisternal segment ; there are similar findings in humans.
Imaging approaches
T2-weighted imaging
The normal course of the fascicles of CN fibers extending from the nuclei toward the surface of the brainstem can sometimes be seen (see Fig. 3 B) during the process of myelination in fetal or neonatal life using T2-weighted imaging, because the tightly packed layers of mature myelin around the nerve fibers have a decreased water content compared with the surrounding incompletely myelinated parenchyma. The fascicles are not typically visualized on standard anatomic imaging in patients who are maturely myelinated but may be outlined by pathologic conditions. In the research realm, advances in high-resolution imaging shows promise for more clear depiction of the fascicular segments at higher field strengths than those currently used clinically. Likewise, the root exit point and AS are not readily appreciated on modern clinical imaging and must be inferred by comparison to anatomic references.
Diffusion tensor imaging
DTI is a technique that allows identification of fiber tracts within the brain parenchyma by virtue of the manner in which they constrain water diffusion. The technique has been used with limited success for visualization of the CN fascicles, largely due to their small size. The tracts associated with CN II and CN III.b are visualized with success. In one patient with CN III palsy, abnormal signal in the region of CN III.b not well seen on standard imaging was demonstrated with thin-section DTI. In the research setting, multishot diffusion-weighted imaging with periodically rotated overlapping parallel lines with enhanced reconstruction (PROPELLER) has been suggested to improve visualization of the visualization of the fascicular component of the CNs in normal volunteers but with insufficient signal-to-noise ratio for routine clinical use.
Selected pathologic entities
The fascicular segment of the CNs may be interrupted by any pathologic condition within the brainstem, including demyelinating disease and neoplastic processes, such as those arising from the glial support cells found in the CNS or other masses. Surgical data on patients treated for hemifacial spasm suggest that the neurovascular compression occurs in the terminal fascicular component of the CN VII.b in many cases, often with compression of the brainstem rather than the cisternal segment itself as generally assumed (discussed later).
c. Cisternal Segment
Anatomic considerations
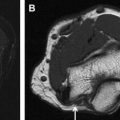
The end of the parenchymal fascicular segment and beginning of the cisternal segment is known anatomically as the root detachment point, corresponding approximately to the AO, where the CN can be visualized surrounded by CSF on imaging ( Fig. 4 B). As described previously, the root exit point is not synonymous with the AO for at least some of the CNs. The term, root exit zone , is considered ambiguous and its use is discouraged. Occasionally, multiple small free rootlets are visible (see Fig. 4 ) that converge on a root or several individual roots.
The proximal nerves in this segment are covered by pia mater and surrounded by CSF within the subarachnoid space. In the cisternal segment, the CNs may adjoin arterial or venous structures as well as the arachnoidal septae. Arterial or venous structures may directly abut, surround, or even divide the cisternal components of the CNs. The cisternal nerve roots extend toward the porus of the dural cave segment (described later) ( Fig. 5 ).
The zone of transition between the oligodendrocyte-myelinated CNS and the Schwann cell–myelinated PNS is called the CNS-PNS transitional zone (TZ) (sometimes given the eponym Obersteiner-Redlich zone). The TZ was initially described as a thinning of the myelin sheaths of spinal nerves just before the junction of the dorsal root and the cord, and the CNS component of the nerve proximal to this point is thought more prone to irritation. This point of transition is visible to the neurosurgeon through the operating microscope, because pia mater can be seen covering the centrally myelinated CNS component.
The length of the centrally myelinated portion of the cisternal segment and, therefore, the site of the TZ varies between the CNs. Among CNs III–XII (as reported by Lang ) the TZ is approximately 1 mm or less from the AO at the surface of the brainstem for CNs IV, V (motor root only), VI, IX, X, and XI. The other CNs vary significantly in the length of the CNS component/location of the TZ. The TZ of CN III.c is typically located 1.9 mm (range 1.0–4.0 mm) from the AO; the TZ of CN VII.c is located 2.1 mm (range 0.5–4.0 mm) from the AO; the TZ for the sensory root of CN V.c is located on average 3.6 mm (range 2.0–6.0 mm) from the AO. The longest centrally myelinated cisternal component of a CN is that of CN VIII.c, with the TZ located most often at or near the porus acousticus, approximately 10.0 mm from the brainstem (range 6.0–15.0 mm).
Imaging approaches
CN evaluation in the cisternal segment most often depends on negative contrast with a heavily T2-weighted appearance of CSF, an approach sometimes termed, magnetic resonance cisternography .
Although the cisternal CNs are generally well visualized, reports of visualization of CN IV.c have been variously reported as inconsistent to excellent, depending on technique and owing to its small size. Visualization is significantly improved with voxel sizes less than the diameter of the nerve. The site of the TZ, although visible to neurosurgeons through the operating microscope, is not readily identifiable through imaging alone, and its approximate location must be inferred through knowledge of the relevant CN anatomy.
Spin-echo T2-weighted imaging
Early MR imaging experience with imaging of the cisternal CNs with 2-D heavily T2-weighted turbo spin-echo imaging using peripheral pulse gating to minimize CSF flow artifacts was successful, particularly in visualizing CNs I, II, III, VII, and VIII. Subsequent studies demonstrated a significant advantage in detection of the CNs with 3-D compared with 2-D fast spin-echo sequences. The addition of driven equilibrium radiofrequency reset pulse (DRIVE) to turbo spin-echo 3-D imaging has been shown to reduce CSF flow artifacts with lower scan times. Comparison of 3-D fast asymmetric spin-echo and 3-D CISS imaging of the cerebellopontine angle cistern by one group of investigators favored 3-D fast asymmetric spin-echo due to “more prominent flow ghosts and magnetic susceptibility artifacts” on CISS.
Steady-state free precession imaging
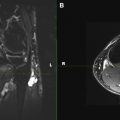
High-resolution steady-state free precession (SSFP) sequences with a heavily T2-weighted appearance have become the mainstay of visualization of the cisternal component of the CNs since their introduction by Casselman and colleagues. Several studies have assessed visualization of the CNs in the cisternal segment. 3-D CISS and the analogous 3-D fast imaging using steady-state acquisition (FIESTA) have been shown to be superior to 2-D T2-weighted images for the cisternal CNs. Additionally, comparison of CISS to 3-D magnetization-prepared rapid gradient echo (MP-RAGE), a T1-weighted 3-D technique, has favored CISS. In patients undergoing surgery for neurovascular compression of CN V.c, comparison of CISS to magnetic resonance angiography (MRA) suggested that CISS more accurately predicts intraoperative findings with respect to the relationship of vascular structures to the CN.
Although commonly thought of as a T2-weighted technique, CISS and FIESTA-C are fully refocused (balanced) steady-state sequences that demonstrate both T2 and T1 components. This combination of weightings is ideal for the evaluation of the CNs because it allows both high spatial resolution with suppression of CSF flow artifacts and the use of contrast agents ( Fig. 6 ). The use of contrast-enhanced CISS images for evaluation of cisternal masses has been proposed by Shigematsu and colleagues, who suggested that the technique could reveal the accurate location of CN VII.c and CN VIII.c relative to cerebellopontine angle masses.