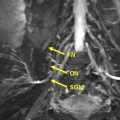
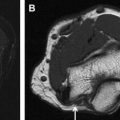
Peripheral nerve surgery represents a broad field of pathologic conditions, medical specialties, and anatomic regions of the body. Anatomic understanding of hierarchical nerve structure and the peripheral nervous system aids diagnosis and management of nerve lesions. Many peripheral nerves coalesce into organized arrays, including the cervical, brachial, and lumbosacral plexuses, controlling motor and sensory functions of the trunk and extremities. Individual or groups of nerves may be affected by various pathologic conditions, including trauma, entrapment, tumor, or iatrogenic damage. Current research efforts focus on enhancing the peripheral nerve regenerative process by targeting Schwann cells, nerve growth factors, and nerve allografts.
Key points
- •
Peripheral nerves may be affected individually or in concert by various pathologic conditions, including trauma, entrapment, tumor, or iatrogenic injury.
- •
After Wallerian (anterograde) and retrograde degeneration, peripheral nerves may undergo physiologic regeneration, aided by Schwann cells and nerve growth factors.
- •
Peripheral nerve sheath tumors include more common benign and infrequent malignant pathologicconditions. The operative approach varies depending on tumor pathology.
- •
Numerous operative techniques foster peripheral nerve regeneration, including nerve repair with coaptation, nerve grafting with autologous or synthetic grafts, and nerve transfers (neurotization).
- •
Goals of nerve transfer in brachial plexus injuries include elbow flexion, shoulder stability, and arm abduction. Numerous intraplexal and extraplexal donor nerves exist for neurotization.
Introduction
The peripheral nervous system represents a complex, organized network of nerves providing sensory, motor, and autonomic control of various end organs within the human body. Because of the anatomy and potential mechanisms of injury, multiple specialties collaborate in the care of patients with peripheral nerve pathologic conditions. The patient population represents a similarly wide spectrum, including neonates with birth-related brachial plexus injuries (BRBPI) to young adults sustaining brachial plexus trauma, and to older adults with nerve entrapments, tumors, or iatrogenic causes of peripheral nerve injury. The variety of pathologic conditions and anatomic regions invites a broad array of surgical techniques used within peripheral nerve surgery. Clear understanding of nerve structure and the peripheral nervous system aids diagnostic and therapeutic approaches to patients with peripheral nerve pathologic conditions.
Introduction
The peripheral nervous system represents a complex, organized network of nerves providing sensory, motor, and autonomic control of various end organs within the human body. Because of the anatomy and potential mechanisms of injury, multiple specialties collaborate in the care of patients with peripheral nerve pathologic conditions. The patient population represents a similarly wide spectrum, including neonates with birth-related brachial plexus injuries (BRBPI) to young adults sustaining brachial plexus trauma, and to older adults with nerve entrapments, tumors, or iatrogenic causes of peripheral nerve injury. The variety of pathologic conditions and anatomic regions invites a broad array of surgical techniques used within peripheral nerve surgery. Clear understanding of nerve structure and the peripheral nervous system aids diagnostic and therapeutic approaches to patients with peripheral nerve pathologic conditions.
Surgical anatomy
Components of a Peripheral Nerve
Peripheral nerves possess a hierarchical internal configuration composed of neural and connective tissue elements surrounded by endoneurium, and multiple myelinated axons join in parallel to form a fascicle. Perineurium envelopes each fascicle and contributes paramount strength to nerve structure. The perineurium also represents the blood nerve barrier. Multiple fascicles travel in parallel, surrounded by internal epineurium and an external layer of epineurium. Connective tissue and vasa nervorum reinforce the external layer of epineurium, providing extra support and blood supply. Several nerves within the human body coalesce into networks, including the cervical, brachial, and lumbosacral plexuses.
Cervical Plexus
The cervical plexus originates deep to the sternocleidomastoid and emerges anterolateral to the middle scalene muscle. Organized into multiple loops, the cervical plexus controls sensory and motor functions within the neck, posterolateral scalp, and superolateral thorax. Ventral rami from C1-C5 contribute to the cervical plexus and split (except C1) into ascending and descending divisions that join segments from adjacent levels. The ansa cervicalis represents a prominent loop of nerves originating from C1 (superior root) and C2-C3 (inferior root) serving primarily motor functions.
Posterior branches from the cervical plexus provide sensory innervation to the anterolateral neck, scalp between the pinna and external occipital protuberance, and superolateral thorax ( Table 1 ). Anterior motor branches from the cervical plexus control suprahyoid and infrahyoid muscles and the diaphragm (see Table 1 ). The ansa cervicalis sends motor branches to infrahyoid muscles, including the sternohyoid, sternothyroid, and omohyoid muscles. Arising mainly from the C4 ventral ramus with contributions from C3 and C5, the phrenic nerve provides the sole motor control of the diaphragm along with innervating the mediastinal pleura and pericardium.
| Nerve Branch | Origin | Function | Distribution or Target |
|---|---|---|---|
| Lesser occipital n. | C2-C3 | Sensory | Posterolateral scalp behind auricle, superolateral neck |
| Greater auricular n. | C2-C3 | Sensory | Skin superficial to parotid gland and around ear; posterior portion of auricle |
| Transverse cervical n. | C2-C3 | Sensory | Anterior neck |
| Supraclavicular n. | C3-C4 | Sensory | Lower neck, superolateral thorax, skin over shoulder |
| Branch traveling with hypoglossal n. | C1 | Motor | Geniohyoid and thyrohyoid mm. |
| Ansa cervicalis | C1-C3 | Motor | Infrahyoid mm.: sternohyoid, sternothyroid, omohyoid mm. |
| C1-C2 branches | C1-C2 | Motor | Rectus capitis anterioris and lateralis mm., longus capitis mm. |
| C2-C4 branches | C2-C4 | Motor | Longus colli and capitis mm. |
| C3-C4 branches | C3-C4 | Motor | Levator scapulae and scalene mm. |
| Phrenic n. | C3-C5 | Motor | Diaphragm |
Brachial Plexus
The brachial plexus usually originates from the cervicothoracic spinal roots of C5-T1. As anatomic variants, a prefixed plexus spans the cervical roots C4-C8, whereas a postfixed plexus encompasses roots C6-T2, with negligible contributions from T1 and C5, respectively. Plexus elements are organized in the following sequence:
Roots → Trunks → Divisions → Cords → Branches.
The brachial plexus runs inferolaterally from the vertebral foramina, between the anterior and middle scalene muscles, to below the clavicle and above the first thoracic rib. Plexus elements continue through the axilla, neighboring the axillary artery, to the ipsilateral upper extremity. The C5 and C6 roots join to form the upper trunk, whereas roots C8 and T1 combine to form the lower trunk. The middle trunk arises from the spinal root of C7 alone. The subclavian artery lies deep to the upper and middle trunks and ventral to the lower trunk.
Excluding the anterior and posterior divisions, each segment of the plexus contains peripheral nerve branches. Representing one of the first major branches, the long thoracic nerve emanates from roots C5-C7 to provide motor control of the serratus anterior muscle. Deficits of this nerve result in the familiar examination finding of scapular winging. Important in brachial plexus repair surgery, the suprascapular nerve originates from the upper trunk, traveling posteriorly through the suprascapular notch to innervate the supraspinatus and infraspinatus muscles.
Each trunk divides into anterior and posterior divisions just proximal to the upper margin of the clavicle. Running below and distal to the clavicle, the divisions coalesce into cords, labeled anatomically:
- •
The lateral cord (from anterior divisions of the upper and middle trunk) resides lateral and superficial to the axillary artery.
- •
The medial cord forms solely from the anterior division of the lower trunk, traveling medial and deep to the axillary artery.
- •
The 3 posterior divisions combine to form the posterior cord, located dorsal to the axillary artery.
Several terminal branches originate from the cords of the plexus to innervate distal targets ( Table 2 ). Between the clavicle and axilla, these cords separate into their respective terminal branches. The lateral cord splits into the musculocutaneous nerve and lateral contribution to the median nerve. The medial cord ultimately forms the ulnar nerve and medial contribution to the median nerve. After sending off the axillary nerve, the posterior cord continues distally as the radial nerve.
| Peripheral Nerve | Nerve Roots | Site of Origin | Innervated Target | Function |
|---|---|---|---|---|
| Dorsal scapular n. | C5 | C5 root | Levator scapulae and rhomboid mm. | Elevate and adduct scapula |
| Suprascapular n. | C5, C6 | Upper trunk | Supraspinatus, infraspinatus mm. | Arm abduction (<90°), external rotation |
| Long thoracic n. | C5-C7 | Roots | Serratus anterior mm. | Stabilizes scapula to chest wall |
| Lateral pectoral n. | C5-C7 | Lateral cord | Pectoralis major/minor mm. | Arm adduction, internal rotation |
| Medial pectoral n. | C8, T1 | Medial cord | Pectoralis major/minor mm. | Arm adduction, internal rotation |
| Thoracodorsal n. | C6-C8 | Posterior cord | Latissimus dorsi mm. | Arm adduction |
| Axillary n. | C5, C6 | Posterior cord | Deltoid, teres minor mm. | Arm abduction (>90°), external rotation |
| Radial n. | C5-C8 | Posterior cord | Triceps, supinator, brachioradialis mm., extensor carpi radialis longus/brevis | Forearm and radial hand extension; forearm supination |
| Posterior interosseous n. | C7, C8 | Radial n. | Extensor carpi ulnaris, extensor digitorum, extensor pollicis longus/brevis, abductor pollicis longus, extensor indicis and digiti minimi | Hand and finger extension, thumb abduction and extension |
| Musculocutaneous n. | C5-C7 | Lateral cord | Biceps, brachialis, and coracobrachialis mm.; forearm radial skin | Forearm flexion and supination; cutaneous sensation to radial aspect of forearm |
| Medial brachial cutaneous n. | T1 | Medial cord | Arm ulnar skin | Cutaneous sensation to ulnar aspect of arm |
| Medial antebrachial cutaneous n. | C8, T1 | Medial cord | Forearm ulnar skin | Cutaneous sensation to ulnar aspect of forearm |
| Ulnar n. | C8, T1 | Medial cord | Flexor carpi ulnaris, lumbricals 3–4, flexor digitorum profundus 3–4, hand interossei, adductor pollicis mm.; abductor, opponens, and flexor digiti minimi mm. | Ulnar hand and finger (digits 4–5) flexion; finger abduction and adduction; thumb adduction; little finger abduction, opposition, and flexion |
| Median n. | C6-T1 | Lateral and Medial cords | Palmaris longus, flexor carpi radialis, pronator teres, flexor digitorum superficialis; lumbricals 1–2, opponens pollicis, abductor and flexor pollicis brevi mm. | Hand and finger flexion; forearm pronation; thumb opposition, abduction, and flexion |
| Anterior interosseous n. | C7-T1 | Median n. | Flexor digitorum profundus 1–2, flexor pollicis longus, pronator quadratus mm. | Finger (digits 2–3) and thumb flexion |
The peripheral nerve branches of the brachial plexus provide motor and sensory control of the upper extremities (see Table 2 ). The anatomic location of the divisions implies the functionality of their distal branches:
- •
The anterior divisions ultimately provide motor control of flexors.
- •
The posterior divisions ultimately control upper extremity extensors.
Lumbosacral Plexus
Serving sensory, motor, and autonomic functions of the lower half of the body, the lumbosacral plexus arises from lumbar roots L1-L5 and sacral roots S1-S4. Although not organized into divisions or cords, the lumbosacral plexus sends off various branches to the lower trunk, reproductive organs, and lower extremities ( Table 3 ). Major proximal branches include the femoral and obturator nerves, both originating from roots L2-L4. The sciatic nerve (L4-S3) traverses the greater sciatic foramen and travels down the dorsal aspect of the thigh, splitting into the tibial and common peroneal nerves proximal to the knee. The common peroneal nerve splits proximally in the upper leg into the deep (L4-L5) and superficial (L5-S1) peroneal nerves. Serving important autonomic functions in the reproductive organs, bowel, and bladder, the pudendal and pelvic nerves originate from roots S2-S4.
| Peripheral Nerve | Origin | Sensory Distribution | Motor Function | Additional Comments |
|---|---|---|---|---|
| Iliohypogastric n. | L1 | Inferior trunk/abdomen | — | |
| Ilio-inguinal n. | L1 | Inguinal region | — | |
| Genitofemoral n. | L1-L2 | Genital region, groin | — | |
| Lateral femoral cutaneous n. | L2-L3 | Lateral thigh | — | Entrapment causes meralgia paresthetica |
| Femoral n. | L2-L4 | Anterior thigh to knee (anterior femoral cutaneous n.); medial leg from knee to foot (saphenous n.) | Hip flexion via iliopsoas and sartorius mm.; knee extension via quadriceps femoris mm. | |
| Obturator n. | L2-L4 | — | Thigh adduction via adductor longus, magnus, and brevis mm., gracilis mm. | |
| Superior gluteal n. | L4-S1 | — | Thigh abduction via gluteus medius and minimus mm.; thigh flexion via tensor fascia lata mm. | |
| Inferior gluteal n. | L5-S2 | — | Thigh abduction via gluteus maximus mm. | Tested with patient prone |
| Sciatic n. | L4-S3 | Posterior thigh (posterior femoral cutaneous n.); lower leg, sole of foot (tibial n.); sural n. | Knee flexion via biceps femoris, semitendinosus, and semimembranosis mm.; adductormagnus mm. | Semimembranosus, semitendinosus, and biceps femoris mm. commonly referred to as hamstrings |
| Tibial n. | L4-L5 | — | Plantarflexion via soleus, plantaris, gastrocnemius mm.; foot inversion via tibialis posterior mm.; toe flexion via flexor hallucis longus/brevis, flexor digitorum longus/brevis mm. | Tibial n. splits into medial and lateral plantar n. that supply mm. in foot |
| Deep peroneal n. | L4-L5 | — | Foot dorsiflexion via tibialis anterior mm.; toe extension via extensor digitorum longus/brevis, extensor hallucis longus mm. | Extensor hallucis longus mm. represents best test for L5 function |
| Superficial peroneal n. | L5-S1 | — | Foot eversion, plantarflexion of pronated foot via peroneus longus/brevis mm. | Common peroneal n. splits into superficial and deep peroneal n. |
| Pudendal n. | S2-S4 | Perineum and external genitalia | Urethral and external anal sphincters; contraction of pelvic floor | Sympathetic branches regulate ejaculation |
| Pelvic n. | S2-S4 | — | Parasympathetic branches control bowel, bladder, and sexual function | Parasympathetic branches regulate erection |
Pathology
Peripheral Nerve Injury
Injuries to peripheral nerves occur through various mechanisms including penetrating or blunt trauma, compression, crush, stretch or traction, and iatrogenic injury. Civilian nerve trauma most commonly involves stretch or crush injury due to motor vehicle accidents, whereas military nerve trauma frequently involves blast injuries. Nearly 5% of all soft tissue injuries involve damage to nearby peripheral nerves. Peripheral nerve trauma affects the upper extremities in greater than 70% of cases. For instance, the brachial plexus frequently sustains injury in high-velocity motorcycle accidents and BRBPI. The mechanism of injury often predicts the type of peripheral nerve damage:
- •
Motorcycle accidents frequently involve nerve root avulsions.
- •
Car collisions typically cause peripheral nerve crush injuries.
- •
BRBPI can cause transient deficits due to stretch injury or more serious deficits due to nerve root avulsion.
Regardless of the injury mechanism, damage to axonal integrity initiates Wallerian degeneration. This process represents the degeneration of axon segments distal to the site of injury in anterograde fashion. Wallerian degeneration begins soon after axonal injury and results in axonal and myelin disintegration. Macrophages responsible for debris removal can be seen along injured peripheral nerves as early as 3 days following trauma. Endoneurial tubes remain intact and devoid of neural components after phagocytic clearance of axonal and myelin debris.
Neuronal damage proximal to the site of injury also occurs because of axonal dysfunction. The decline in retrograde transport of nutrients along the axon toward the neuronal cell body ultimately leads to a breakdown of cell constituents. Microscopy of degenerating neurons reveals chromatolysis, or the destruction of Nissel substance in the nerve cell body. Distal muscle fibers also undergo degeneration and fibrosis when lacking peripheral nerve innervation. Fibrotic change initiates 3 weeks after axonal damage and completes within 18 months of denervation. The timeline of muscle atrophy, Wallerian degeneration, and loss of Schwann cells in the distal nerve carries significant implications for nerve grafting and repair.
Sunderland Classification of Peripheral Nerve Injury
Despite their higher resistance to mechanical deformation than central nerves, peripheral nerves undergo reproducible phases of injury. The Sunderland classification represents the most commonly used system of grading peripheral nerve damage. Divided into 5 ascending degrees of severity, the Sunderland classification stratifies nerve injury based on damage to neural anatomic components ( Table 4 ).
| Sunderland Degree | Components Damaged | Clinical Outcome Expected | Additional Comments |
|---|---|---|---|
| I | No structural damage; only conduction loss | Complete recovery | Consistent with neurapraxia |
| II | Axon, myelin | Complete recovery | Longer recovery period than degree I injuries |
| III | Axon, endoneurium, fascicle | Incomplete recovery with residual deficit | Intrafascicular hemorrhage and fibrosis complicate recovery |
| IV | Axon, endoneurium, fascicle, perineurium, internal epineurium | Partial spontaneous recovery; limited function of involved distal targets | Typically requires surgical repair |
| V | Entire nerve structure | Negligible recovery | Typically requires surgical repair |
Grade I injuries spare the continuity of the axon, but result in electrical conduction loss. Negligible structural damage with loss of nerve function describes a neurapraxia. The conduction deficit typically resolves, leading to complete recovery. A Sunderland degree II injury involves severing the axon and subsequent Wallerian degeneration distal to the injury. All structural components beyond the axonal level are preserved. Grade III classification indicates damage or disorganization to axons and endoneurium within the fascicles. In addition to Wallerian degeneration, retrograde injury is more pronounced than seen in grade II injuries. Sunderland degree IV injuries involve structural disruption to axons, fascicles, and perineurium with a loss of fascicular anatomy. The most severe class of peripheral nerve injury, Sunderland grade V, involves complete nerve discontinuity. With structural damage to all peripheral nerve anatomic components, nerve regeneration without intervention does not typically occur.
Peripheral Nerve Regeneration and Neuroma Formation
Distal targets of peripheral nerves release trophic factors to promote growth and survival of axons and neuronal cell bodies. Following axonal damage, proximal axonal sprouts grow distally, seeking these trophic factors. Chemotactic factors released by distal targets also help guide sprouting axons to the appropriate destination. Peripheral nerve regeneration occurs at an approximate rate of 1 mm/d or 1 inch/mo. After extending beyond a discontinuity, growing axons may enter endoneurial tubes previously vacated by Wallerian degeneration. The endoneurial tubes, along with trophic and chemotactic factors, act as guides to direct axonal regeneration toward the proper distal target. The entrance of Schwann cells into the endoneurial tubes further supports peripheral nerve reconstitution via myelin formation around budding axons.
The misdirection of axonal sprouting results in neuroma formation. Budding axons that do not properly cannulate an endoneurial tube grow astray, creating a focal tangle of regenerating axons, Schwann cells, and fibrotic tissue. Scarring of endoneurial tubes exacerbates this process as sprouting axons lack proper channels to enter. Intrafascicular hemorrhage and edema in significant nerve injury also contribute to the process of scarring in neuroma formation.
Multiple factors influence the success of peripheral nerve regeneration following injury. In general, younger patients show better rates of recovery and functional improvement than older patients. The location of the lesion also bears considerable weight on the success of peripheral nerve regeneration. Proximal lesions, including nerve root avulsions, exhibit poor rates of recovery. Similarly, proximal injuries require prolonged regeneration times to reach their targets, increasing the risk of muscle atrophy before reinnervation.
Schwann Cells and Nerve Growth Factors
Schwann cells and nerve growth factors (NGFs) play critical roles in the development, maintenance, and regeneration of peripheral nerves. Beyond providing myelin sheaths to enhance action potential conduction, Schwann cells aid peripheral nerve regeneration along with NGFs:
- •
Schwann cells contribute to the formation of longitudinal Hanken-Bungner’s cell cords that guide sprouting axons toward the appropriate target.
- •
Schwann cells secrete NGFs and neurotrophins to provide sustenance for regenerating axons.
- •
Schwann cells also produce collagen and laminin, integral components of peripheral nerve basal laminae.
- •
Acting as positive chemotactic agents, NGFs help guide sprouting axons to their destination.
- •
Neurotrophic factors stunt retrograde degeneration from the proximal nerve stump.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree