Why Apply Cardiac Magnetic Resonance Imaging to the Fetus: Is there a Need for Another Imaging Modality?
Assessment of fetal cardiac anatomy and function is an important component of the overall care of the newborn with cardiac disease. Fetal echocardiography has been the mainstay of cardiac assessment in utero, and successful diagnoses and monitoring of fetal therapy have been well-documented in the literature. Nevertheless, conventional fetal echocardiography is limited by the fact that it is not a technology that can readily offer three-dimensional (3D) visualization (which limits assessment of ventricular volumes and mass, for example), although recent advances over the past 10 years have enabled fetal echocardiography to provide for the reconstruction of two-dimensional (2D) images into 3D images through techniques such as spatiotemporal image correlation (STIC), and “real-time” 3D transducers and processing algorithms. Fetal echocardiography has a relatively limited field of view, can be constrained by acoustic windows and soft tissue characterization is lacking. Development of an alternative, noninvasive imaging modality to complement fetal echocardiography would be desirable and magnetic resonance imaging (MRI)—by virtue of its 3D capabilities to quantitatively assess morphology, ventricular volume, mass, and flow as well as its wide field of view—is uniquely suited to be this complementary imaging modality. MRI has been used for fetal extracardiac malformations for many years. It has been stated from experts in the field that “[Fetal MRI is] firmly established as an excellent complementary tool [to fetal ultrasongraphy] capable of providing exquisite anatomic detail” and sonography and “MRI have played critical roles in the identification of those fetuses most likely to benefit from different treatment strategies.” With such a track record, it seems logical to extend this technique to fetal cardiovascular evaluation as well.
Clinical needs for the additional quantitative information can be obtained with a 3D imaging modality such as MRI. One of these needs is accurate measurement of ventricular volume and mass, which is illustrated in cases such as hypoplastic left heart syndrome or pulmonary atresia with intact ventricular septum, in which determination of ventricular adequacy is critically important in management. Quantitatively, fetal echocardiography relies on geometrical assumptions to determine ventricular volumes and mass that are invalid in the oddly shaped hearts (or a compressed heart as when a diaphragmatic hernia is present) found in congenital heart disease. Accurate functional data obtained by MRI can aid in the medical decision-making process when deciding, for example, whether a patient can undergo a one- or two-ventricle repair when born. In addition, with the advent of fetal cardiac interventional techniques, accurate functional data obtained by MRI may be key in directing and evaluating the results of these interventions.
Another functional 3D parameter that can be provided by MRI is quantitative blood flow. When calculating blood flow by echocardiography, a sample volume is placed in the vessel of interest and the velocity-time integral is measured along with the diameter of the vessel. A number of assumptions are involved in this calculation including a circular cross-sectional area of the vessel and flat velocity profile. When cardiac MRI velocity mapping is utilized, velocity is measured “through-plane” across the cross-sectional area of the vessel, and when integrating this velocity across the cross-sectional area, blood flow (not solely velocity) can be obtained. This is a more direct measurement of blood flow and minimizes the assumptions made. In addition, blood flow measurements generally validate ventricular volume measurements made by MRI, confirming internal consistency. MRI velocity mapping has progressed to the stage at which now “instantaneous” or “real-time” flow measurements have been a reality ex utero since 2004 ; the future holds promise for this to be applied in utero. Accurate fetal flow measurement would be useful in patients with poor ventricular performance such as twin-twin transfusion syndrome and fetal heart block.
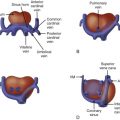
Three-dimensional anatomic imaging in the fetus has obvious advantages in patients with structural heart disease. The malformed cardiovascular system can be visualized more easily in this manner rather than inferring the 3D structure from 2D imaging. The wide field-of-view of MRI affords a more global picture of the cardiovascular system as well as the syndromes associated with congenital heart disease such as heterotaxy. For example, because the trachea is filled with fluid, MRI can determine the branching of the tracheobronchial tree in utero and sidedness of the aortic arch (see Figure 46-2C later); this is a challenge for echocardiography. Because a number of patients with congenital heart disease have extracardiac malformations, MRI is poised to be a “one-stop shop” for fetal evaluation for both cardiac and extracardiac lesions.
MRI utilization in the fetus has other potential advantages. MRI has been used to determine blood oxygen levels ex utero using the “BOLD” (blood oxygen level dependent) technique, and recent reports suggest it can be used in the fetus as well. MRI can also be used to assess myocardial metabolism and myocardial iron stores and holds the possibility of being used for interventions, in a manner similar to that of echocardiography. The applicability of MRI to the fetus can be utilized for research into the hemodynamic abnormalities that may cause or be a result of pathological heart defects. Fetuses may be followed over time to assess structure and hemodynamics after being identified with congenital heart disease.
The instances previously discussed are meant to convey that the development of fetal cardiac MRI is complementary to fetal echocardiography and is unlikely at this point in time to supplant it. For example, fetal echocardiography is limited to the acoustic windows available; MRI has no such limitation and can be used where windows are less than optimal. To assess soft tissues, ex vivo fetal imaging comparing MRI with echocardiography revealed that “MRI projections had better image quality and revealed more structural details than the sonographic views.” Finally, an important capability of cardiac MRI is that it can build images over multiple heartbeats, “averaging-out” or embedding ventricular performance over time into the image created, as opposed to the instantaneous ventricular performance obtained by echocardiography. The imager or clinician does not, therefore, have to evaluate multiple heartbeats and “average” the ventricular performance; it is embedded in the image created. This can complement the “instantaneous” temporal images and data derived from echocardiography.
The Development of Fetal Cardiac MRI for Visualizing Cardiovascular Morphology and Myocardial Motion
Although fetal MRI of extracardiac structures has played an important role in perinatal management for many years, extension of these technique to anatomical and functional fetal heart imaging has been limited. To obtain high-quality MRI images of cardiac structure and hemodynamics, data acquisition typically must be segmented over multiple heartbeats, necessitating synchronization to the cardiac cycle. This synchronization is generally achieved with electrocardiographic (ECG) triggering. However, detection of a fetal ECG signal can be problematic, particularly within the bore of an MRI scanner. The fetal ECG recording is typically corrupted by the maternal ECG reading and movement, and the MRI scanner itself induces significant artifacts via rapidly switching magnetic field gradients. In addition, during segmented image acquisition, MRI relies on the subject remaining motionless in the scanner and, with active fetuses, this again poses problems that must be overcome.
The main approach to fetal cardiac MRI avoids cardiac synchronization entirely by using single-shot and real-time imaging techniques, a method that can obtain bright blood anatomical and functional imaging in milliseconds. Considerable efforts have been devoted to the development of these rapid real-time imaging strategies including non-Cartesian k-space sampling, parallel imaging, and ultrafast MRI pulse sequence techniques. Besides precluding the need for cardiac synchronization, real-time imaging offers other potential advantages in insensitivity to arrhythmia, gross fetal or maternal motion and potential for interactive control of scan plane and other parameters.
An alternative approach to overcoming the problems associated with ECG measurement within the MRI system is to derive a physiological synchronization signal from the MRI data itself. This strategy, dubbed “self-gating,” offers the possibility of synchronizing MRI data acquisition with the motion of the fetal heart, providing the high spatial and temporal resolution benefits of segmented acquisition. This has already been demonstrated ex utero in adult and pediatric patients. “Real-time” MRI can be used for cine imaging of vascular structures and ventricular function as well as quantitative flow measures using real-time phase encoded velocity mapping.
Doppler ultrasound is commonly used to detect fetal heart rate and is a third approach to fetal cardiac imaging. The feasibility and safety of performing fetal ultrasound cardiotocography (CTG) in an MRI scanner have been evaluated independently in several studies. Commercial ultrasound scanners were modified with electronic shielding and magnetically compatible components for use in the MRI scanner. The safety of performing fetal CTG in a 1.5-T MRI system was most recently evaluated by Michel and coworkers. The monitoring of maternal and fetal heart rates, fetal movements before, during, and after MRI scanning for 10-minute periods showed no pathological accelerations, short-term or long-term changes in basal fetal heart rate, and no signs of fetal stress. An earlier attempt to use CTG monitoring to gate the MRI sequence was hampered by severe artifacts in the MRI images created by the unshielded ultrasound device. This study, however, demonstrated the feasibility of using CTG signals to cardiac-gate MRI pulse sequences in adult subjects. Although the investigators have successfully shielded an ultrasound machine for use with MRI, the same technology is yet to be optimized for fetal cardiac MRI.
Finally, a fourth approach leverages recent advances in ECG signal processing and offers the possibility of noninvasively gating the MRI scan to the fetal heart rate via a fetal ECG signal. Modifications of the same vectorcardiogram technology that has been successful in adult cardiac MRI may make fetal ECG gating feasible.
Safety of Fetal MRI
As with any imaging technique, safety should always be considered. Fetal MRI has been used as early as 1983, and to date, no negative side effects or delayed sequelae are known to be associated with fetal MRI. Isolate cell, animal, and human studies have confirmed this finding. Fetal MRI is considered by physicians working in the field as “firmly established as an excellent complementary tool to fetal ultrasongraphy capable of providing exquisite anatomic detail in many case.” In addition, these physicians have stated that sonography and “MRI have played critical roles in the identification of those fetuses most likely to benefit from different treatment strategies.”
Multiple animal research studies have been published with regard to the developing fetus and exposure to magnetic fields. Behr and colleagues. exposed chick embryos to different static and time-varying gradient field strengths and found no influence from MRI on the embryo mortality, hatching rate, or the vitality of the chickens. In mice exposed to a magnetic field at 0.35 T in midgestation, Heinrichs and associates found no gross effects of embryotoxicity and some “evidence” of reduction in crown-rump length. Yip and coworkers studied cell proliferation and migration effects at 1.5 T on chicks as well as axonal outgrowth in the sympathetic nervous system and found no effects. In pregnant mice exposed to 4.7 T (more than three times the field strength of a routine 1.5 T scanner) for 8 hours (more than eight times the exposure of a typical study) on day 9 or 12 of gestation, significant differences were found in biological endpoints such as fetal weight reduction, crown-rump length reduction, postpartum death rate increase, and a reduction in daily sperm production. This is in contrast to the findings of Magin and colleagues, who found no effects on fetal growth and postnatal development in mice as a result of long-term exposure to 4T. Chew and associates reported no statistical effects in the rate of blastocyst formation between control groups and those exposed to 1.5 T with MRI clinical sequences.
A number of human studies also confirm the safety of fetal MRI. Baker and coworkers performed a 3-year follow-up study of children imaged in utero and no demonstrable increase in disease or disability could be related to the echo planar imaging (EPI) used at 0.5 T on 20 pregnant women. Similarly, Myers and colleagues investigated the effects of EPI on 74 pregnant women exposed to 0.5 T, utilizing 148 women not exposed to MRI as a control group; no statistical difference was observed with regard to intrauterine growth retardation. There have also been a few studies on fetal heart rate (a measure of fetal distress) during MRI encompassing 34 pregnant patients exposed to 0.5 to 1.5 T, and none of the studies could detect any variance in fetal heart rate. Although certainly not the same as undergoing a scan, a study by Kanal and associates surveyed female MRI technologists to establish static fringe field effects and received 1915 responses covering 1421 pregnancies. They studied factors such as fertility, length of gestation, birth weight, pregnancy outcome, and offspring gender and found no statistical significant associations with working in an MRI. Shellock and Crues reviewed a number of studies on the safety of fetal MRI as part of an overall review of MRI safety and concluded that the “overall findings from these studies indicate that there is no substantial evidence of injury or harm to the fetus, however, additional research on this topic in warranted.”
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree