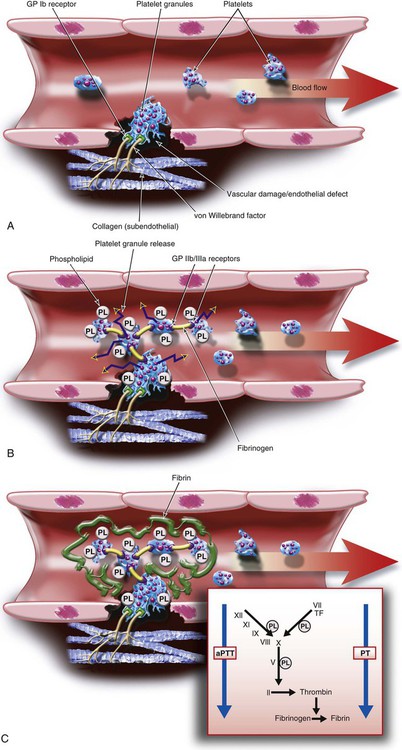
Chapter 22 Vascular injury leads to thrombus formation. As a first step, a platelet plug is formed, which is then surrounded and strengthened by a fibrin polymer meshwork. Several steps are involved in this process (Fig. 22-1): 1. Platelet adhesion: During the first step of hemostasis, von Willebrand factor multimers bind to exposed subendothelial collagen with one part of their structure and to the glycoprotein (GP)Ib receptor on the surface of platelets with another, thereby anchoring the platelet to the site of injury. 2. Platelet aggregation: Platelet adhesion leads to activation of the anchored platelet, which results in three major reactions: • Release of platelet granules: Platelet granules contain platelet agonists such as thromboxane A2, adenosine diphosphate (ADP), and epinephrine. Thromboxane A2 is synthesized with the help of the enzyme cyclooxygenase (COX)-1. Once released into the bloodstream, the agonists bind to receptors on the surface of platelets trying to float by, thus activating and recruiting them to the site of injury. The platelet receptors for ADP are termed P2Y1 and P2Y12, and it appears both must be activated for platelet aggregation to take place. • Activation of platelet surface GPIIb/IIIa receptors: Activation of the platelet leads to a change in shape of the GPIIb/IIIa receptor, enabling it to bind fibrinogen. One end of the fibrinogen molecule binds to the GPIIb/IIIa receptor of one activated platelet and the other to the receptor of another platelet, thus bridging platelets and creating a platelet plug. • Flip-flop of phospholipids: In nonactivated platelets, phospholipids are located on the inner side of the cell membrane. Platelet activation leads to a flip-flop of these phospholipids to the outside of the platelet membrane, where then they can bind coagulation factors and calcium and facilitate the various reactions of the coagulation cascade that lead to formation of fibrin. 3. Plasmatic coagulation: Vascular injury brings tissue factor on extracellular cells (fibroblasts, monocytes) into contact with circulating coagulation factor VIIa, thereby initiating the coagulation cascade that leads to thrombin and eventually fibrin formation. Coagulation factor activation reactions require phospholipids, which are provided by platelets during the flip-flop mechanism that occurs during platelet aggregation. A fibrin meshwork forms around the platelets anchored at the site of vascular injury. A thrombus is formed. Venous thrombosis occurs mostly via the plasma coagulation system, with only minor platelet participation. In contrast, platelets play a major role in arterial thrombus formation, with additional participation of the plasma coagulation system. This paradigm helps explain why drugs that block the plasmatic coagulation reaction (i.e., anticoagulants) are very active in preventing venous thrombosis and also effective in preventing arterial thrombosis, whereas antiplatelet drugs, which successfully prevent arterial thrombosis, are less or not at all effective in venous disease. Table 22-1 lists the important antiplatelet agents and anticoagulants, discussed in further detail in the following sections. TABLE 22-1 Antiplatelet Agents and Anticoagulants †Drug approval and dosing regimens may vary depending on indication, country of use, presence or absence of renal or liver dysfunction, and concomitant use of antiplatelet drugs or anticoagulants. Aspirin (acetylsalicylic acid) inhibits the enzyme COX-1, which is needed to form thromboxane A2 in platelets. Thromboxane A2 is normally released from platelet granules upon platelet adhesion and during platelet aggregation (see Fig. 22-1, B) and serves as an agonist to activate and thereby recruit other platelets to the platelet plug. Because platelets do not synthesize new cyclooxygenase and aspirin binds irreversibly to the enzyme, aspirin’s action lasts for the lifespan of a platelet (i.e., 7-10 days). Complete inactivation of platelet COX-1 is typically achieved with a daily aspirin dose of 160 mg. When used as an antithrombotic drug, aspirin is maximally effective at doses between 50 and 325 mg/day. Higher doses do not improve efficacy. However, there is considerable interindividual variability in aspirin’s ability to inhibit COX-1. “Aspirin resistance” is a laboratory phenomenon in which there is an inability of aspirin to inhibit one or more in vitro tests of platelet function, such as platelet aggregometry, the Platelet Function Analyzer (PFA-100), the VerifyNow Aspirin rapid platelet function assay, or measurement of thromboxane generation in vitro or in vivo via serum levels of thromboxane B2 or urinary levels of 11-dehydrothromboxane B2. “Aspirin failure” is a clinical observation of treatment failure. Aspirin resistance does not necessarily lead to treatment failure, nor is clinical aspirin failure necessarily due to aspirin resistance. The platelet GPIIb/IIIa receptors (also termed integrin αIIbβ3) are the sites where fibrinogen binds during platelet aggregation (see Fig. 22-1, B), leading to cross-linking of platelets and platelet plug formation. Several inhibitors of this receptor have been developed for clinical use.
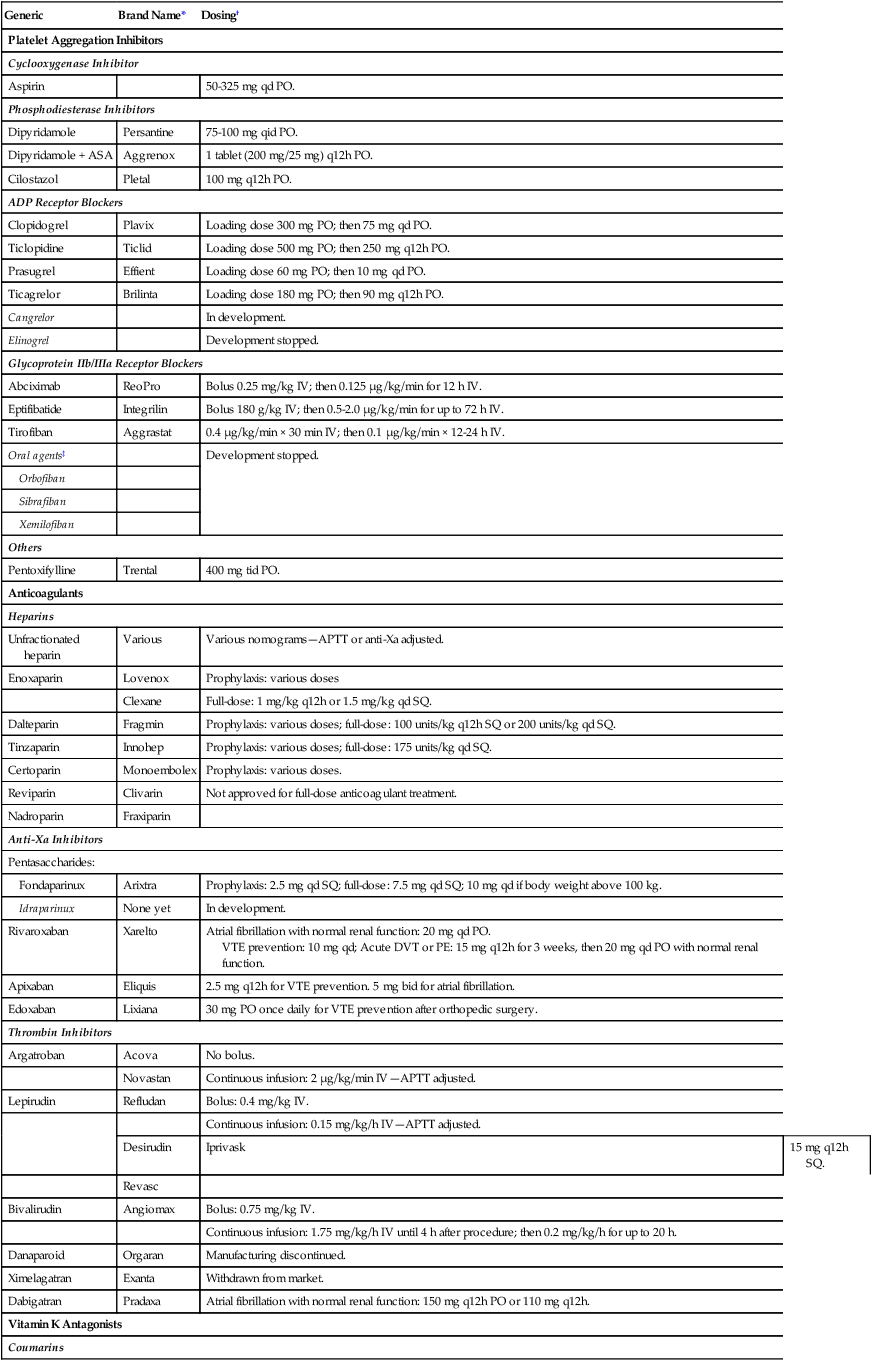
Antiplatelet Agents and Anticoagulants
Physiology of Blood Clotting
Pathophysiology of Blood Clotting
Generic
Brand Name*
Dosing†
Platelet Aggregation Inhibitors
Cyclooxygenase Inhibitor
Aspirin
50-325 mg qd PO.
Phosphodiesterase Inhibitors
Dipyridamole
Persantine
75-100 mg qid PO.
Dipyridamole + ASA
Aggrenox
1 tablet (200 mg/25 mg) q12h PO.
Cilostazol
Pletal
100 mg q12h PO.
ADP Receptor Blockers
Clopidogrel
Plavix
Loading dose 300 mg PO; then 75 mg qd PO.
Ticlopidine
Ticlid
Loading dose 500 mg PO; then 250 mg q12h PO.
Prasugrel
Effient
Loading dose 60 mg PO; then 10 mg qd PO.
Ticagrelor
Brilinta
Loading dose 180 mg PO; then 90 mg q12h PO.
Cangrelor
In development.
Elinogrel
Development stopped.
Glycoprotein IIb/IIIa Receptor Blockers
Abciximab
ReoPro
Bolus 0.25 mg/kg IV; then 0.125 µg/kg/min for 12 h IV.
Eptifibatide
Integrilin
Bolus 180 g/kg IV; then 0.5-2.0 µg/kg/min for up to 72 h IV.
Tirofiban
Aggrastat
0.4 µg/kg/min × 30 min IV; then 0.1 µg/kg/min × 12-24 h IV.
Oral agents‡
Development stopped.
Orbofiban
Sibrafiban
Xemilofiban
Others
Pentoxifylline
Trental
400 mg tid PO.
Anticoagulants
Heparins
Unfractionated heparin
Various
Various nomograms—APTT or anti-Xa adjusted.
Enoxaparin
Lovenox
Prophylaxis: various doses
Clexane
Full-dose: 1 mg/kg q12h or 1.5 mg/kg qd SQ.
Dalteparin
Fragmin
Prophylaxis: various doses; full-dose: 100 units/kg q12h SQ or 200 units/kg qd SQ.
Tinzaparin
Innohep
Prophylaxis: various doses; full-dose: 175 units/kg qd SQ.
Certoparin
Monoembolex
Prophylaxis: various doses.
Reviparin
Clivarin
Not approved for full-dose anticoagulant treatment.
Nadroparin
Fraxiparin
Anti-Xa Inhibitors
Pentasaccharides:
Fondaparinux
Arixtra
Prophylaxis: 2.5 mg qd SQ; full-dose: 7.5 mg qd SQ; 10 mg qd if body weight above 100 kg.
Idraparinux
None yet
In development.
Rivaroxaban
Xarelto
Atrial fibrillation with normal renal function: 20 mg qd PO.
VTE prevention: 10 mg qd; Acute DVT or PE: 15 mg q12h for 3 weeks, then 20 mg qd PO with normal renal function.
Apixaban
Eliquis
2.5 mg q12h for VTE prevention. 5 mg bid for atrial fibrillation.
Edoxaban
Lixiana
30 mg PO once daily for VTE prevention after orthopedic surgery.
Thrombin Inhibitors
Argatroban
Acova
No bolus.
Novastan
Continuous infusion: 2 µg/kg/min IV—APTT adjusted.
Lepirudin
Refludan
Bolus: 0.4 mg/kg IV.
Continuous infusion: 0.15 mg/kg/h IV—APTT adjusted.
Desirudin
Iprivask
15 mg q12h SQ.
Revasc
Bivalirudin
Angiomax
Bolus: 0.75 mg/kg IV.
Continuous infusion: 1.75 mg/kg/h IV until 4 h after procedure; then 0.2 mg/kg/h for up to 20 h.
Danaparoid
Orgaran
Manufacturing discontinued.
Ximelagatran
Exanta
Withdrawn from market.
Dabigatran
Pradaxa
Atrial fibrillation with normal renal function: 150 mg q12h PO or 110 mg q12h.
Vitamin K Antagonists
Coumarins
Warfarin
Coumadin
Interindividual variability; dosing is INR adjusted.
Jantoven
Phenprocoumon
Marcumar
Falithrom
Phenpro
Acenocoumarol
Sinthrome
Tioclomarol
Apegmone
Indandiones
Fluindione
Previscan
Anisindione
Miradon
Phenindione
Dindevan
Pindione

Antiplatelet Agents
Aspirin
Glycoprotein IIb/Ill Receptor Antagonists
Radiology Key
Fastest Radiology Insight Engine