BENIGN AND MALIGNANT NEUROECTODERMAL AND OTHER NEUROGENIC TUMORS
KEY POINTS
- Neurogenic tumors in the head run a gamut from very benign to highly malignant behavior.
- Proper imaging can greatly enhance chances for cure in cases where gross total resection is important in a plan for local control.
OLFACTORY NEUROBLASTOMA (ESTHESIONEUROBLASTOMA)
Patterns of Disease and Pathology
Olfactory neuroblastoma is a neurogenic tumor arising from specialized olfactory epithelium situated relatively high in the nasal cavity.1 In adults, the olfactory epithelium lines the upper surface of the superior turbinate, a corresponding area of the nasal septum, and the undersurface of the cribriform plate. The bipolar receptor cells in the olfactory epithelium terminate in the olfactory bulb, making the olfactory nerve the only cranial nerve whose first-order neuron terminates in a distal epithelium. Olfactory neuroblastoma most often arises from the basal layer of this epithelium. The neoplasm may occur anywhere from the first to eighth decade of life but peaks in the 10- to 30-year age range without a significant gender predilection.2,3.
Olfactory neuroblastoma frequently invades the ethmoid sinuses, orbit, and anterior cranial fossa (Figs. 30.1–30.6). Spread across the cribriform plate is the potentially most devastating event with regard to the effect on survival. Tumor can invade the meninges at the anterior skull base and even spread to the pia without obvious destruction of the cribriform plate. However, bone destruction at the anterior skull base is normally obvious once the brain and/or meninges are involved (Figs. 30.1–30.3). Cerebrospinal fluid dissemination is unusual at presentation, but it is a common mode of local failure (Fig. 30.6). Spread along the dura is not uncommon at presentation. The nasal septum may be destroyed. More lateral spread is to the medial wall of the maxillary sinus, the ethmoid sinuses, the pterygopalatine fossa, and the orbit. Posterior spread to the central skull base and sphenoid sinus may be present in advanced cases, but the tumor is usually more anterior than that at the time of initial presentation.


FIGURE 30.1. Contrast-enhanced magnetic resonance study of a patient with olfactory neuroblastoma. A: Contrast-enhancedT1-weighted image showing the mass centered in the high nasal cavity extending through the ethmoid sinuses and anterior skull base with invasion of the medial aspect of the orbit (arrows). B: T2-weighted image to correlate with that in figure (A) showing the same overall extent of the tumor with differentiation of tumor margins from obstructive sinus changes (arrowheads).


FIGURE 30.2. Contrast-enhanced computed tomography of a patient with olfactory neuroblastoma. A: Tumor within the nasal cavity (arrowhead) bilaterally invades through the cribriform plate to extend likely transdural into the anterior cranial fossa (arrows). B: Sagittal image showing the overall anterior to posterior extent of the mass and only equivocal evidence of spread beyond the anterior skull base (arrows) that would be much better demonstrated on magnetic resonance imaging.






FIGURE 30.3. Contrast-enhanced magnetic resonance (MR) imaging and computed tomography (CT) study in a patient with olfactory neuroblastoma. A: Coronal contrast-enhanced CT showing the mass originating in the olfactory mucosa of the upper nasal cavity extending across the anterior skull base to spread intracranially (arrowheads). A tumoral cyst is noted superior to the more solid component (arrow). B: Coronal bone window CT study shows related bony defect in the ethmoids and anterior skull base. C: Non–contrast-enhancedT1-weighted (T1W) image in the same patient showing the lesion extending from the nasal cavity to the anterior cranial fossa (arrows) through a narrower defect in the anterior skull base (arrowhead). D: Contrast-enhanced T1W image roughly correlating with the CT image in (A) showing the solid component of the mass (arrowheads) and the tumor-related cyst (arrow). E: T2-weighted (T2W) coronal image correlating with CT in (A) and T1W MR in (B) showing the solid component of the tumor (arrowheads) to be isointense the brain and the tumor-related cyst obviously bright (arrow). F: Axial T2W images show the tumor-related cyst and edema in adjacent brain (arrows). The brain edema suggests likely pial involvement of the brain or at least edema due to adjacent vascular congestion. However, brain invasion must be considered possible once brain edema is noted.



FIGURE 30.4. Computed tomography examinations of two patients with olfactory neuroblastomas that calcified. A: Four contiguous sections showing the overall degree of calcification and how the lesion invades the medial aspect of the orbit and lacrimal sac region (arrows). B: Enlarged view of one of the images in (A) showing the detail of tumoral calcification and involvement of adjacent bony orbit and maxillary sinus (arrows). C: A second patient with a different pattern of tumoral calcification.

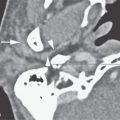
FIGURE 30.5. Contrast-enhanced computed tomography of a patient with a very limited olfactory neuroblastoma. The tumor is manifest only as nonspecific soft tissue thickening along the nasal septum (arrows).








FIGURE 30.6. Computed tomography study of a patient treated with olfactory neuroblastoma demonstrating patterns of recurrent disease in this tumor following treatment. A: Bone windows show the repaired surgical defect in the anterior skull base and no definite evidence of recurrence based on bone detail. Sinonasal changes are all reactive and not due to tumor. B: At the area of dural closure anteriorally along the resected inner table of the frontal sinus, there are dural reactive changes that are nonspecific. However, there is nodular recurrence along the meninges of the left temporal lobe (arrowheads). C; More cephalad section shows abnormal dural thickening (arrows) on the right side and less specific dural thickening on the left side (arrowhead). DC: Multiple dural and possible pial nodules of recurrent tumor (arrows). ED: Close-up view of the dural(arrows) and pial disease along the left temporal lobe with abnormal pial enhancement within the sylvian fissure(arrowhead). FE: More remote meningeal seeding manifesting at the cranial cervical junction (arrow). This patient had disseminated disease throughout the spinal subarachnoid space as well. GF: Metastatic level 2A lymphadenopathy showing a very aggressive extranodal spread pattern (arrow) present encasing the jugular vein (arrowhead). HG: Multiple positive lymph nodes in level 1B on both sides (arrows) and level 2A on left (arrowhead).
Lymph node metastases to retropharyngeal and cervical groups are possible (Fig. 30.6).
Local failure will usually manifest at the skull base margin and/or as a meningeal spread pattern. Retropharyngeal node involvement may be the earliest and only manifestation of recurrence.
Imaging Appearance
The gross margins of olfactory neuroblastomas are often irregular and indistinct; although the mass is well defined, the invasive nature of the lesion is usually clear from attendant bone destruction frequently present even in small lesions (Figs. 30.1–30.4). Calcifications may be present (Fig. 30.4). Histologically, there is a neurofibrillary matrix with clusters of small round to oval cells grouped by vascular septae; this accounts for the computed tomography (CT) and magnetic resonance (MR) appearance. On T1-weighted images, the tumors are isointense or slightly hypointense to brain; on T2-weighted images, they are usually isointense but may be hyperintense to brain4(Fig. 30.5).4 The lesions normally enhance markedly and diffusely on contrast-enhanced computed tomography (CECT) and contrast-enhanced magnetic resonance (CEMR), reflecting the vascular septae in the stroma4(Figs. 30.1–30.3).4
The tumors are tenacious with a tendency for local recurrence. Dural and leptomeningeal dissemination is possible, especially with recurrent disease5(Fig. 30.6).5 Metastases occur in at least 20% of patients, most commonly to retropharyngeal cervical nodes and the lungs, and both of these sites are common modes of recurrent disease (Fig. 30.6).
Treatment
Treatment is usually a combination of surgery, radiation, and chemotherapy.2–6,7 Overall survival is approximately 50% at 5 years and is mainly dependent on the extent at the time of initial diagnosis and the adequacy of the initial resection. The latter being true, imaging plays a key role in helping to establish the full extent of the tumor so that gross total resection margins are properly anticipated.
NEUROBLASTOMA
Patterns of Disease and Pathology
Neuroblastomas are neurogenic neoplasms arising from cells derived from embryonic sympathetic neuroblasts. They are seen most commonly in the retroperitoneal-adrenal region, but they can arise wherever sympathetic ganglia are located. These lesions are most commonly encountered in children and account for 10% to 20% of childhood malignancies. In the head and neck region, these are most commonly encountered as metastatic lesions to the skull and facial bones, in particular the orbits. The metastases may be osteoblastic, osteolytic, or mixed.
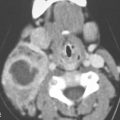
Primary neuroblastoma in the head and neck arises from the cervical sympathetic plexus and accounts for very few of the lesions. These may present in early childhood with feeding and swallowing difficulty due to bulk of the mass compressing the pharynx and/or Horner syndrome (Figs. 30.7 and 30.8).




FIGURE 30.7. Non–contrast-enhanced computed tomography in a patient with cervical neuroblastoma. A: Calcified mass in the sympathetic plexus region (arrow). B: Mass continues inferiorly medial to the position of the sympathetic plexus (arrows) with more central classification (arrowhead). C: Continued abnormal calcification along the course of the sympathetic plexus (arrow) with suggestion of a calcified metastatic lymph node (arrowhead). D: The inferior extent of the calcified mass in the right tracheoesophageal groove (arrowheads) with calcifications present either in the mass or in related metastatic lymph nodes (arrows).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree