Benign Testicular Lesions
Etiology and Clinical Presentation
Benign scrotal or testicular swellings and masses have many etiologies and different clinical presentations, as listed in Tables 78-1 and 78-2 . Of palpable nodules, 31% to 47% are benign at surgery.
| Condition | Symptoms | Signs | Comments |
|---|---|---|---|
| Torsion | Acute onset of severe pain, usually postpubertal | Pain not relieved by scrotal elevation, high-riding testis, absent cremasteric reflex | Surgical emergency |
| Epididymo-orchitis | Severe acute onset of pain, older age group | Edema, tenderness, erythema | Positive urinalysis |
| Torsion of appendix testis | Gradual onset of pain, usually prepubertal | Tenderness localized to anterosuperior testis, cremasteric reflex preserved | Managed conservatively |
| Trauma | History of injury | Depends on severity of injury | May result in infarction, rupture, or torsion of testis. |
| Infarction | Depends on cause | Not specific | Possible causes include torsion, epididymo-orchitis, vasculitis, hypercoagulable states, sickle cell disease. |
| Abscess | Pain, fever, failure to improve with antibiotics in case of abscess secondary to epididymo-orchitis | Febrile; tender, swollen scrotum Signs may be masked in patients with acquired immunodeficiency syndrome | Known complication of mumps, smallpox, scarlet fever, influenza, typhoid, and sinusitis. Fournier’s gangrene may extend to involve the testis. |
| Hematocele/pyocele | Depends on cause | Edematous and swollen in the acute phase, scrotal skin thickening, calcification when chronic | Hematoceles occur after trauma, including iatrogenic trauma. Pyoceles form from rupture of an abscess into, or infection of, a hydrocele. |
| Incarcerated inguinal hernia | Sudden irreducibility of inguinal hernia with pain | Nonreducible, edematous, red inguinal mass | Usually presents in the setting of a preexisting inguinal hernia. |
| Condition | Symptoms | Signs | Comments |
|---|---|---|---|
| Hydrocele | Painless mass that may increase slowly in size | Transillumination positive | Reactive hydrocele may be associated with epididymo-orchitis or other inflammation. |
| Testicular cyst | None | Usually nonpalpable | Incidental finding on ultrasonography. |
| Varicocele | Scrotal swelling, infertility | More common on the left; “bag of worms” feel on palpation, pronounced on Valsalva maneuver | Sudden onset of a right-sided varicocele, or any irreducible varicocele, may be due to retroperitoneal pathology (e.g., renal cell carcinoma) compressing the testicular vein. |
| Epidermoid cyst | Painless mass or none | Palpable mass or none | Often removed surgically because it may be difficult to differentiate from malignancy. |
| Tubular ectasia of rete testis | None | None | Incidentally discovered; may be associated with prior infection, trauma, or scrotal surgery |
| Adrenal rests | Often manifest as bilateral scrotal swelling | Bilateral scrotal masses | Usually associated with elevated corticotropin level |
| Spermatocele | Asymptomatic or may manifest as small focal scrotal lump | Discrete soft mass near epididymis, freely moving and superior to testis with positive transillumination | Arises from obstructed efferent ductules usually in patients with prior vasectomy. |
| Testicular microlithiasis | Asymptomatic | None | Incidental diagnosis, may be premalignant, follow-up imaging is often advised. |
| Scrotal pearl | Asymptomatic or may manifest as nodular swelling | Hard nodule may or may not be felt. | Generally idiopathic and benign; may be from a torsed appendix testis. |
Imaging
Testicular Torsion, Testicular Infarction, and Torsion of the Testicular Appendage
Testicular torsion occurs when the spermatic cord is twisted, compromising the blood flow to and from the testis. Torsion can be classified as intravaginal or extravaginal. Intravaginal torsion occurs when the mesenteric attachment of the spermatic cord on the testis is narrow, allowing the testis to rotate within the cavity of the tunica vaginalis like a “bell clapper.” It typically affects males between the ages of 12 and 18 years, probably owing to the fivefold increase in testicular volume at puberty. Extravaginal torsion, in which the entire testis, epididymis, and tunica vaginalis twist in a vertical axis, is rare and is typically seen prenatally (75%) or in neonates. Intravaginal torsion is characterized by pain of sudden or insidious onset, whereas extravaginal or neonatal torsion may be completely asymptomatic, and a unilateral mass in the inguinal region or high scrotum may be the only manifestation. The extent of the spermatic cord twist and its duration are the two most important factors. The initial disruption of blood supply will be to the venous and lymphatic drainage, rather than to the arterial supply of the testes, and venous infarction occurs earlier and at lesser levels of torsion.
Magnetic Resonance Imaging.
Magnetic resonance imaging (MRI) has been used to differentiate subacute testicular torsion from epididymitis with a high degree of accuracy. On MRI, an enlarged spermatic cord (secondary to edema) with no increase in cord vascularity (seen as lack of vascular flow voids) and a “whirlpool pattern” and “torsion knot” (best seen on T2-weighted coronal views) are specific for torsion.
Contrast-enhanced MRI can help in the diagnosis of segmental testicular infarction ( Figure 78-1 ). Dynamic contrast-enhanced subtraction MRI can diagnose testicular torsion and detect testicular necrosis with a high degree of sensitivity and accuracy.

Ultrasonography.
On ultrasonography, the initial appearance of the testis is hypoechoic and enlarged secondary to congestion accompanied by a small hydrocele. Later, increased echogenicity and heterogeneity can be seen from hemorrhagic change. The epididymis also may be involved ( Figure 78-2 ). A small hypoechoic testis, with an enlarged echogenic epididymis, is seen with a chronic missed torsion. There may be an abrupt change in caliber of the spermatic cord below the point of torsion. Ultrasound demonstration of the “whirlpool” sign in the spermatic cord with absent or reduced flow distal to the whirlpool is a reliable indicator of torsion. Skin thickening may manifest as venous congestion.

Color Doppler imaging allows visualization of intratesticular blood flow, which is either reduced or absent in torsion. This distinction may be less apparent when orchitis is complicated by testicular infarction. Technical factors, including equipment and operator experience, may limit the quality of the study. A useful caveat to use in such conditions is to compare the findings with the contralateral normal testis. Color Doppler flow is difficult to detect in testes less than 1 mL in volume (i.e., in children). Techniques including power Doppler imaging and the use of contrast agents may improve detection of intratesticular flow.
The most common cause of scrotal pain in a young child is torsion of the testicular appendix. The ultrasound appearance of the twisted testicular appendage has been described as an avascular hypoechoic or echogenic mass adjacent to a normally perfused testis and surrounded by an area of increased color Doppler flow.
Nuclear Medicine.
Traditionally, testicular scintigraphy has been used in the assessment of testicular torsion, but this method suffers from the constraints of limited access out of hours, complex equipment needs, and prolonged examination times.
Imaging Algorithm.
An imaging algorithm for testicular torsion, adapted from the American College of Radiology Appropriateness Criteria for Acute Scrotal Pain without Antecedent Mass or Trauma is proposed in Figure 78-3 and Table 78-3 .

| Modality | Accuracy | Limitations | Pitfalls |
|---|---|---|---|
| MRI | 93%-100% | Motion sensitive, requires more time to perform than ultrasonography, skill to interpret, relatively expensive | T2*-weighted images may not be sensitive for detection of late contrast enhancement and are vulnerable to susceptibility artifacts that result from the air/tissue interface, which may distort images and hamper accurate measurement of signal intensity. |
| Ultrasonography | 89%-100% | Lower sensitivity in prepubertal and younger children | Blood flow can be preserved in torsion and detorsion, when hyperemia can be mistaken for orchitis. |
| Nuclear medicine | 90% sensitivity 60% specificity | Cannot be applied to small children Lower availability of trained personnel to perform and interpret the examination | Hyperemic epididymis may be misinterpreted as a halo, producing false-positive study. Photon-deficient areas secondary to hydrocele, spermatocele, uncommonly edematous appendix testis, and rarely an inguinal hernia can be mistaken for an avascular testis. |
Epididymitis, Epididymo-Orchitis, and Testicular Abscess
Epididymitis is the most common cause of acute scrotal pain in postpubertal men. In 20% of patients, testicular extension results in epididymo-orchitis. Primary orchitis may result from infectious agents such as mumps. Epididymitis in older men usually results from a lower urinary tract infection with the common causative organisms being Escherichia coli, Pseudomonas, and Klebsiella. In younger men, organisms such as Chlamydia and Neisseria gonorrhoeae are more common etiologic agents. Rarely, tuberculosis may cause epididymo-orchitis. Mild repetitive trauma to the scrotum such as caused by riding a bicycle also may cause mild noninfective “mechanical” epididymo-orchitis.
Testicular abscess is usually a complication of epididymo-orchitis, although it may also result from undiagnosed testicular torsion, gangrenous or infected tumor, or primary pyogenic or outcome of primary orchitis. A testicular abscess may complicate into pyocele or a fistula to the skin.
Magnetic Resonance Imaging.
MRI is not used as a primary modality, but epididymo-orchitis may be diagnosed in suspected cases of torsion based on increased vascularity of the spermatic cord, epididymis, and testis.
Ultrasonography.
Ultrasound findings of acute epididymitis include diffuse or focal involvement with low echogenicity or, rarely, high echogenicity (if there is coexisting hemorrhage), and increased blood flow on color Doppler imaging. There is usually evidence of inflammation in the rest of the testis, as well in the form of generalized swelling and hyperemia ( Figure 78-4 ). Associated findings, such as reactive hydrocele or pyocele and scrotal wall edema, can further support the diagnosis. Testicular ischemia and infarction may occur when the vascularity of the testis is compromised by venous occlusion. Changes of chronic epididymo-orchitis include persistent swelling of the epididymis as a heterogeneous mass and a striated appearance of the testis.

Primary orchitis such as that secondary to mumps may have nonspecific findings and mimic tumor and transient torsion/detorsion. However, the presence of increased venous flow suggests orchitis, because intratesticular venous flow is usually difficult to detect in normal testes.
Testicular abscess is most commonly seen as an irregular, hypoechoic to anechoic mass with areas of mixed echogenicity. It is usually distinguished from tumors on the basis of clinical symptoms.
Testicular Trauma
The primary causes of scrotal trauma include blunt, penetrating, degloving, and thermal injuries. Blunt scrotal trauma is by far the most common cause of testicular injury and usually results from athletic injury, motor vehicle accident, or assault. The right testis is more commonly injured than the left. Testicular trauma may result in testicular hematoma, traumatic hydrocele or hematocele, testicular fracture, rupture, and infarction.
Magnetic Resonance Imaging.
MRI is generally not used to evaluate primary testicular trauma, although it may be performed for concomitant penile trauma.
Ultrasonography.
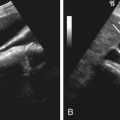
Ultrasonography is ideal for the assessment of scrotal trauma because it provides rapid and accurate assessment of scrotal contents and their integrity. Hematoma may be intratesticular or extratesticular and appear hyperechoic (acutely), heterogeneous (subacute), or predominantly hypoechoic (chronic) ( Figure 78-5 ). Without surgical exploration, intratesticular hematoma has a poor prognosis.

Hydrocele is commonly seen with trauma ( Figure 78-6 ). Simple hydroceles usually appear uniformly hypoechoic. Rupture of the bulbar urethra may result in leakage of urine into the scrotum, mimicking a hydrocele. Hematocele (blood within the tunica vaginalis) also may occur in trauma. Heterogeneity of the testicular parenchyma with associated hematocele suggests testicular rupture.

A testicular fracture is a break in the continuity of the testicular parenchyma with an intact tunica albuginea. A testicular rupture involves discontinuity of the tunica albuginea with extrusion of testicular parenchymal contents into the scrotal sac. Testicular rupture necessitates emergent surgery, whereas testicular fracture with preserved vascularity may be managed conservatively. Testicular fracture without preserved vascularity also necessitates emergent surgery, owing to the presence of testicular ischemia. Heterogeneous testicular parenchymal echotexture, with focal hyperechoic or hypoechoic areas, corresponds to areas of hemorrhage or infarction ( Figure 78-7 ). Contour abnormality is the single most important predictor of testicular rupture.

Hydrocele, Hematocele, and Pyocele
Hydrocele is the most common cause of unilateral or bilateral diffuse painless scrotal enlargement. Congenital hydrocele results from a patent processus vaginalis resulting in open communication between the scrotal sac and peritoneum. It usually resolves by 18 months of age. Acquired hydroceles are the result of trauma, epididymitis, and torsion. Blood, pus, or urine may accumulate in the tunica vaginalis spaces. Hematoceles and pyoceles are less common than simple hydroceles.
Magnetic Resonance Imaging.
MRI is not generally used to evaluate hydroceles, hematoceles, or pyoceles.
Ultrasonography.
Hydroceles are characteristically anechoic collections with good sound transmission anterolateral to the testis because of the attachment of the testis to the epididymis and scrotal wall posteriorly. Low-level echoes from fibrin bodies or cholesterol crystals may occasionally be visualized ( Figure 78-8 ). Both hematoceles and pyoceles are complex fluid collections that may contain multiple internal septa and debris (see Figure 78-6 ).

Nuclear Medicine.
Although radionuclide studies were traditionally used to diagnose hydrocele, this is no longer done.
Varicocele
A varicocele is a collection of abnormally dilated, tortuous, and elongated veins of the pampiniform plexus located posterior to the testis, accompanying the epididymis and the vas deferens within the spermatic cord. It can manifest as pain and/or swelling or may be detected incidentally during a workup for infertility. The cause of varicocele is unclear. Idiopathic varicoceles are thought to be due to incompetent valves in the testicular vein that permit retrograde passage of blood through the spermatic cord into the pampiniform plexus. Idiopathic varicoceles are more common on the left side where the testicular vein enters the left renal vein at a perpendicular angle. The right testicular vein enters obliquely into the inferior vena cava, and this appears to have some protective effect on the right side. Idiopathic varicoceles are bilateral in up to 70% of cases.
Secondary varicocele is uncommon. It is caused by compression of the renal vein by tumor or can result from an aberrant or obstructed renal vein. Because varicoceles are much less common on the right side, the finding of a right-sided varicocele in the absence of a left-sided varicocele should prompt further investigation to exclude an associated abdominal mass causing compressive symptoms. This also applies to older patients with a recent-onset varicocele. Secondary varicocele on the left may result from “nutcracker syndrome,” in which the superior mesenteric artery compresses the left renal vein.
Radiography.
Although venography is still considered to be the gold standard, it is time consuming and invasive. A normal venogram is one in which a single testicular vein is seen up to the inguinal ligament and into the spermatic cord. If a varicocele is present, the internal spermatic vein will be enlarged and there will be reflux into the abdominal, inguinal, scrotal, or pelvic portions of the spermatic vein. There also will be venous collateralization and formation of anastomotic channels. Venography is now most commonly performed before definitive treatment with venous embolization.
Computed Tomography.
CT may be used to evaluate for abdominal and retroperitoneal masses as a cause of varicocele.
Magnetic Resonance Imaging.
MR venography may be used when conventional venography is contraindicated or to demonstrate recurrent varicoceles after surgery.
Ultrasonography.
Ultrasonography is the primary modality for evaluation of varicoceles. The veins of the pampiniform plexus normally range from 0.5 to 1.5 mm in diameter, with a main draining vein up to 2 mm in diameter. On ultrasound evaluation, a varicocele consists of multiple, serpentine, anechoic structures more than 3 mm in diameter, creating a tortuous collection located adjacent or proximal to the upper pole of the testis and the head of the epididymis. Performance of a Valsalva maneuver or examination with color Doppler imaging in the upright position is important because it causes an increase in vessel size and some varicoceles will only then become apparent ( Figure 78-9 ). Rarely, varicoceles may be intratesticular, either in a subcapsular location or around the mediastinum testis.

Benign Cystic Scrotal Lesions
Cystic testicular lesions are usually benign and detected incidentally. Malignant testicular lesions can rarely show cystic degeneration secondary to hemorrhage or necrosis. An abnormal rind of parenchyma with increased echogenicity usually surrounds malignant cysts and provides a clue to the diagnosis. The common benign cystic scrotal lesions that can be characterized with ultrasonography include tunica albuginea cysts, intratesticular simple cysts, tubular ectasia of the rete testis, spermatocele, and epidermoid cysts.
Magnetic Resonance Imaging.
Simple cysts of the testis, epididymis, and tunica should have the same imaging features as a cyst elsewhere: low T1 and high T2 signal, imperceptible walls, and no enhancement.
Epidermoid cysts are nonenhancing, with variable signal intensity on T1-weighted images. They are usually of high signal intensity on T2-weighted images, sometimes with internal foci of low signal intensity. There is a low signal intensity rim on T1- and T2-weighted sequences. The low-intensity outer rim is believed to be due to the outer fibrous capsule and adjacent compact keratin, whereas central hypointensity is believed to be due to dense central debris and rarely calcification. Tubular ectasia of the rete testis has characteristic MRI features, but MRI is rarely needed because the ultrasound appearance is usually diagnostic. The lesion is uniformly of low intensity on T1-weighted sequences and isointense to hyperintense to testicular parenchyma on T2-weighted images ( Figure 78-10 ). This lesion does not enhance after administration of gadolinium.

Ultrasonography.
Cysts of the tunica albuginea are located within the tunica. The mean age at presentation is approximately 40 years, and frequently patients may present with clinically palpable firm scrotal nodules, although they may be asymptomatic and incidentally discovered. They vary in size from 2 to 30 mm and are usually well defined, solitary, and unilocular ( Figure 78-11 ) but may be multilocular. Complex tunica albuginea cysts may simulate a testicular neoplasm.

Simple testicular cysts are rare and usually detected incidentally in men 40 years of age or older. They are typically filled with serous fluid and may range from 2 to 18 mm. On ultrasonography, they are well-defined, anechoic with thin smooth walls and posterior acoustic enhancement. They are usually located near the mediastinum testis, suggesting that they may originate from the rete testis. Simple cysts usually require no treatment.
Epidermoid cysts represent 1% of all testicular tumors. They range from 1 to 3 cm in diameter, and most commonly manifest during the second to fourth decades with a painless testicular nodule or incidentally. The ultrasound appearance varies with maturity, compactness, and quantity of keratin present within the cyst. A target appearance, a solid mass with an echogenic rim, and a characteristic “onion ring” configuration with alternating layers of hyperechogenicity and hypoechogenicity have been described ( Figure 78-12 ). These cysts are usually avascular. When an epidermoid cyst is suspected, testicular-sparing surgery by enucleation may be pursued rather than orchiectomy. MRI may be helpful in further characterizing the lesion preoperatively.