Chapter 9 Breast Implants and the Reconstructed Breast
Implant Types
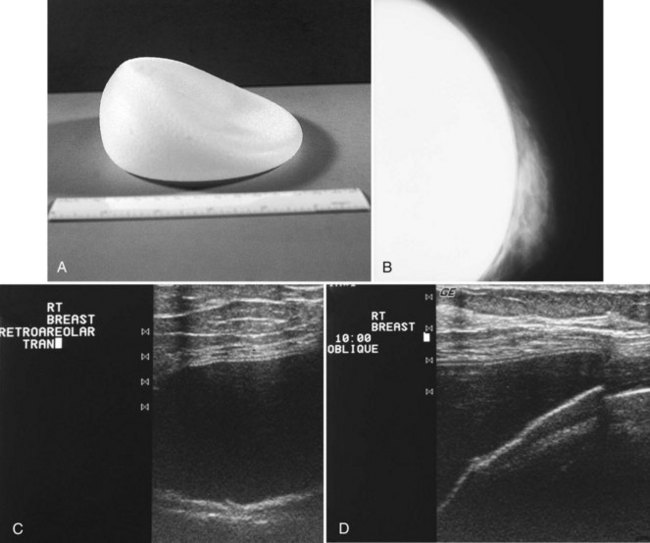
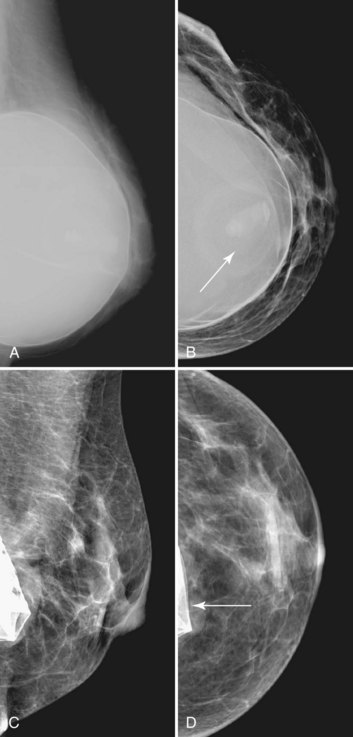
The most common breast implants are single-lumen and filled with either silicone or saline. Silicone implants are composed of a silicone elastomer shell filled with silicone made from a synthetic polymer of cross-linked chains of dimethylsiloxane that makes the implant soft and movable (Box 9-1 and Fig. 9-1). The outer envelope can be textured or smooth, polyurethane-coated or uncoated. The inner silicone can be a gel, a liquid, or a solid form. Saline implants are composed of an outer silicone shell and an inner envelope filled with saline (Fig. 9-2A to D). Of note, newer generation silicone implants have had a very small rupture rate in augmentation patients. Double- or triple-lumen implants have two or more envelopes inside one another, and each can contain saline or silicone gel. A common double-lumen implant is the saline outer, silicone inner implant. More recent common double-lumen implants have silicone outer and saline inner components. All implants are placed behind the breast tissue, and some implants are placed behind the pectoralis muscle.
Mammography of Normal Implants
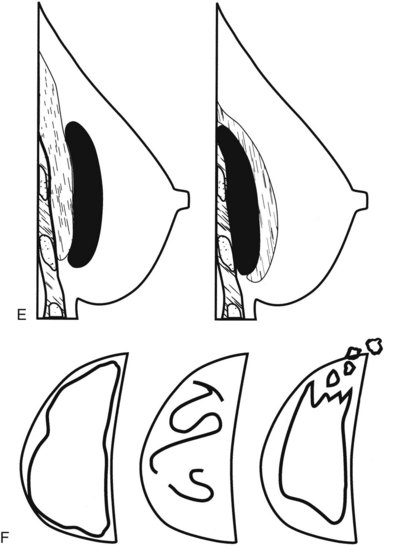
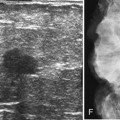
Surgeons place silicone gel-filled implants behind the breast tissue on the chest wall in the subglandular or subpectoral position (see Fig. 9-2E and F). In either case, the body generally forms a fibrous capsule around the implant. The fibrous capsule is usually soft, nonpalpable, and undetectable to physical examination, but with time, the capsule hardens or calcifies in some individuals. If a silicone implant ruptures, this fibrous capsule holds the silicone within as long as it, too, does not rupture.
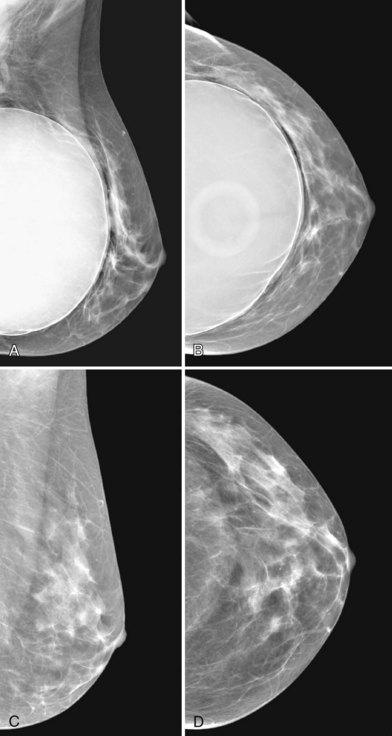
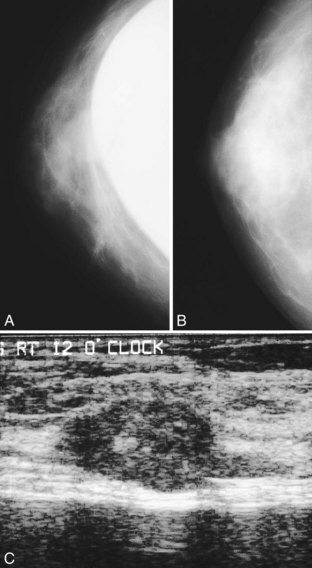
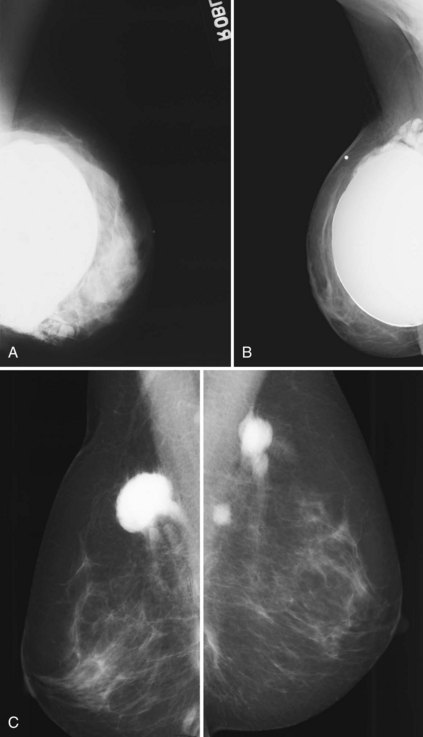
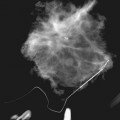
A normal silicone implant is quite dense and completely opaque, and obscures and displaces much of the surrounding breast tissue. On mammography the implant appears as a smooth white oval opacity near the chest wall (Fig. 9-3). The pectoralis muscle curves over the implant in subpectoral implants and lies underneath the implant in subglandular implants. A silicone gel-filled implant is not as compressible as breast tissue and can be ruptured if compressed too hard during mammography or closed capsulotomy. Because limited compression decreases visualization of the surrounding breast tissue for breast cancer screening, the Mammography Quality Standards Act (MQSA) recommends four views of each implanted breast. Two views are implant-displaced views, in which the technologist pinches the breast tissue in front of the implant to compress it, and two views include the implant but do not use much compression (Box 9-2). Breast tissue is evaluated on the implant-displaced views; implant integrity is evaluated on limited-compression mammograms in which the implant is surrounded by noncompressed breast tissue. Even with the implant-displaced views, the radiologist sees only about 80% of the breast tissue because it is hidden by the implant.
Box 9-2 Mammography of Implants
CC, craniocaudal; ML, mediolateral; MLO, mediolateral oblique.
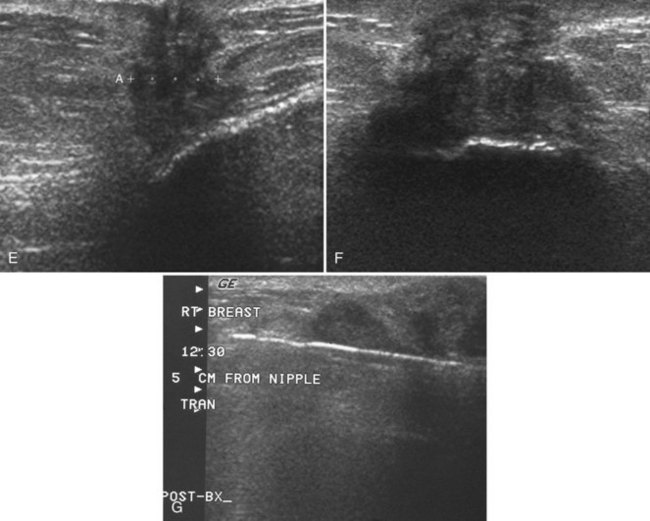
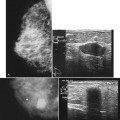
The implant-displaced technique does not completely resolve the problem of imaging small breast cancers, but it does optimize the amount of breast tissue displayed on the mammogram. Both physical examination and breast ultrasound as an adjunct to mammography are helpful in evaluating mammographically detected breast masses or palpable findings because mammography is limited with implants in place. Ultrasound can be especially helpful in determining whether a true mass exists, because ultrasound can evaluate the entire breadth of the breast tissue down to the implant. Ultrasound also distinguishes breast masses from the snowstorm appearance of silicone granulomas caused by ruptured implants (Fig. 9-4).
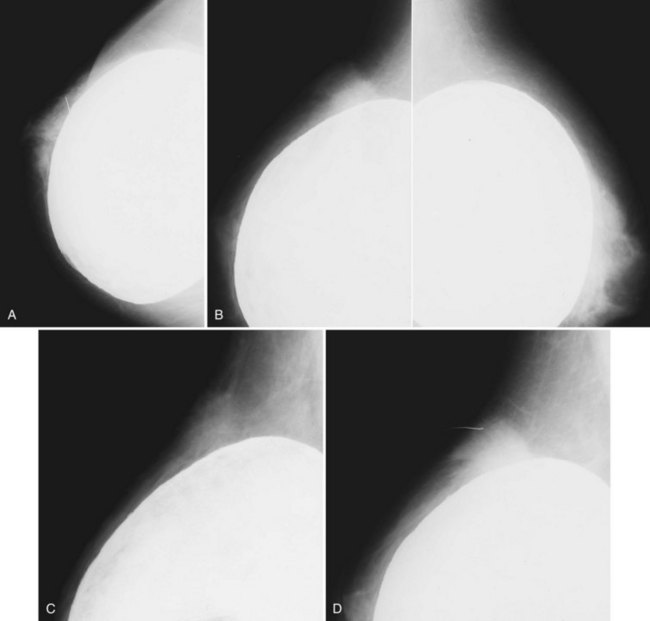
Unlike opaque silicone implants, saline implants contain radiolucent saline surrounded by a dense silicone outer envelope, in which small wrinkles may be seen. When a saline implant ruptures, the saline diffuses into the breast tissue and the envelope shrinks back against the chest wall (Fig. 9-5). In contradistinction, when a silicone implant ruptures, most of the silicone may be contained by the fibrous capsule and the implant retains much of its shape and volume. Saline outer, silicone inner double-lumen implants have an outer envelope containing saline surrounding a dense inner silicone implant filling.
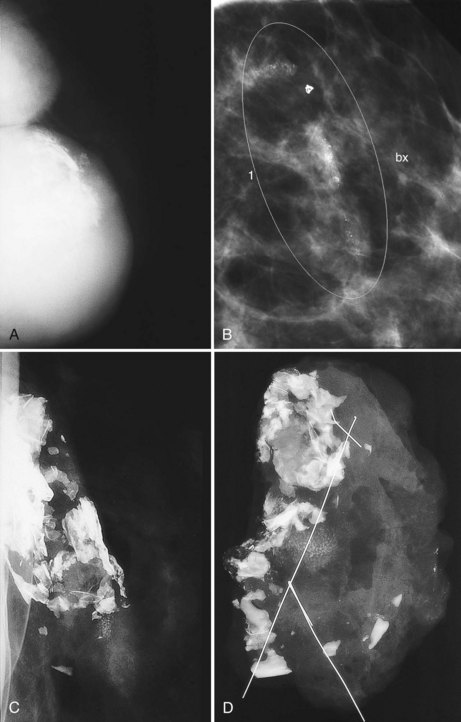
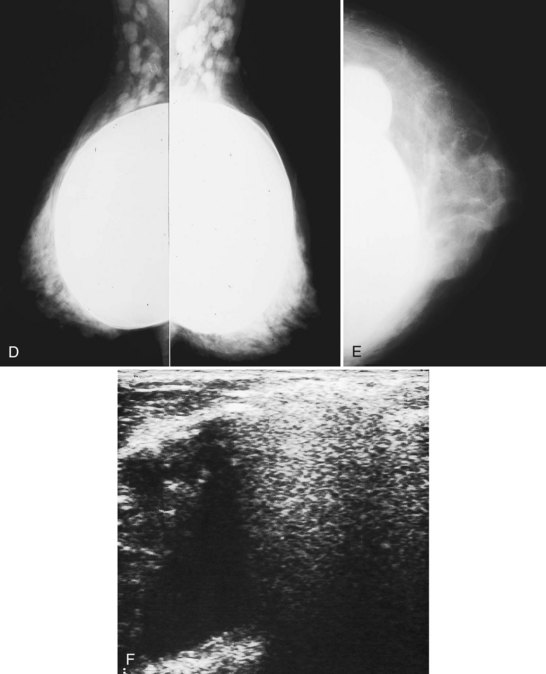
The fibrous capsule surrounding implants of any type is not usually visible unless it calcifies. A calcified fibrous capsule contains dystrophic sheetlike calcifications and appears bumpy on the nondisplaced-implant views (Fig. 9-6A). Implant-displaced views displace the capsular calcifications away from the implant for analysis if one is concerned that the calcifications are in breast parenchyma rather than in the implant capsule. Spot magnification mammograms also help the radiologist analyze intraparenchymal calcifications and distinguish them from capsular calcifications.
If an implant ruptures, the surgeon removes the ruptured implant but does not always remove the fibrous capsule. If the fibrous capsule has calcified, the mammogram shows dystrophic calcifications in a sheetlike curvilinear pattern because they reside in the retained fibrous capsule. These calcifications can be hard to distinguish from cancer and can prompt biopsy (see Fig. 9-6B). Another specific type of capsular calcification that can be mistaken for cancer and will sometimes prompt biopsy is from calcifying polyurethane-covered implants. These implants are covered with a spongelike material, and when they calcify produce a typical fine meshlike calcification that can mimic ductal carcinoma in situ (see Fig. 9-6C and D).
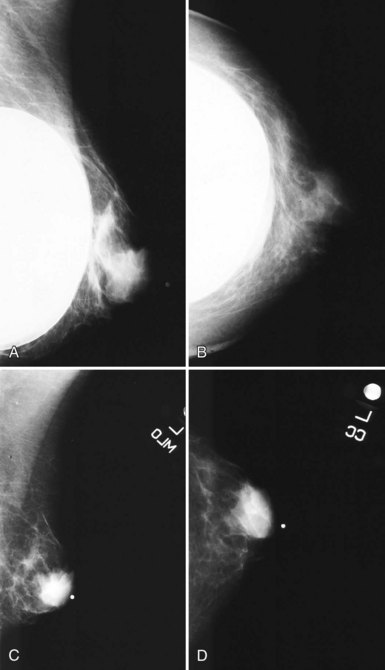
When evaluating breast tissue for cancer in women with implants, it is important to inspect both the implant-displaced views and the breast tissue adjacent to the implant on the nonimplant-displaced views. The standard views may display a mass near the implant or the fibrous capsule not evident on implant-displaced views. Masses on the fibrous capsule can be pushed away from the field of view with the implant-displaced views (Fig. 9-7). However, for masses or suspicious calcifications, spot compression or other fine-detail views can be used in women with implants, just as with any other woman. Needle localization, ultrasound-guided core biopsy, and stereotactic core biopsy all can be performed in women with implants as well. The radiologist just has to obtain informed consent from the woman, including implant rupture as a possible complication for percutaneous biopsies.
Implant Complications and Rupture
Untoward complications associated with silicone gel-filled breast implants include contracture of the fibrous capsule, calcification of the fibrous capsule, hematoma, infection, implant rupture, and the controversial silicone gel “bleed,” in which silicone gel leaks outside the implant through an intact envelope (Box 9-3). Capsular contracture is the most common complication, with a reported incidence of more than 70% in some older series and only about a 20% incidence in more recent series.
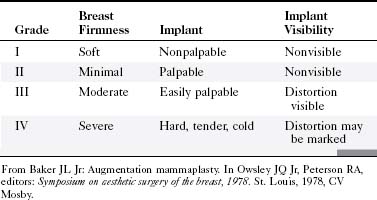
Implants can undergo capsular contraction, becoming hard and resistant, leading to a round, hard appearance and feel. The Baker classification of capsular formation on implants describes increasing levels of capsular contracture (Table 9-1). The incidence of capsular contracture may be diminished by the use of textured submuscular implants, although the use of such implants remains controversial. Open surgical capsulotomy, in which the hardened implant capsule is removed, was used to solve the problem of capsular contracture. Alternatively, surgeons would squeeze the implant to break the hardened fibrous capsule to allow the implant to become soft and pliable again, called closed capsulotomy. Unfortunately, closed capsulotomy could result in implant rupture.
Different types of fillers for implants were examined in trials, resulting in varying rates of fibrous capsule contracture. Munker and colleagues reported on Trilucent implants in 27 patients who elected to exchange their implants for a fourth-generation cohesive silicone implant. Of these 27 patients, 14 had a change in the volume of their implants but not all were aware of the change, and capsular contracture was not present (Baker grade II) (see Table 9-1); 55% of the implants had thickening or color changes caused by peroxidation of the triglyceride contents, and the implant capsule was adherent to breast tissues—in particular, the pectoralis muscle, which led to prolonged operative times. Rizkalla and colleagues reported similar results, with a reoperation rate of 20% (10/50) and an implant deflation rate of 10% (5/50). The Medical Devices Agency in the United Kingdom (which merged with the Medicines Control Agency in April 2003 to form the Medicines and Healthcare Products Regulatory Agency) withdrew the Trilucent implant from the market in March 1999, with a subsequent recommendation in June 2000 that the implants be removed from patients. A new type of alloplastic material for implants that contains carboxymethylcellulose, called Hydrogel, was introduced into the European market. Of 12 patients with 20 implants placed between 1996 and 1997, as reported by Cicchetti and colleagues, none showed immediate complications and had Baker grade I or II capsular contracture at 3.5 years of follow-up.
Implant Rupture
Implant integrity is classified as intact, intact with gel bleed, intracapsular rupture, or extracapsular rupture (Table 9-2). Extracapsular rupture is defined as implant rupture with silicone gel extruded outside a broken fibrous capsule. Intracapsular rupture is defined as implant rupture with silicone gel still contained within an intact fibrous capsule. Gel bleed is defined as silicone gel leakage through an intact implant envelope, although the existence of gel bleed versus small, undetected ruptures remains controversial.
Table 9-2 Implant Rupture Types
| Rupture Types | Silicone Location | Envelope Status |
|---|---|---|
| Intracapsular rupture | Fibrous capsule contains silicone gel | Envelope ruptured |
| Extracapsular rupture | Silicone gel outside fibrous capsule | Envelope ruptured |
| Gel bleed (controversial) | Silicone outside envelope | Envelope intact |
Implant Imaging
Mammography
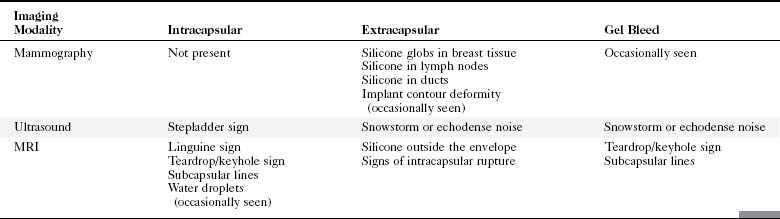
A retrospective review of screening mammograms in 350 asymptomatic women with breast implants showed an incidence of asymptomatic implant rupture of 5%. Mammography shows extracapsular rupture as silicone extravasation outside the implant envelope with blobs of silicone in the breast tissue (Fig. 9-8A and B), within implant ducts, or as a contour abnormality caused by extruded silicone in contiguity with the implant (Table 9-3). After extracapsular rupture, the surgeon removes the implant. Removal of all extravasated silicone is often impossible without removing much of the breast tissue, so the surgeon may leave some extravasated silicone in the breast. Later, when a new silicone implant is placed, residual silicone from the old ruptured implant makes it impossible to tell on mammography if the new implant has ruptured (see Fig. 9-8C). Silicone within axillary lymph nodes implies extravasation of silicone outside the fibrous capsule, because the silicone has traveled to the lymph nodes. This means that there must be an extracapsular rupture as well (see Fig. 9-8D).
Silicone implant contour lobulation indicates either capsular contracture, herniation of an intact implant envelope through a break in the surrounding fibrous capsule, or a contained implant leak (Table 9-4). Because intracapsular rupture is defined as implant rupture with silicone gel still contained within an intact fibrous capsule, mammography may show an intracapsular rupture as a normal-looking or bulging implant, depending on the shape of the fibrous capsule. Because both implant lobulation and a contained leak have the same mammographic appearance, radiologists use ultrasound and MRI to make the diagnosis of a rupture (see Fig. 9-8E and F). Mammography cannot identify intracapsular ruptures when the implant contour is normal, nor can it show posterior implant ruptures on the chest wall.
Table 9-4 False-Positive Imaging Findings for Rupture
| Imaging Modality | Sign | Differential Diagnosis |
|---|---|---|
| Mammography | Implant contour deformity | |
| Intraparenchymal silicone | Previous leak with the ruptured implant removed | |
| Ultrasound | Stepladder sign | |
| MRI |
Direct silicone or paraffin injections were used overseas for breast augmentation; free silicone, paraffin, or other materials were injected directly into the breast tissue. The injections are foreign bodies and therefore result in multiple tiny round eggshell-type calcifications that obscure the underlying breast tissue. Because these silicone or injection granulomas may become quite hard, both physical examination and mammography of the underlying tissue are nearly impossible (Fig. 9-9). Ultrasound of patients with silicone injections shows multiple areas of snowstorm or echodense noise that cast shadows throughout the breast and obscure the breast tissue, thus rendering evaluation for breast cancer difficult.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree