Hepatocellular carcinoma (HCC) is a leading cause of morbidity and mortality, ranking fifth for men and eighth for woman as source of primary malignancy.1,2 The highest incidences of HCC are found in sub-Saharan Africa and Eastern Asia, with age-adjusted incidence rates of 17.43 and 6.77 per 100,000 in men and women of developing countries, compared to 8.71 and 2.86 per 100,000 in men and women of developed regions of the world.1 However, the incidence of HCC in developed countries, including the United States, has risen significantly in the past 2 decades, in part attributed to the increased prevalence of hepatitis B and C during this same period.3 Establishing generalizable standards of practice for treatment in HCC is confounded by wide variations in prognostic features present at diagnosis, severity of underlying liver disease, and access to medical services around the world. Three curative options of resection, liver transplant, and ablation exist as first-line treatment modalities for early HCC, achieving 5-year survival rates of 50% to 70%.4 Arterial embolization techniques are primary therapy for more advanced stages of HCC in patients who are nonsurgical candidates. At these advanced stages of HCC, chemoembolization has been shown to be an effective palliative therapy that can also improve patient survival.5,6 Chemoembolization combines the effects of targeted ischemia with high local chemotherapeutic drug concentration and prolonged drug dwell time in the tumor. Hepatic cancers receive blood primarily from the hepatic artery, in contrast to normal liver, which derives the majority of its blood flow from the portal circulation.7,8 This is the basis for targeted chemoembolization of tumor cells via the hepatic artery. Lipiodol, a brand of iodized esters of poppyseed oil used as a contrast agent, is selectively retained within tumor cells and serves as the delivery vehicle of the chemotherapy agents. Particle embolization after infusion of the chemotherapy emulsion increases the dwell time of the cytotoxic agents by slowing the rate of their efflux from the hepatic circulation. In addition, ischemia causes cell death directly and potentiates intracellular drug uptake by impeding the function of metabolically active cell membrane pumps that normally function to clear cytotoxic chemicals from the cytosol. Because the liver metabolizes most of the chemotherapeutic agents, systemic toxicity is reduced. Chemoembolization is the most widely used primary treatment for unresectable HCC, with the best outcomes in patients with reasonably preserved liver function (Child-Pugh class A-B [Table 64-1]), Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 1 (Table 64-2), and without macrovascular invasion or extrahepatic spread.9,10 TABLE 64-1 Child-Pugh Classification of Liver Disease TABLE 64-2 ECOG, Eastern Cooperative Oncology Group. From Tuite CM, Sun W, and Soulen MC. General assessment of the patient with cancer for the interventional oncologist. J Vasc Interv Radiol 2006;17:753–8. There is an additional neoadjuvant role for chemoembolization as a therapy for those awaiting liver transplantation. Neoadjuvant treatment may decrease the risk of tumor progression outside of criteria that would preclude liver transplantation.5,11,12 The only absolute contraindication to chemoembolization is decompensated baseline liver function. Patients at high risk of acute liver failure include those with greater than 50% replacement of liver by tumor, a lactate dehydrogenase level above 425 IU/L, aspartate aminotransferase (AST) level above 100 IU/L, and total bilirubin level above 2 mg/dL.13 Relative contraindications include those unlikely to benefit from therapy due to poor prognosis, such as widely metastatic disease, Child-Pugh class C liver disease, or decompensated performance status (ECOG 3-4). Patients at risk for liver failure include those with markedly elevated bilirubin, hepatic encephalopathy, transjugular intrahepatic portosystemic shunt (TIPS), main portal vein occlusion, or hepatofugal portal blood flow.6 In these settings, segmental or subsegmental chemoembolization may be performed. Chemoembolization in the setting of biliary stenting, sphincterotomy, or bilioenteric anastomosis increases the risk of abscess formation.14 Portal vein thrombosis can be managed by adjustment of the embolization protocol to limit the degree of embolization and distribution of chemotherapeutic agent15 or by documenting sufficient hepatic collateral flow is present to compensate for lack of portal flow.16 Contraindications to the angiographic procedure include anaphylactoid contrast media allergy, renal insufficiency, uncorrectable coagulopathy, and peripheral vascular disease preventing establishment of arterial access. Contraindications to chemotherapy such as severe cytopenias, renal insufficiency, and cardiac dysfunction may preclude treatment.6,17 • Fluoroscopic unit capable of digital subtraction angiography (DSA) of sufficient quality to carefully evaluate the visceral vasculature, preferably equipped with cone-beam computed tomography (CT) capability • A 4F hydrophilic Cobra catheter and hydrophilic guidewire suffice for about half of procedures. • Standard reverse-curve visceral catheters (Simmons, SOS, Rosch) • Microcatheters 105 to 130 cm in length with 0.025- to 0.027-inch inner lumen are specifically designed for chemoembolization. • 0.014- to 0.018-inch guidewires for use with microcatheters
Chemoembolization for Hepatocellular Carcinoma
Clinical Relevance
Indications
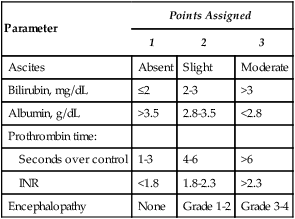
Parameter
Points Assigned
1
2
3
Ascites
Absent
Slight
Moderate
Bilirubin, mg/dL
≤2
2-3
>3
Albumin, g/dL
>3.5
2.8-3.5
<2.8
Prothrombin time:
Seconds over control
1-3
4-6
>6
INR
<1.8
1.8-2.3
>2.3
Encephalopathy
None
Grade 1-2
Grade 3-4
Grade
Description
0
Fully active, able to carry on all predisease performance without restriction
1
Restricted in physically strenuous activity but ambulatory and able to carry out work of a light or sedentary nature
2
Ambulatory and capable of self-care but unable to carry out any activities; up and about > 50% of waking hours
3
Capable of only limited self-care, confined to bed or chair > 50% of waking hours
4
Completely disabled; cannot carry on any self-care; totally confined to bed or chair
5
Dead
Contraindications
Equipment
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Chemoembolization for Hepatocellular Carcinoma






