Abstract
Chorionic villus sampling (CVS) is a procedure in which small samples of placental tissue are obtained for prenatal diagnosis of aneuploidy or for deoxyribonucleic acid analysis. CVS is an outpatient procedure usually performed between 10 weeks’ and 14 weeks’ gestation. The main advantage of CVS compared with amniocentesis is that the results are available earlier in pregnancy, allowing for earlier and safer methods of pregnancy termination if abnormal results are detected. This chapter provides a detailed discussion regarding the indications, risks, technique, and complications associated with this procedure.
Keywords
CVS, chorionic villus sampling, genetics, placenta
Introduction
Chorionic villus sampling (CVS) is a procedure in which small samples of placental tissue are obtained for prenatal diagnosis of aneuploidy or for DNA analysis. CVS is an outpatient procedure usually performed between 10 and 14 weeks’ gestation. The main advantage of CVS compared with amniocentesis is that the results are available earlier in pregnancy, allowing for earlier and safer methods of pregnancy termination if abnormal results are detected.
Procedure
Description, Technique, and Equipment
Before CVS, the patient should be counseled regarding the purpose, risks, and alternatives to the procedure, and informed consent should be obtained. Transvaginal ultrasound (US) should be performed to determine the number of embryos, chorionicity, fetal viability, and any early US evidence of fetal anomalies.
CVS can be performed transabdominally or transcervically ( Figs. 113.1 and 113.2 ). No differences in fetal loss rates were noted by Jackson et al. after transcervical versus transabdominal CVS. The procedure should be performed with US by a physician who is experienced in performing the latter. A sonographer who is trained to recognize the US appearance of a transabdominal needle and a transcervical catheter ( Fig. 113.3 ) is crucial for a successful procedure.
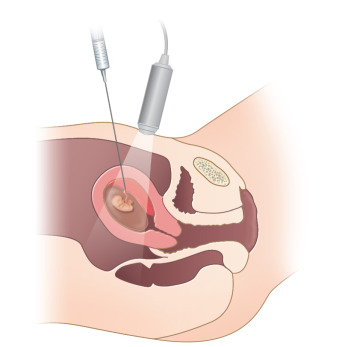
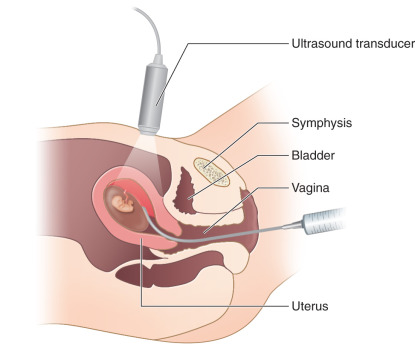
Transcervical Chorionic Villus Sampling
Before transcervical CVS, obtaining cervicovaginal cultures is recommended and pathogens should be appropriately treated. The supplies needed for the procedure include antiseptic solution, 4-inch × 4-inch gauze pads, a sterile speculum, a single-toothed tenaculum or ring forceps (optional), a 26-cm-long polyethylene cannula with an outer diameter of 1.5 mm and a soft stainless steel blunt stylet (alternatively, biopsy forceps may be used ), a 20-mL syringe containing nutrient medium, a tissue culture dish or specimen tube, sterile US gel, and US probe cover ( Fig. 113.4 ).
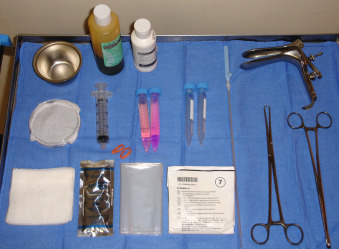
The patient is placed in the lithotomy position, and the speculum is inserted into the vagina. When the cervix can be seen, the vagina and cervix are prepared with antiseptic solution. Some operators use either a single-toothed tenaculum or ring forceps to grasp the anterior lip of the cervix to manipulate the cervix and uterine position gently; others perform the procedure without these instruments. A full bladder may facilitate the ideal angle between the anterior uterine wall and the endocervical canal. The transcervical catheter can be bent to mimic the curve of the endocervical canal noted on the US scan (a uterine sound can be placed in the cervical canal to help with visualization before catheter placement).
While the sonographer provides direct transabdominal US guidance, the physician can carefully insert the transcervical catheter through the endocervical canal and into the placenta. The stylet is removed, and the 20-mL syringe containing several milliliters of nutrient medium is attached to the transcervical catheter. Using negative pressure, the catheter is gently moved back and forth within the placenta to aspirate placental villi . While the syringe is under negative pressure, the catheter is removed, and the sample is placed into a plastic tissue culture dish or specimen tube. The physician examines the sample under direct light or a microscope to ensure that adequate placental villi were obtained.
Transabdominal Chorionic Villus Sampling
Supplies needed for the procedure include antiseptic solution, 4-inch × 4-inch gauze pads, a sterile drape, nutrient medium, tissue culture dish or specimen tube, sterile US gel, US probe cover, a 9-cm-long or 12-cm-long 20-gauge spinal needle with stylet, and a 20-mL syringe and holder to mount the syringe ( Fig. 113.5 ).