Cirrhosis
Etiology
Virtually any chronic insult to the liver, if sufficiently severe and long-standing, may result in cirrhosis. In the United States, the most common causes are hepatitis C virus (HCV) infection and alcohol ingestion, whereas in Asia and sub-Saharan Africa, chronic hepatitis B virus (HBV) infection is the most frequent culprit. Nonalcoholic fatty liver disease (NAFLD) is increasing in prevalence and is now the third most common cause of cirrhosis in North America and in parts of Europe and South America. Other common causes in adults include nonviral infections (mainly, parasitic [e.g., schistosomiasis]), autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, genetic disorders (hemochromatosis, Wilson’s disease, alpha-1 antitrypsin deficiency, and glycogen storage diseases), medications (e.g., amiodarone and methotrexate), and veno-occlusive disease (especially Budd-Chiari syndrome; sinusoidal obstruction syndrome generally does not cause cirrhosis). Cryptogenic cirrhosis describes cirrhosis of unknown cause; once a frequent diagnosis, the term has become less common in the past 2 decades with the discovery of HCV and the recognition of NAFLD.
The fundamental pathogenic mechanism of cirrhosis is that of injury and repair. Hepatocytes may be injured by a variety of etiologic factors, initiating an inflammatory response. Atypical, dysfunctional hepatocytes aggressively multiply to counteract the effects of hepatocyte injury, forming regenerative nodules. Isolated insults cause self-limited inflammation, and the liver’s wound-healing mechanisms successfully repair the damage. Repetitive or chronic injuries, however, overwhelm these restorative processes, leading to the dysregulated production of cytokines (e.g., transforming growth factor beta) and activation of profibrogenic perisinusoidal stellate cells. The end result is the unchecked deposition of excess macromolecules and collagen in the extracellular matrix (fibrosis).
Fibrosis initially occurs in the space of Disse, where it obliterates normal endothelial fenestrations. Further scar deposition in the perisinusoidal and sinusoidal spaces obstructs sinusoidal blood flow and causes portal hypertension. Because of the elevated intrahepatic resistance, portal venous blood is shunted to systemic veins with lower pressures and returned to the heart without passing through the liver. The microvascular architecture of the liver remodels in response. Abnormal connections develop between the portal vein and terminal hepatic vein, which contribute to the shunting of blood from the hepatic parenchyma. As a result of portal hypertension and microvascular hepatic alterations, varices form, ascites develops, and hypersplenism occurs. Toxins normally metabolized and eliminated by the liver accumulate in the blood and contribute to the development of hepatic encephalopathy.
Loss of hepatocytes affect hepatic synthetic function, resulting in reduced levels of essential proteins (e.g., blood clotting factors and albumin) and causing coagulation abnormalities as well as reduced intravascular oncotic pressure. Atypical hepatocytes have impaired ability to secrete bile into canaliculi, which not only causes intestinal fat and vitamin malabsorption and steatorrhea but also results in cholestasis (accumulation of bile within hepatocytes), which may perpetuate hepatocellular damage. Because the liver is required for the normal metabolism and supply of glucose and lipids to the rest of the body, hepatocellular dysfunction leads to muscle breakdown, inefficient mobilization of energy stores, and redistribution of adipose tissue. Clearance of estrogens may be reduced, leading to gynecomastia in males.
Systemically, patients with cirrhosis exhibit a hyperdynamic circulation as a result of decreased systemic vascular resistance and peripheral vasodilation, mediated by nitric oxide among other factors. End consequences include increased cardiac output, splanchnic vasodilation, renal vasoconstriction and hypoperfusion, and salt and water retention. Additionally, the intestinal mucosa becomes hyperpermeable, perhaps related to reduced oncotic pressure; this may account for bacterial translocation from the gut to the peritoneal space, resulting in spontaneous bacterial peritonitis.
Prevalence and Epidemiology
The prevalence of cirrhosis is difficult to ascertain and is probably underestimated because large numbers of patients with compensated cirrhosis go undetected. This is especially the case in patients with cirrhosis resulting from NAFLD or HCV infection, conditions with asymptomatic phases of long duration. In the United States, the prevalence of cirrhosis is estimated to be 0.27%, which would represent 633,323 American adults based on 2010 U.S. Census data. However, cirrhosis is undoubtedly more frequent in Africa and Asia, where vertically transmitted HBV infection is common, although precise estimates are unavailable.
Age, sex, and ethnicity all influence the risk for cirrhosis. Cirrhosis rarely afflicts children (<18 years) or the elderly (>75 years), occurring 80% of the time in patients between the ages of 25 to 64. Sixty-five percent of cases occur in males. Although there is little variation in the prevalence of cirrhosis among different ethnicities, there are differences with regard to cirrhosis-related death. In 2001, chronic liver disease with cirrhosis was the 12th leading cause of death in the United States; however, it was the 6th and 7th most frequent cause of death in Native Americans and Hispanics, respectively.
Clinical Presentation
Cirrhosis is often indolent and unsuspected until its complications arise. Some asymptomatic patients present for the evaluation of unrelated problems and are incidentally found to have cirrhosis based on physical examination, laboratory findings, or imaging. Owing to the large number of abnormalities in liver function caused by cirrhosis and the central role the liver plays in whole-body physiology, symptomatic patients may present with a broad spectrum of clinical manifestations.
Common manifesting symptoms of compensated cirrhosis include fatigue, weakness, anorexia, jaundice, itching, and easy bruising. On physical examination, patients may exhibit muscular and temporal wasting, ecchymoses, and jaundice. Dermatologic stigmata of chronic liver disease, such as spider angiomas and palmar erythema, are present in 33% of cases. Gynecomastia occurs in the majority of male patients. Hypogonadism, secondary to either primary gonadal injury or disruption of the hypothalamic-pituitary axis, results in testicular atrophy, impotence, infertility, and feminization. The liver itself may be enlarged (characteristically observed in alcoholic and nonalcoholic fatty liver disease, storage diseases, and Budd-Chiari syndrome) or shrunken with firm edges (characteristically observed in viral causes). Portal hypertension frequently causes splenomegaly and caput medusae.
With hepatic decompensation, more advanced signs and symptoms may manifest. Ascites, the most common complication of cirrhosis, occurs as a result of portal hypertension and salt and water retention. Over 60% of patients with cirrhosis develop ascites within 10 years of diagnosis; and once ascites occurs, 2-year survival is only approximately 50%. Spontaneous bacterial peritonitis is a life-threatening complication of ascites and is one of the reasons for the high ascites-related fatality.
Portosystemic collaterals, or varices, develop as a result of portal hypertension. The most clinically significant varices are located in the distal esophagus and upper stomach. These esophagogastric varices occur in approximately 60% of patients with cirrhosis, with 30% of them experiencing bleeding complications.
Hepatic encephalopathy refers to a spectrum of neuropsychiatric abnormalities caused by the retention of ammonia and other toxic substances normally metabolized and cleared by the liver. Precipitating factors include dehydration, electrolyte and metabolic derangements, gastrointestinal bleeding, infections, and sedating medications. Transjugular intrahepatic portosystemic shunt (TIPS) and surgical shunt procedures hasten the development of hepatic encephalopathy in approximately 40% of patients.
Hepatorenal syndrome refers to acute renal failure in patients with cirrhosis without underlying renal abnormalities. It is thought to occur because of splanchnic vasodilatation, which results in unremitting, hormone-mediated renovascular vasoconstriction and reduced renal perfusion. Occurring in approximately 40% of patients with cirrhosis and ascites by 5 years, hepatorenal syndrome is a terminal complication of cirrhosis and has a poor prognosis.
In the majority of patients, the diagnosis of cirrhosis is based on history, physical examination, basic laboratory studies, and imaging studies. In general, serum aminotransferase levels are mildly elevated (generally, <100 units/L), the total bilirubin level is proportionally elevated, and the albumin value is reduced. Because the liver synthesizes many of the blood clotting factors, the prothrombin time may be prolonged. Hyponatremia, secondary to impaired free water secretion, and renal insufficiency are seen in advanced cirrhosis and are poor prognostic signs. Serum alpha-fetoprotein levels are sometimes elevated in patients presenting with cirrhosis complicated by hepatocellular carcinoma (HCC).
Once cirrhosis has been diagnosed, the severity historically has been graded by the Child-Turcotte-Pugh classification (e.g., Child class A, B, or C), with higher point totals coinciding with greater short-term and perioperative fatality. More recently, the Model for End-Stage Liver Disease (MELD) score has gained preference. This score is calculated from a logarithmic formula using the patient’s total bilirubin, international normalized ratio, and creatinine concentration.
The natural history of cirrhosis is that of an inexorably progressive condition. Within 10 years, 58% of patients with cirrhosis develop various forms of hepatic decompensation. The annual incidence of HCC in patients with cirrhosis is approximately 5% but ranges from less than 1% up to 10%, depending on the cause of cirrhosis and presence of comorbidities, such as human immunodeficiency virus infection or diabetes, which may increase the hepatocarcinogenesis risk.
Pathology
Cirrhosis is a diffuse process affecting the architecture of the entire liver; localized scarring does not constitute cirrhosis. In advanced cirrhosis, it is not possible to determine the cause with certainty. However, in early-stage cirrhosis, different hepatic insults may cause varied patterns of cirrhosis.
Gross examination of the cirrhotic liver typically reveals a shrunken and firm organ, although the liver may be enlarged depending on the cause. Cirrhosis is defined histologically by the presence of fibrous septa that divide the liver parenchyma into nodules. The septa range from delicate fibrous bands to large fibrous tracts that obliterate multiple lobules. The fibrous septa may connect portal tracts and centrilobular hepatic terminal veins in portal-portal, portal-central, or central-central patterns. The nodules are variably sized and arbitrarily defined as micronodules (<3 mm) or macronodules (>3 mm).
By far, the most common nodules are regenerative (RNs). These tend to be relatively uniform in size and appearance. Occasionally, nodules may arise that are conspicuous in terms of size, color, texture, or degree of bulging from the cut surface of the liver. These may represent low- or high-grade dysplastic nodules (DNs) or HCC. Low-grade DNs have been alternatively referred to as large RNs and macroregenerative nodules. The hepatocytes in low-grade DNs are essentially indistinguishable from background hepatocytes, although subtle variations in cell size may be seen. High-grade DNs are distinguished microscopically from low-grade DNs via recognition of cytologic alterations (increased nuclear to cytoplasmic ratio) and architectural changes (thickening of hepatocyte cell plates and pseudo-gland formation). Grossly, high-grade DNs frequently show a “nodule-in-nodule” appearance, reflecting patchy proliferative activity. Low-grade DNs are considered benign, whereas high-grade DNs are thought to be precursors of HCC.
Recently, focal nodular hyperplasia (FNH)-like lesions have been described in cirrhosis. Both macroscopically and microscopically, these lesions appear identical to FNH in the noncirrhotic liver. Unlike true FNH, which is thought to arise in response to congenital arteriovenous malformations, FNH-like lesions are thought to arise from acquired vascular flow perturbations associated with cirrhosis.
Imaging
Imaging studies, including ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI), are used to assess liver size, the biliary tree, patency of hepatic vasculature, and sequelae of portal hypertension (e.g., ascites, varices, and splenomegaly) and to screen for HCC. Although the portosystemic pressure gradient may be directly measured via transjugular cannulation of the hepatic veins, this is invasive and often unnecessary because portal hypertension can be inferred by endoscopic findings of varices and sequelae of portal hypertension seen on imaging.
On cross-sectional imaging, the cirrhotic liver may demonstrate a nodular surface, widened fissures between lobes, and an increase in size of the hypertrophied caudate lobe relative to the atrophied right lobe. The ratio of caudate lobe hypertrophy to right lobe atrophy has most recently been defined by the modified ratio of caudate to right lobe, in which the lobes are divided anatomically by the bifurcation of the right portal vein. A ratio greater than 0.65 predicts cirrhosis with high specificity and positive predictive value. Depending on the modality and imaging technique, fibrotic reticulations, fatty changes, and the presence of various hepatocellular nodules (RNs, DNs, HCC, FNH-like lesions) may be visible. The critical diagnostic distinction is between malignant nodules (e.g., HCC) and nonmalignant nodules (e.g., RNs, DNs, and FNH-like lesions). Although DNs have higher rates of malignant transformation than RNs, the transformation rate is not sufficiently high to warrant ablation or other intervention, nor to increase imaging surveillance frequency or alter surveillance strategies. The natural history of FNH-like lesions in cirrhosis is unknown, but these lesions are considered benign. Simple hepatic cysts and hemangiomas are observed less frequently in the cirrhotic liver than in the noncirrhotic liver. These lesions are described elsewhere in this text. Peribiliary cysts are serous cysts that are hypothesized to represent obstructed periductal glands in patients who have severe liver disease. Recognition of these cysts on imaging helps radiologists to avoid the incorrect diagnosis of dilated bile ducts, abscesses, or cystic neoplasms.
Classic vascular manifestations of cirrhosis include hepatic artery dilatation, tortuosity of hepatic arteries within the liver (“corkscrew” arteries, which may be observed with high-resolution CT and MRI), portal vein dilation in early portal hypertension, portal vein occlusion in late portal hypertension as a result of sluggish portal flow, and formation of shunts. Intrahepatic shunts (arterioportal and arteriovenous) may manifest on dynamic imaging studies as small (<2 cm) hypervascular pseudo-lesions and may be mistaken for nodules. Portosystemic shunts manifest as varices (e.g., esophagogastric and paraumbilical); these are usually located outside the liver, although intrahepatic varices may also occur. In addition to varices, other sequelae of portal hypertension (e.g., ascites, splenomegaly, peribiliary cysts, and Gamna-Gandy bodies) may be noted ( Figures 41-1 to 41-3 ).



Computed Tomography
Technical Considerations.
The primary indications for performing multiphasic CT in patients with cirrhosis are the evaluation of disease progression, surveillance for HCC, and follow-up of known lesions. Contrast enhancement of vessels, liver, and other solid organs may be impaired in patients with cirrhosis owing to third spacing of fluid and leakage of contrast material into the pulmonary interstitium during passage through the right-sided circulation. For this reason, patients with cirrhosis may require higher rates and concentrations of contrast material than patients without cirrhosis. The critical phases for image acquisition are the late arterial (typically at 35 to 40 seconds), portal venous (at 60 to 80 seconds), and equilibrium (at 3 to 5 minutes). Unenhanced images usually are recommended as cirrhotic nodules that may be intrinsically hyperdense because of copper or iron deposition or high glycogen content. These nodules may thus appear hyperdense at hepatic arterial phase and be mistakenly characterized as hypervascular if unenhanced images are not acquired first. Some authors also advocate early arterial phase images (typically at 20 to 25 seconds) to detect very early enhancement of malignant lesions and permit more precise characterization of lesion enhancement features, but this strategy has not been proved to be superior for lesion diagnosis in large clinical trials and increases the radiation dose. CT hepatic artery angiography, CT portography, and CT after transarterial administration of iodized oil may be performed in select cases but are not used routinely in the assessment of cirrhotic patients; their discussion is beyond the scope of this chapter.
Fibrosis.
On unenhanced images, the attenuation of normal liver is typically approximately 10 Hounsfield units greater than that of the spleen. However, the density of the cirrhotic liver or of focal lesions may be reduced (e.g., by steatosis, fibrosis, or edema) or increased (e.g., by iron or copper deposition). Fibrosis typically is not visible on unenhanced CT; if sufficiently severe, fibrosis may manifest as a diffuse lacework of hypoattenuating bands or as mottled areas of decreased density. Regions of confluent fibrosis are characterized as hypoattenuating wedge-shaped or geographically shaped regions radiating from the portal hilus and causing retraction of the overlying hepatic capsule ( Figure 41-4 ). Involvement of the medial segment of the left lobe or anterior segment of the right lobe is characteristic, but other segments may be involved as well. Confluent fibrosis is observed more commonly in alcoholic liver disease and primary sclerosing cholangitis than in viral and other liver diseases. On contrast-enhanced CT, fibrosis, especially if confluent, may show progressive enhancement and appear hyperattenuating on delayed images. Confluent fibrosis occasionally may be masslike and cause diagnostic confusion. Differentiation from HCC is usually possible based on the characteristic capsular retraction, volume loss, and progressive enhancement pattern associated with confluent fibrosis. In difficult cases, follow-up imaging may be necessary; progressive volume loss, if observed, clinches the diagnosis of confluent fibrosis.

Nodules.
FNH-like lesions are small, ranging up to approximately 1 cm in diameter. Isodense on unenhanced imaging, these lesions hyperenhance on the arterial phase and then fade to isoattenuation on more delayed images. On both unenhanced and contrast-enhanced CT, RNs usually are isodense relative to the surrounding hepatic parenchyma and are difficult to visualize ( Figures 41-5 and 41-6 ), although siderotic RNs may be hyperdense. On unenhanced CT, large DNs are hyperattenuating relative to surrounding parenchyma because of the presence of increased iron and glycogen, whereas small DNs remain isodense. Conversely, DNs enhance simultaneously with normal parenchyma on contrast-enhanced CT, appearing isodense. With increased dedifferentiation, however, these DNs may appear hyperattenuating because of increased vascularity and consequent increased contrast uptake.


HCCs may have a varied appearance at CT depending on tumor size, vascularity, steatosis, cholestasis, hemorrhage, and necrosis. On unenhanced CT, HCCs generally appear as hypoattenuating or heterogeneously attenuating lesions. Intralesional fat or blood products may be difficult to identify on CT; these features are more readily observed on MRI. After administration of contrast agents, HCCs become hyperattenuating or heterogeneously enhancing during the arterial phase, then wash out to hypoattenuation on venous and delayed phases. If a tumor capsule is present, it often enhances progressively and retains contrast material on delayed images. Vascular invasion, if present, is more easily appreciated on contrast-enhanced images.
Perfusional pseudo-lesions secondary to arteriovenous or arterioportal shunts enhance during the arterial phase and then fade to isoattenuation on images acquired during the portal venous and equilibrium phases. As opposed to true nodules, pseudo-lesions tend to have ill-defined or straight borders and may have blood vessels running through their center. In diagnostically challenging cases, longitudinal imaging is necessary. At follow-up, pseudo-lesions regress or are stable but rarely grow.
Magnetic Resonance Imaging
Technical Considerations.
MRI provides the best performance characteristics for the diagnosis of cirrhosis but requires longer scan times than CT and is more costly. Controversy exists regarding which MR technique and contrast agent is optimal for evaluation of cirrhotic patients. For T1-weighted imaging, dual-phase in-phase and out-of-phase gradient echo images are commonly acquired because these permit assessment of parenchymal and lesional fat content in addition to characterizing T1 relaxation properties. T2-weighted fast spin echo imaging is useful for evaluating bile ducts, cysts, and fluid collections; single-shot fast spin echo sequences are particularly useful for this purpose. T2-weighted imaging also helps characterize RNs and DNs but has low sensitivity for detecting HCC. On diffusion weighted imaging, HCCs may have restricted diffusion and appear bright on diffusion weighted images and dark on corresponding apparent diffusion coefficient maps.
The most common dynamic technique to evaluate the liver is MRI after a rapid bolus injection of gadolinium-based contrast. Volumetric three-dimensional T1-weighted, fat-saturated spoiled gradient echo acquisitions are usually used for dynamic imaging. The key phases for extracellular contrast agents are the hepatic arterial-dominant phase (in which the acquisition of the center of k space coincides with peak arterial perfusion of hepatic nodules), portal venous phase (acquired 60 to 80 seconds after gadolinium injection [see Figure 41-6 ]), and equilibrium phase (acquired 3 to 5 minutes after injection). For purposes of HCC screening and evaluating response to locoregional therapy (i.e., tumor ablation, transcatheter arterial chemoembolization), extracellular agents such as Magnevist are typically preferred because of their high gadolinium concentration and mechanism of action, which includes an equilibrium phase to detect tumor washout. Hepatobiliary agents such as Eovist can be helpful in distinguishing true masses from perfusional pseudo-lesions, because the latter will not appear as a hypointense filling defect on 20-minute delayed imaging.
Superparamagnetic iron oxides (SPIOs) may be given to evaluate phagocytic function of hepatic Kupffer cells. The agent is administered as a slow infusion, typically over 30 minutes. Uptake of SPIOs results in T2 and T2* shortening. Lesions with Kupffer cells (most RNs and DNs) lose signal intensity on SPIO-enhanced T2-weighted and T2*-weighted images, whereas lesions deficient in Kupffer cells (most HCCs and cirrhotic scars) do not lose signal intensity and appear relatively hyperintense. There is controversy regarding whether spin echo or gradient echo techniques are most well suited for evaluating SPIO uptake. In select centers, double-contrast–enhanced imaging, in which SPIO followed by gadolinium are administered sequentially in the same examination, is performed. The sequential administration of two agents theoretically improves the characterization of cirrhotic nodules by permitting the assessment of two complementary biological features (phagocytic activity of Kupffer cells and vascularity).
Fibrosis.
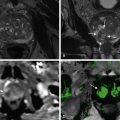
Fibrosis in the cirrhotic liver has low signal intensity on unenhanced T1-weighted images and high signal on T2-weighted images. Similar to its appearance on CT, confluent fibrosis tends to have straight borders and is associated with progressive volume loss over time at follow-up imaging. When gadolinium is administered, fibrotic tissue slowly enhances in the arterial phase and retains contrast on portal venous and delayed images. As a result, fibrosis has relatively high signal on delayed gadolinium-enhanced images (see Figures 41-4 and 41-5 ). Fibrosis, which lacks Kupffer cells, also has relatively high signal intensity on T2-weighted and T2*-weighted images after SPIO administration. Double-contrast–enhanced gradient echo images show the reticulations and bands of liver fibrosis to greatest advantage; the fibrotic tissue appears hyperintense as a result of gadolinium accumulation, while the background liver parenchyma appears hypointense because of SPIO accumulation. Such images also show other features of cirrhosis, including capsular thickening ( Figure 41-7 ) and nonuniform distribution of fibrosis ( Figure 41-8 ).


Nodules.
FNH-like lesions are typically isointense on T1-weighted and isointense to hypointense on T2-weighted imaging. Similar to their appearance on contrast-enhanced CT, these lesions enhance during the hepatic arterial phase after injection of gadolinium-based agents and then fade to isointensity on more delayed phases. Like FNH, they also take up Eovist and appear isointense to parenchyma on the hepatocyte phase.
RNs have a variable appearance at unenhanced T1-weighted imaging and may be hypointense, isointense, or hyperintense. Some RNs are steatotic and lose signal intensity on out-of-phase compared with in-phase imaging ( Figure 41-9 ). Most RNs are isointense and not detectable at T2-weighted imaging, whereas some RNs, particularly those with high iron concentration, are hypointense at T2-weighted and T2*-weighted imaging. High signal intensity on T2-weighted images is distinctly unusual for RNs; marked T2 hyperintensity suggests a simple cyst, whereas mild to moderate T2 hyperintensity raises suspicion for HCC. On administration of gadolinium, RNs enhance to a degree similar to that of surrounding parenchyma and usually appear isointense. However, both RNs and DNs can demonstrate arterial enhancement but then fade to isointensity on delayed imaging (rather than washing out). RNs and DNs typically take up hepatobiliary contrast agents and appear isointense at 20 minutes. When SPIOs are administered, RNs take up the iron particles and, as a result of superparamagnetic effects, appear hypointense on T2-weighted and T2*-weighted images (see Figure 41-5 ). DNs, like RNs, have variable signal intensity on unenhanced T1-weighted images and appear isointense or hypointense on T2-weighted images. On gadolinium and SPIO-enhanced imaging, low-grade DNs typically are difficult to distinguish from RNs, whereas high-grade dysplastic nodules may resemble well-differentiated HCCs.

HCCs display variable signal intensity on unenhanced T1-weighted imaging. They characteristically appear hyperintense at T2-weighted imaging, but T2-weighted imaging has limited sensitivity for HCCs and may render them invisible or mildly hypointense. After administration of gadolinium, hypervascular HCCs enhance rapidly to high signal intensity on arterial phase images and then become hypointense on portal venous and delayed images. When Eovist is administered, HCCs almost always appear hypointense on the hepatocyte phase. Rarely, well-differentiated HCCs can take up and excrete hepatobiliary agents. Compared with RNs and DNs, HCCs tend to have a reduced concentration of Kupffer cells and diminished phagocytic capacity. Hence, when SPIOs are administered, HCCs do not take up the particles and have high signal intensity relative to the adjacent non-neoplastic parenchyma. Focal areas of fibrosis also have high signal on such images and may be mistaken for HCCs on low-resolution SPIO-enhanced images. High-resolution SPIO-enhanced imaging usually permits differentiation of fibrosis (reticulated morphology) from HCC (nodular morphology). In challenging cases, gadolinium administration or follow-up imaging is necessary.
Perfusional pseudo-lesions may be indistinguishable from FNH-like lesions. Similar to FNH-like lesions, the pseudo-lesions hyperenhance during the arterial phase and then fade to isointensity. Both will typically appear isointense at delayed hepatobiliary imaging. Differentiation is not clinically important, however, because both FNH-like lesions and perfusional pseudo-lesions are benign. If these entities are suspected, follow-up imaging rather than intervention is recommended.
Ultrasound
Technical Considerations.
Because of its low cost and lack of radiation, ultrasonography is typically the initial imaging modality performed in patients with cirrhosis. It provides information about liver size and shape and, if high-frequency transducers are used, may detect subtle nodularity along the liver surface to establish the diagnosis of cirrhosis in clinically equivocal cases. In patients with established cirrhosis, ultrasound plays an important role in assessing sequelae of portal hypertension (e.g., splenomegaly and ascites). On gray-scale images, features suggestive of portal hypertension include dilation of the portal vein to greater than 13 mm, splenic vein to greater than 11 mm, and superior mesenteric vein to greater than 12 mm. Color Doppler tracings can be used to further characterize portal vein patency and direction of flow. Portal hypertension is associated with increased pulsatility of the portal vein Doppler tracing and loss of the normal triphasic hepatic vein Doppler tracing. When portal flow is hepatofugal, the liver has progressed to end-stage disease and shunt placement or liver transplantation may be required. Color Doppler tracings can also be used to monitor shunt patency as well as patency of vascular anastomoses after transplantation.
In Europe, Asia, and select centers in North America, microbubble-based contrast agents may be administered intravenously to assess lesion vascularity using ultrasonography. Similar to CT and MRI, multiple phases are acquired: arterial phase (15 to 30 seconds after injection), portal phase (30 to 60 seconds after injection), and sinusoidal (or blood pool) phase (60 to 240 seconds after injection).
Nodules.
Ultrasound has limited sensitivity for cirrhosis-associated nodules. Discrete RNs are rarely identified even if they are obvious on other imaging modalities. Coarsening and heterogeneity of the liver echotexture may suggest the presence of RNs, but this finding is neither sensitive nor specific, and the liver parenchyma may appear normal even if cirrhosis is advanced (see Figures 41-4 and 41-10 ). Another indirect clue to the presence of RNs includes nodularity of the liver surface, most reliably appreciated if high-resolution transducers are used. In this setting, the RNs themselves are usually not visible and their presence is inferred by the bulging of the liver capsule.