Introduction
Over the past few decades, the management of lumbosacral spinal disease has become notoriously difficult, whereby complex biomechanics and heterogeneity in presenting conditions have challenged treating clinicians. In response, classification systems emerged, leading to expanded characterization of disease and improvements in treatment guidelines. Though the earliest classifications of the lumbosacral region were simply descriptive, advances in knowledge and technology have since led to systems resulting in measurable improvements in patient outcomes. Over time, the introduction of imaging techniques, such as plain radiographic films and tomography followed by CT, MRI, angiography, and myelography led to major advancements in taxonomic development. These advancements led to the identification of various phenotypes or measurements that could be used to categorize lumbosacral disease and injury, assess boney anomalies, disc vitality, and alignment of the lumbosacral spine in relation to adjacent vertebrae, discs, and soft tissues. This chapter aims to highlight lumbosacral classifications with emphasis on measurement, phenotypic variation, and the clinical relevance of the various systems proposed.
Current Classifications
Alignment (Spinal Curvature)
Adult spinal deformity
Adult spinal deformity (ASD) has conventionally been defined as aberrant curvature or alignment of the spinal vertebrae. While the vast majority of spinal deformity falls into the category of degenerative scoliosis, there are frequent incidences of ASD in patients with persistent adolescent idiopathic scoliosis. As such, increased precedence is being placed on examining other, atypical deformities as novel research continues to unravel the complex, heterogeneous nature of the various disorders that fall within the diagnosis. In 2006, the SRS and Schwab classifications were developed to address such issues and have subsequently been combined in routine clinical use. The SRS-Schwab classification leverages full-spine (free-standing) radiographs to assess the hip joints and femoral heads in relation to a patient’s underlying deformity. In 2008, Kuntz described the CKIV Classification, which utilized some of the parameters in the SRS classification, such as thoracic kyphosis (TK) and lumbar lordosis (LL), but placed precedence on developing an age-dependent system that was further subcategorized by abnormal characteristics (location, severity, pattern) as well as overall global spinal alignment ( Table 1 ). Finally, in 2012, the SRS-Schwab classification was modified to include updated pelvic parameters, such as pelvic tilt (PT), pelvic incidence-lumbar lordosis (PI-LL) mismatch, and Cobb angle, as these factors had been previously correlated with clinical outcomes, such as disability, pain, and quality of life scores ( Table 2 ).
| Patient age (year) |
| Infantile (0–2) |
| Juvenile (3–9) |
| Adolescent (10–18) |
| Adult (19–60) |
| Geriatric (> 60) |
| Spinal abnormality |
| Scoliotic, kyphotic, lordotic, mixed deformity curves |
| Major structural deformity curve standing deformity curve with greatest deviation from age-appropriate NUSA for 98.5% of the asymptomatic population |
| Scoliotic deformity curves |
| Scoliotic major structural deformity curve > age-appropriate NUSA for 98.5% of the population |
| Minor structural scoliotic curves remain > 25 degrees on side-bending radiographs |
| Scoliotic curves named for curve apex in spinal zones |
| Location (disc) |
| Occipitocervical: O-C2 |
| Cervical: C2/C3 disc-C6/C7 disc |
| Cervicothoracic: C7-T1 |
| Proximal thoracic: T1/T2 disc-T5 |
| Main thoracic: T5/T6 disc-T11/T12 disc |
| Thoracolumbar: T12-L1 |
| Lumbar: L1/L2 disc: L4/L5 disc |
| Lumbosacral: L5-S1 |
| Kyphotic and lordotic deformity curves |
| Kyphotic major structural deformity curve > age-appropriate NUSA mean + 2.5 SD |
| Lordotic major structural deformity curve < age-appropriate NUSA mean − 2.5 SD |
| Minor structural kyphotic curves remain > adult NUSA mean + 1 SD on extension radiographs |
| Minor structural lordotic curves remain < adult NUSA mean − 1 SD on flexion radiographs |
| Kyphotic and lordotic curves named for Sagittal angle in spinal zones |
| Location (Vertebra) |
| Occipitocervical: O-C2 |
| Cervical: C2-C7 |
| Cervicothoracic: C6-T2 |
| Proximal thoracic: T1-T5 |
| Main thoracic: T4-T12 |
| Thoracolumbar: T10-L2 |
| Lumbar: L1-L5 |
| Lumbosacral: L4-S1 |
| Scoliokyphotic and scoliolordotic deformity curves |
| Structural scoliotic curve plus structural kyphotic curve in the same zone |
| Structural scoliotic curve plus structural lordotic curve in the same zone |
| Coronal imbalance |
| ± Coronal imbalance, C7-S1 CVA (greater or less than age-appropriate NUSA mean ± 2.5 SD) |
| Sagittal imbalance |
| ± Sagittal imbalance, C7-S1 SVA (greater or less than age-appropriate NUSA mean ± 2.5 SD) |
| Pelvic alignment, neutral |
| Coronal rotation PO > adult NUSA mean ± 2.5 SD |
| Sagittal rotation PT > or < adult NUSA mean ± 2.5 SD |
| Primary curve types |
| Single thoracic (ST) |
| Double thoracic (DT) |
| Double major (DM) |
| Triple major (TM) |
| Thoracolumbar (TL) |
| Lumbar “de novo”/idiopathic (L) |
| Primary sagittal plane deformity (SP) |
| Adult spinal deformity modifiers |
| Regional sagittal modifier (include only if outside normal range as listed) |
| (PT) Proximal thoracic (T2-T5): ≥+20 degrees |
| (MT) Main thoracic (T5-T12): ≥+50 degrees |
| (TL) Thoracolumbar (T10-L2): ≥+20 degrees |
| (L) Lumbar (T12-S1): ≥−40 degrees |
| Lumbar degenerative modifier (include only if present) |
| (DDD) 2 disc height and facet arthropathy based on X-ray include the lowest involved level between L1 and S1 |
| (LIS) Listhesis (rotational, lateral antero, retro) ≥ 3 mm includes the lowest level between L1 and L5 |
| (JCT) Junctional L5-S1 curve ≥ 10 degrees (intersection angle superior endplates L5 and S1) |
| Global balance modifier (include only if imbalance present) |
| (SB) Sagittal C7 plumb ≥ 5 cm anterior or posterior to sacral promontory |
| (CB) Coronal C7 plumb ≥ 3 cm right or left of CSVL |
| SRS definition of regions |
| Thoracic: apex T2-T11-T12 disc |
| Thoracolumbar: apex T12-L1 |
| Lumbar: apex L1-L2 disc-L4 |
| Criteria for specific major curve types |
| Thoracic curves |
| • Curve ≥ 40 degrees |
| • Apical vertebral body lateral to C7 plumbline |
| • T1 rib or clavicle angle ≥ 10 degrees upper thoracic curves |
| Thoracolumbar and lumbar curves |
| • Curve ≥ 30 degrees |
| • Apical vertebral body lateral to CSVL |
| Primary sagittal plane deformity |
| • No major coronal curve |
| • One or more regional sagittal measurements (PT, MT, TL, L) outside |
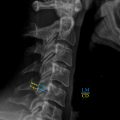
The currently used classification systems allow for an understanding of the impact of disease, mostly by leveraging radiographic findings, yet fail to accurately prognosticate the likelihood of complication rates. The most commonly utilized system is the SRS classification, and while it provides pertinent information regarding previously unconsidered factors (pelvic tilt, incidence, etc.) it fails to take into consideration other potentially relevant factors (comorbidities, treatment, alignment, and patient goals). Perhaps the most commonly utilized system is the pelvic incidence/lumbar lordosis (PI-LL) mismatch calculation, which was described by the creators of the Schwab classification ( Fig. 1 ). The PI-LL mismatch is a quick, simple calculation that is commonly applied for evaluation and preoperative planning. Furthermore, the PI-LL mismatch is a diagnostic marker in other pathologies, such as adjacent segment disease after lumbar fusion or pelvic dissociation. A PI-LL mismatch < 10 degrees has traditionally been associated with improved postoperative health-related quality of life scores, though some recent evidence may suggest that differences up to 20 degrees may lead to acceptable outcomes as well.

Recent literature indicates the need to reevaluate our current parameters to identify associations with perioperative complication rates in ASD patients. While studies, such as Lafage et al., have described age-specific ASD classification systems, there still is ample opportunity to improve upon the current ASD schemes, as some researchers believe the patients’ symptomology should have a larger role in their classification and treatment. The current heterogeneity in the described classification systems at the time of the writing of this chapter further serves as evidence supporting the need for a more comprehensive system.
Facet Joints
Zygapophysial (“facet”) joints are small articulations found along the entire length of the spine, providing stability and a restricted range of motion between adjacent vertebral segments. As such, with increasing age and cumulative biomechanical stress, the facet joints may undergo progressive degeneration and become a consistent source of back pain and disability. Facet joints located in the lumbar spine are perhaps most susceptible to such pathology, as corresponding vertebral segments and intervertebral discs experience greater flexibility, axial loads, and other concomitant degenerative changes. Further, facet joint pathology has been notoriously difficult to identify through routine history and physical examination, as frequently the cause of pain, disability, or radicular symptoms is multifaceted and may require invasive diagnostic procedures to identify. Such considerations have led to the development of numerous imaging-based classification systems, with hopes that specific facet phenotypes may best correlate with presenting symptoms and help identify which patients will respond appropriately to specific interventions.
Lumbar facet arthropathy
Lumbar facet arthropathy (LFA) is a form of osteoarthritis that occurs in the lumbar zygapophysial joints. LFA is often portrayed as a result of degenerative processes, such as excessive loading or abnormal movement patterns, but may occur as a result of trauma as well. Generally speaking, LFA occurs as the synovium in the facet joint begins to fail, leading to erosions, decreased disc space, and subchondral osteosclerosis. LFA presents most commonly at L4-L5, though all levels of the lumbar vertebral column may be involved, and often results in complaints of chronic low back pain or radiculopathy due to spinal cord compression. Three classification systems are currently utilized to describe lumbar facet arthropathy ( Tables 3–5 ).
| Grade | Description |
|---|---|
| 1 | Uniformly thick cartilage which covers the articular surface plus well-defined cartilage over articular processes |
| 2 | Cartilage covers the entire articular surface with evidence of irregular regions or erosion, interspace is irregular or noncrescentic |
| 3 | Cartilage incompletely covers the articular surfaces with underlying bone exposed |
| 4 | Cartilage is absent from the articular surface (may have trace amounts). Voids are evident with low MRI signals within the interspace |
| Grade | Criteria |
|---|---|
| 0 | Normal facet joint space (2 ± 4 mm width) |
| 1 | Narrowing of the facet joint space (< 2 mm) and/or small osteophytes and/or mild hypertrophy of the articular processes |
| 2 | Narrowing of the facet joint space and/or moderate osteophytes and/or moderate hypertrophy of the articular process and/or mild subarticular bone erosions |
| 3 | Narrowing of the facet joint space and/or large osteophytes and/or severe hypertrophy of the articular process and/or severe subarticular bone erosions and/or subchondral cysts |
| Grade | Description |
|---|---|
| 1 | Normal; no degeneration |
| 2 | Mild; joint space narrowing or mild osteophyte |
| 3 | Moderate; sclerosis or moderate osteophyte |
| 4 | Severe; marked osteophyte or subchondral cysts |
Despite expansive efforts concerning LFA classification schema development, there is no current consensus on any of these classifications. A study by Kettler et al. in 2006, which set kappa or ICC at > 0.40 (moderate agreement), was carried out to determine which system to recommend statistically for clinical use. Their efforts yielded two classifications that met their recommendation standards, namely the Pathria and Weishaupt systems. Additionally, they included the Grogan system, which is also utilized in clinical care, despite the that the classification did not meet study standards. As a whole, the agreement set on this study is low compared to the research community’s standards. The heterogeneity in classification systems as well as the lack of agreement at the time of the writing of this chapter necessitates further evaluation. Examination of which of the aforementioned systems correlates “better” with patient symptoms or clinical outcomes could be completed in a comparative analysis similar to that described by Kettler et al. in 2006.
Vertebral Endplate
Endplate degeneration
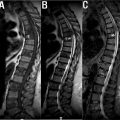
Modic changes (MC) are currently defined as signal changes in vertebral subchondral bone marrow present on magnetic resonance. The sentinel study by M.T. Modic et al., first described Modic changes in 1989 using T1-weighted sagittal MRI. These changes have been strongly associated with degenerative spinal pathologies, specifically degenerative disc disease. Type 1 MC has been correlated with low back pain (LBP) in numerous studies although its role in the pathogenies of LBP is under scrutiny. To date, three main types of MC have been delineated ( Fig. 2 ).

Currently, only Modic type 1 has been associated with low back pain in study populations, despite recent evidence the Modic type 2 changes may be more prevalent in individuals with greater fat mass. According to a study by Mitra et al., Modic changes represent a pathologic progression from acute (Modic type 1) to other chronic forms with Modic type 2 resulting in improvement of the patients’ symptoms. Overall, most hypotheses about the pathogenesis and role of Modic changes in degeneration are speculative, despite the classification’s widespread use in the research community. Continued analysis of the role of type 3 changes in spinal pathologies, with specific attention to the progression of degenerative etiologies, is necessary to elucidate the relationship with patient symptoms. Furthermore, it is important to note that recent studies have proposed a combined classification system that utilizes not only Modic changes, but Pfirrmann grading, high-intensity zones, and loss of disc height as well. Continued analysis of algorithmic approaches that leverage multiple classical scoring systems may provide better prognostic utility when evaluating the spine for degeneration.
Schmorl’s nodes
Schmorl’s nodes (SN) are a subclassification of herniation that results in the nucleus pulpous (NP) being pushed through the cartilage and endplate. In most cases, SN are asymptomatic findings on imaging modalities but may become painful over time. Despite numerous postulates as to the pathogenesis of SN (autoimmunity, trauma, degeneration, etc.), no single theory is currently accepted. Previous studies indicated SN is associated with the degeneration of the vertebral discs. With the wide variability of reported prevalence data, 3.8%–76%, precedence has been placed not only on examining the different etiologies of SN but on classifying it as well. Currently, at least one study has proposed a classification system. The classification is broken into five categories based on the contour, morphology, and typology of the lesion.
Despite continued research, such as that by Mok et al. which described the association between SN and disc degeneration, the clinical relevance of SN remains uncertain as the explanatory mechanism or pathogenesis to lower back pain has not been fully elucidated. Additionally, conflicting evidence about the progression of SN to disc degenerative symptomology exists, making prognostication inherently difficult. The initial results of the classification system described by Samartzis et al are promising with an excellent intraobserver (kappa = 0.88) and good interobserver reliability (mean kappa = 0.79). Despite the initial findings, it is important to note that the intraobserver reliability was calculated with a small observer sample size ( n = 2) and is a logical starting place for future analysis. Additionally, further studies investigating the prognostic utility and reliability of the aforementioned system as well as mechanistic evaluations of the pathophysiology of SN is necessary.
Intervertebral Discs
Annular fissure
Annular fissures are defined as a disruption in at least one layer of the annulus fibrous. Annular fissures occur not only between the annular fibers but from the vertebral column as well. Despite the typical asymptomatic nature of annular fissures, some may progress to simple annular fissures without herniation or herniated nucleus pulposus with radiculopathy depending on the severity and extent of the insult. Currently, fissures are classified by their involvement, location, and continuity ( Table 6 ).
| Grade | Description |
| 0 | No visible tears, normal nucleus |
| 1 | Tears are less than one-third of the distance through the annulus fibrosus |
| 2 | Tears extend further toward the disc edge, there is no presence of bulging, compression, or gross deformity |
| 3 | Tears have completely disrupted the disc and are encroaching on PLL |
The current classification for annular fissures, despite its descriptive value, fails in regard to prognostic usefulness. Despite the commonality of annular fissures, we cannot currently reliably identify them on MRI as well as grade or classify them based on clinical symptoms. Currently, multiple studies have explored the presence of high-intensity zones (HIZ) as a means of elucidating the existence or complexity of annular fissures, even though they can occur as a result of alternative pathological processes. Preliminary analysis indicates that the signal intensity on T2-weighted images has correlated with annular fissures, whereas the presence of intensity on a T1-weighted image appears to suggest different pathophysiology. Systematizing annular fissure MRI findings into a classification system has been suggested, but as of the writing of this chapter it has not been fabricated. It is the current recommendation of experts in the field to develop large-scale studies assessing the role of HIZ in annular fissure while cultivating a standardized classification system that utilizes the pathophysiologic progression of annular fissures.
Disc degeneration
Disc degeneration is one of the most common spine-related presentations. Degeneration is currently considered to be related to aggravating factors, such as surgery, metabolic injury, or trauma which leads to deteriorating processes (chondrocyte proliferation, cell death, dehydration, etc.) over time. As a result of degeneration, the spine may become more susceptible to injury and progress to unfavorable symptomologies such as developing chronic low back pain. The most commonly utilized classification systems for degeneration, Pfirrmann grades and Thompson Classification, will be examined in the subsequent sections.
Pfirrmann grading
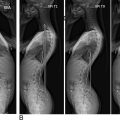
In 2001, Pfirrmann et al. created a scoring system for disc degeneration based on T2-weighted magnetic resonance findings. Specifically, the score aims to provide standardized nomenclature for describing disc abnormalities among spinal surgeons. The Pfirrmann classification system utilizes an algorithmic approach leveraging four criteria for describing disc degeneration, specifically structure, signal intensity, height, and the ability to distinguish the nucleus from the annulus. The scores are classified as one of the five grades depending on MRI findings such as disc height, intensity, and the distinction between anatomical landmarks ( Fig. 3 ).
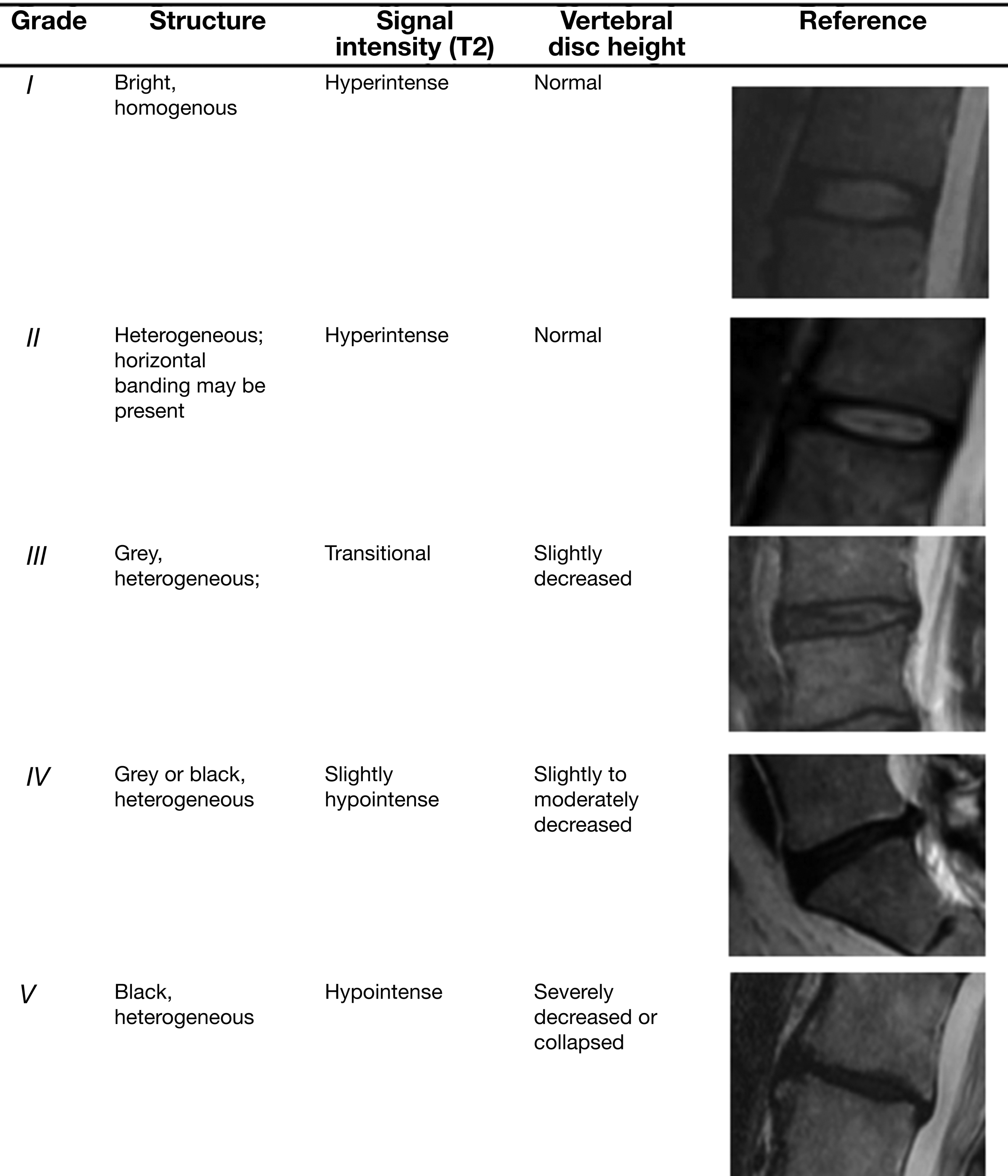
The strengths of the Pfirrmann system lie in its simplicity, completeness, and proven research utility. Despite demonstrated reliability, specifically, intrarater, of Pfirrmann grading the classification falls short as multiple limitations of the system have been considered. First, Pfirrmann grading has been described as being subjective. As such, some studies have found that the Pfirrmann system only provides acceptable agreement among different observers, which calls its communicational consistency into question. Furthermore, the Pfirrmann grading system has been under recent scrutiny for the lack of interobserver reliability, particularly when comparing disc degeneration between the elderly and younger patients. Additionally, some argue that the 5-level system should be abandoned in favor of an 8-level system that expands grade C to 2 grades (3 and 4) and Grade D to 3 grades (5, 6, and 7). This allows clinicians to more accurately take into account the effect of age on the degeneration of the spine. Finally, advancements in biomechanical and magnetic resonance imaging technologies, such as glycosaminoglycan chemical exchange saturation, micro-nano structural, and 9.4 T MRI, have let experts speculate that each could improve upon the currently utilized Pfirrmann system. Of note, decreased variability in scoring severity with the adoption of these technologies has been postulated.
Thompson classification
In 1990, Thompson et al. develop a gross morphological classification scheme for the lumbar intervertebral disc. The grading system separates midsagittal sections into five succinct categories ( Table 7 ) of degeneration.
| Grade | Nucleus | Annulus | Endplate | Vertebral Body |
|---|---|---|---|---|
| 1 | Bulging gel | Discrete fibrous laminae | Hyaline, uniform thickness | Rounded margins |
| 2 | Peripheral white fibrous tissue | Mucinous material between laminae | Irregular thickness | Pointed margins |
| 3 | Consolidated fibrous tissue | Extensive mucinous infiltration; loss of annular-nuclear demarcation | Focal defects in cartilage | Small chondrophytes or osteophytes at margins |
| 4 | Horizontal clefts parallel to endplate | Focal disruptions | Fibrocartilage extending from subchondral bone; irregularity and focal sclerosis in subchondral bone | Osteophytes < 2 mm |
| 5 | Clefts extended through nucleus and annulus | Diffuse sclerosis | Osteophytes > 2 mm |
While not as clinically useful as its counterpart, namely the Pfirrmann system, the Thompson classification exhibits an excellent intraobserver agreement (87%–91%) with an adequate to good interobserver agreement (61%–88%). Despite being in use today, the clinical utility of the Thompson classification has fallen under scrutiny. The inherent limitations of the Thompson system, namely that it requires in vitro disc samples, curtail the system’s widespread usage, limiting it to mostly the academic realm.
Disc herniation
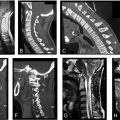
Disc herniation is described as a part of the disc nucleus that has slipped, ruptured, or bulged into the spinal canal. Herniation is often the result of increased loading or pressure on the vertebral column which leads to degeneration over time, though sudden insults may also lead to herniation. As the disc protrudes, patients may experience radicular symptoms, including pain, numbness/tingling, or weakness. To date, multiple studies have examined individuals with herniations through multiple modalities (e.g., autopsies, computerized tomography (CT), magnetic resonance imaging (MRI), etc.). There are currently three main classification systems to describe disc herniations, as outlined in Tables 8–10 and Fig. 4 .
| Type | Description |
|---|---|
| Normal | Normal disc pathology |
| Bulge | Symmetric circumferential extension of the disc beyond interspace |
| Protrusion | Focal extension of the disc beyond interspace |
| Extrusion | The base of disc material is smaller than any other disc material dimension |
| Morphology |
| Protrusion |
| Extrusion |
| Intravertebral |
| Containment |
| Continuity |
| Relation with PLL complex |
| Volume |
| Composition |
| Location |
| (a) | ||
|---|---|---|
| Features | Score | |
| Clinical manifestation (CM) | ||
| Pain | ||
| Slight lower limb(s) pain, tolerable without analgesics for > 6 weeks | 1 | |
| Heavy lower limb(s) pain, tolerable with analgesics for > 6 weeks | 2 | |
| Severe lower limb(s) pain, intolerable with analgesics for > 6 weeks | 3 | |
| Nervous function | ||
| Slight involvement of nerves (numbness in any nerve between L4-S1) | 1 | |
| Involvement of nerves (strength decreasing 1–2 grade in digital extensor L4-S1) | 2 | |
| Muscle strength decreasing ≥ 3 grade, plus dysfunction of the sphincter (foot-drop or uroschesis) | 3 | |
| Straight Let Raising Test (SLRT) | ||
| ≥ +70 degrees | 1 | |
| +30–50 | 2 | |
| ≤ +30 degrees | 3 | |
| Imaging findings | ||
| CT or MRI cross-section | ||
| Central | ||
| 1. Protrusion of spinal sagittal diameter < 30% | 1 | |
| 2. Protrusion of spinal sagittal diameter 30%–50% | 2 | |
| 3. Protrusion of spinal sagittal diameter > 50% | 3 | |
| Paramedian | ||
| 1. lateral recess stenosis by protrusion < 30% | 1 | |
| 2. lateral recess stenosis by protrusion 30%–50% | 2 | |
| 3. lateral recess stenosis by protrusion > 50% | 3 | |
| Foraminal | ||
| 1. Intervertebral foramen stenosis by protrusion < 30% | 1 | |
| 2. Intervertebral foramen stenosis by protrusion 30%–50% | 2 | |
| 3. Intervertebral foramen stenosis by protrusion > 50% | 3 | |
| (b) | ||
|---|---|---|
| 6-Score V-Type Criteria | ||
| Type | Points | |
| I | 2 | |
| II | 3 | |
| III | 4 | |
| A | CM score of 3 and image finding score of 1 | |
| B | CM score of 2 and image finding score of 2 | |
| C | CM score of 1 and image finding score of 3 | |
| IV | 5 | |
| V | 6 | |

Currently, the Jensen and CTF classification system is the most commonly utilized system for describing lumbar disc pathology. Researchers and physicians dispute the clinical relevance of these systems. In an attempt to revitalize the classification of disc herniations, two separate research teams developed their own original systems. While the MSU system offers great interobserver reliability, as high as 98%, it is not without its faults. Recent criticism of the system cites oversimplicity in the system, which led the authors to question the classification’s clinical relevance. The Hao system currently leverages a 6-score V-type system that can be used to score and evaluate future patients. Due to the novelty of the study, little follow-up, aside from the initially reported 98% interexaminer reliability, has been initiated. As a whole, the current heterogeneity in classification systems amplifies the need for further evaluation to determine clinical correlations.
Trauma and fracture classifications
Lumbosacral fractures are complex and exhibit heterogeneous nature as a result of the multiple ways they may be sustained. Over the years, numerous classification systems have been developed and subsequently discarded. In light of novel imaging modalities, increased efforts have been made to develop a singular, original classification system that not only organizes lumbosacral fractures but allows for consistent treatment across providers. To date, despite the best efforts by experts, no such system exists. This had led to the use of multiple classification schemes, which are explored in the following sections.
Brief history of the classification of lumbosacral fractures
The exploration and development of lumbosacral fracture classification systems has been a long, arduous journey. The advent of these classification systems can be traced back to the mid-1900s when Boehler described five categories of spinal fractures. While the system leveraged physiologically relevant information, namely the type of injury (compression, flexion, extension, etc.), it was ultimately served as a starting for other early classification systems, such as Watson-Jones (1938) and Nicoll (1949). While each of these systems utilized different techniques to explain fracture subtleties, spinal solidity, and anatomy respectively, they both placed precedence on the ligamental stability in their schemes.
Fracture classification was reinvigorated in 1963 with the advent of the Holdsworth classification. Drawing on the previously described models, specifically the role of the posterior spinous ligament in the Watson-Jones model and bony anatomy in the Nicoll model, Holdsworth proposed a two-column theory that placed precedence on the biomechanical stress or trauma of each injury subtype, thereby integrating all three of the previously proposed systems. An attempt to reexamine Holdsworth scheme, focused specifically on the anterior column, was carried out by Kelly and Whiteside in 1968. Despite the limited sample ( n = 11), the classification paved the way for subsequent classifications, namely Denis and McAfee.
While each of the aforementioned classification systems served as the chief schema in their respective times, all were eventually replaced in favor of novel mechanistic considerations or a more objective measurement provided by the advancement in imaging modalities. It is important to note that despite the use of newer imaging modalities, some more recent classification systems, namely the McAfee, Ferguson-Allen, and McCormack Classification (Load Sharing Classification), were eventually replaced as pathophysiology implications were elucidated.
Roy-Camille classification
In 1984, Roy-Camille proposed a classification system for sacral fractures, placing attention on U-shaped injury patterns. The system categorizes the fractures into three types based on the location of the cranial or rostral portion. Injury types 1 and 2 describe degeneration or trauma as a result of flexion whereas type III predominately indicates extension injuries ( Table 11 ).