Computed Tomography Enterography: How It Differs From Routine Abdominal Computed Tomography
Computed tomography enterography (CTE) reflects individualization of the abdominal pelvic CT technique for patients with small bowel disorders. CTE provides visualization of the small bowel lumen, wall, and perienteric tissues by distending the small bowel with large volumes of oral contrast and obtaining multiplanar, high-resolution images of the bowel during appropriate phase(s) of enhancement. It differs from routine abdominal pelvic CT in the amount and type of oral contrast given to the patient prior to scanning, timing of image acquisition with respect to the intravenous (IV) contrast bolus, routine reconstruction of thin multiplanar images, and other factors related to patient-specific indications (e.g., Crohn’s disease or obscure gastrointestinal [GI] bleeding). CT acquisition for CT enteroclysis is similar, but the luminal small bowel distention is achieved by infusion of enteric contrast via a nasojejunal tube.
Indications
CTE is performed in patients with known or suspected Crohn’s disease, patients with obscure GI bleeding, diarrhea of unknown cause (often in association with imaging of the pancreas), and in patients with abdominal pain of unknown cause in an outpatient setting. Multiphase CTE is often used as a complementary technique with capsule endoscopy in patients with obscure GI bleeding, and its complementarity with other tests is reviewed later in this chapter. CTE may also be used to evaluate for the presence and cause of low-grade obstruction (when an enteroclysis cannot be performed), and to examine for findings or complications of other small bowel disorders (e.g., small bowel diverticulosis, celiac disease, polyposis syndromes).
Technique
The CTE technique is adapted to the indication for the examination. Table 38-1 describes common variations of CTE technique, linking them to clinical indications. All types of CTE require the ingestion of large amounts of oral contrast and adaptation of the CT x-ray tube potential and tube current to adjust for patient size and diagnostic task to minimize radiation dose appropriately, yet provide an accurate diagnosis.
| Indication | Phase(s) of Enhancement | Dose Level | Comments |
|---|---|---|---|
| R/O small bowel disease | Enteric or portal | Routine abdominopelvic CT | Neutral oral contrast, high resolution * |
| R/O or FU Crohn’s disease | Enteric or portal | Less than for routine abdominopelvic CT | Neutral oral contrast, high resolution * |
| Active GI bleed | Arterial and portal or delayed | Routine abdominopelvic CT | Neutral oral contrast, high resolution * |
| Obscure GI bleed | Arterial, enteric and delayed | More than for routine abdominopelvic CT (because of more than one phase of enhancement) | Neutral oral contrast, high resolution * |
| Polyposis syndrome | Noncontrast | Less than for routine abdominopelvic CT | Enteroclysis preferred, positive oral contrast, high resolution |
| Low-grade obstruction | Enteric or portal | Routine abdominopelvic CT | Enteroclysis preferred, neutral oral contrast, high resolution |
* Axial slice thickness is ≤3 mm, with routine coronal ± sagittal multiplanar reconstructions or interactive 3D viewing of volumetric CT data at a computer workstation.
For CTE, 1300 to 2000 mL of oral contrast is typically administered to the patient over an hour in divided increments (e.g., 450 mL at 60, 45, and 15 minutes prior to the examination). Contrast agents are generally divided into neutral enteric agents that demonstrate CT number attenuation similar to that of water and positive enteric contrast agents containing barium or iodine, which have CT numbers much greater than those of enhancing structures. Neutral agents are preferred for most small bowel indications because they allow greater conspicuity of many small bowel pathologies, such as segmental inflammation or masses, which usually are hyperenhancing compared with the adjacent small bowel wall. Positive agents are useful when the suspected pathology is known to be an intraluminal filling defect (e.g., polyposis syndromes), serosal metastases are suspected, or IV iodinated dye is contraindicated. Although a variety of neutral enteric contrast agents are available, many centers use commercially available products, which often contain sorbitol, which retards absorption of water across the small bowel wall, improves small bowel distention compared with water alone, and provides an expanded time window during which small bowel imaging can be achieved with a high degree of reproducibility. Alternatively, locust bean gum or polyethylene glycol may also be used. If patients have difficulty with oral ingestion of these neutral agents, they can complete ingestion of the recommended volume with water. Patients need to be informed that enteric agents using sorbitol or polyethylene glycol may cause loose bowel movements as the agent is expelled shortly after the test. Because of the requirement for the ingestion of large volumes, CTE using non–water agents is an outpatient procedure, and radiologists and referring clinicians should consider alternatives for hospitalized patients (for whom routine abdominal-pelvic CT or CTE with water may be preferred). Also, approximately 30% of jejunal loops will be collapsed using the peroral CTE technique ; therefore, so in the presence of known or suspected jejunal pathology, CT enteroclysis or magnetic resonance (MR) enterography, in which the jejunum is imaged repeatedly with different pulse sequences, may be preferred.
IV contrast is typically delivered at a rate of 3 to 5 mL/s, with imaging during the enteric or portal phase of enhancement. The enteric phase of enhancement refers to imaging during peak enhancement of the small bowel wall, which is generally regarded as 50 seconds after the initiation of IV contrast material. The enteric phase demonstrates findings of Crohn’s disease, hyperenhancing polyps, and most small bowel vascular lesions. The timing of the enteric phase is similar to that of the pancreatic phase because the superior mesenteric and portal veins are opacified with IV contrast media and the hepatic veins have not yet filled with contrast; thus, synchronous evaluation of the pancreas can often be performed with a single acquisition. In the hepatic or portal phase of enhancement, hepatic veins are filled with contrast; this may be a more appropriate phase of enhancement if correlative imaging of the liver is also needed. For the most common indication of imaging Crohn’s disease, active Crohn’s disease is adequately demonstrated in the enteric and portal phases.
Because one of the goals of CTE is to display small fistulas and intramural or intraluminal masses, high-resolution CT imaging is required, with the axial slice thickness generally reconstructed on the order of 3 mm or less. Multiplanar coronal reformatted images are required, and sagittal reformatted images are also generally obtained to aid in the evaluation of suspected small bowel segments and the mesenteric vasculature. The pitch or CT scanner table speed is adjusted so that the abdomen and pelvis can be imaged within a single breath-hold (i.e., 6 to 15 seconds). For most clinical indications (e.g., to rule out small bowel disease, evaluate Crohn’s disease), only a single phase of contrast enhancement is required.
Irrespective of the indication, the CTE acquisition technique should be adapted to patient size and diagnostic goal. One of the best radiation dose-saving techniques is for the radiologist to limit the number of phases acquired to those that are necessary for the suspected diagnosis. Multiple phases of enhancement are reserved for the investigation of obscure GI bleeding or situations in which multiphasic imaging of solid organs is required. Tube current adaptation can be carried out through the use of technique charts or automatic exposure control. The routine use of automatic exposure control will lower radiation dose by approximately 30% in large CT practices. Additionally, tube energy selection (kV) can be performed using technique charts or vendor-supplied kV selection programs, which take into account patient size, iodine contrast-to-noise ratio (CNR), and CT system limitations. Selection of kV and technique charts generally reduce radiation dose by an additional 20% to 30%. One of the advantages of lower kV is that it not only reduces radiation dose, but increases the iodine signal and iodine-related CNR, which can be used to increase the conspicuity of hyperenhancing bowel segments and masses or lower the radiation dose.
The dose level chosen for different types of enterography examinations depends on the image contrast between the suspected small bowel pathology and normal, enhancing bowel wall, as well as other pathology, which may be suspected in the solid organs (see Table 38-1 ). In Crohn’s disease, the diagnostic task requires differentiating relatively large differences in CT number between normal and hyperenhancing bowel segments, so routine abdominal pelvic CT dose levels can be lowered by 30% to 50% without sacrificing diagnostic performance. Similarly, if using positive enteric contrast to evaluate for filling defects such as polyps in polyposis syndromes, the CT number differences between the suspected pathology and adjacent bright lumen are large, and the radiation dose can be substantially reduced (analogous to CT colonography). In contrast, CTE performed to evaluate synchronously for pancreatic or small bowel masses or hepatic metastases will require routine abdominal pelvic dose level settings so that important low-contrast structures and masses can be visualized adequately.
The lower radiation dose levels frequently used in CTE, along with the thinner slices, often result in noisier images, which can be accurately interpreted without diagnostic compromise in the setting of Crohn’s disease. A variety of CT image noise reduction methods can be used (e.g., iterative reconstruction) to improve the image quality of the lower dose CTE images so that they resemble those obtained with a routine dose. CT noise reduction is especially useful when reducing radiation dose with low-kV techniques to improve disease conspicuity and compensate for the increased image noise associated with low-kV imaging.
Interpretation of Computed Tomography Enterography Images
Routine CT enterography performed for general indications of Crohn’s disease requires only the review of axial and coronal images, although interactive viewing with a volume viewer or in other planes is often helpful. Similar to fluoroscopy, a systematic approach to view the bowel is required to maximize disease detection. Because Crohn’s disease, GI bleeding, and small bowel tumor exclusion are frequent indications for CTE, CT findings in these entities will first be considered, with emphasis on the differential diagnosis. Appropriate diagnosis relies on combining knowledge of the intraluminal morphology seen at fluoroscopy with the mural and intraluminal enhancement seen at cross-sectional imaging.
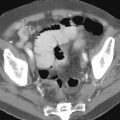
Normal small bowel findings at CTE are similar to those observed fluoroscopically. The jejunal caliber is slightly larger than the ileum, and the jejunal valvulae conniventes are slightly thicker and more closely spaced than in the ileum. Approximately 30% of jejunal folds will be collapsed and should not be confused with small bowel masses. In the enteric phase of enhancement, the jejunum enhances to a significantly greater degree than the ileum, with the attenuation difference normalizing in later phases ( Fig. 38-1 ).

Suspected Crohn’s Disease
CTE images in patients with known or suspected Crohn’s disease should be scrutinized for CT findings of mural inflammation anywhere along the GI tract, penetrating disease (e.g., sinus tracts, fistulas, phlegmon, and abscesses), bowel obstruction, and inflammatory bowel disease (IBD)–related extraenteric findings, including mesenteric venous thromboses, sacroiliitis, and primary sclerosing cholangitis. Chapters 40 and 54 describe the imaging features of Crohn’s disease in greater detail.
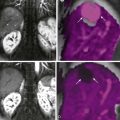
CT findings of mural inflammation include segmental mural hyperenhancement, mural thickening, mural stratification, abnormal mural or perienteric fat, and the comb sign. Mural hyperenhancement refers to segmental attenuation (or enhancement) greater than that of adjacent small bowel loops. Although not specific, it is generally considered to be the most sensitive findings for Crohn’s disease–related inflammation, and it is highly correlated with endoscopic and histologic findings of inflammation. Because the jejunum enhances more avidly than the ileum, comparison to only nearby loops is important in this evaluation. Jejunal hyperenhancement associated with jejunal Crohn’s disease is often more difficult for radiologists to appreciate because of the prominence of the valvulae conniventes, or jejunal folds ( Fig. 38-2 ). Other imaging features such as penetrating ulcers, mural thickening, and disruption of the fold pattern are often seen in jejunal Crohn’s disease, which frequently involves a longer length and multiple segments compared with ileal Crohn’s disease.

A wide variety of diseases can also cause mural hyperenhancement; however, patchy and asymmetric hyperenhancement or continuous hyperenhancement along the mesenteric border are pathognomonic for Crohn’s disease (Video 38-1; Table 38-2 ). Contraction of small bowel loops can cause increased attenuation of a small bowel loop, but is usually readily distinguished by smooth tapering of the adjacent distended segments on either end. Infection, backwash ileitis, angioedema (often from angiotensin-converting enzyme [ACE] inhibitors), vasculitis, and bowel ischemia can all cause segmental hyperenhancement of bowel loops. However, these entities are not usually associated with asymmetric enhancement or penetrating complications. When faced with equivocal hyperenhancement, comparing two nearby distended bowel loops and taking into account secondary signs of Crohn’s disease (e.g., mural stratification, perienteric edema, and the comb sign) are often helpful. Radiation enteritis can also produce narrowed and hyperenhancing bowel loops, but it is easily distinguished from Crohn’s disease because of the history of radiation and symmetric hyperenhancement.
| Disease Entity | Comments | Differential Imaging Features |
|---|---|---|
| Crohn’s disease | Jejunal involvement frequently overlooked | Asymmetric, patchy or mesenteric border hyperenhancement; multifocality; perianal disease or other penetrating complications |
| Backwash ileitis | Symmetric involvement; continuous with pancolonic involvement, patulous ileocecal valve | |
| Angioedema | Often associated with abdominal pain and use of ACE inhibitors | Symmetric involvement, dilated loop, ascites |
| Contraction, peristalsis | Delayed images often help | Smooth tapering on proximal and distal ends; compare with other phases of enhancement |
| Radiation enteritis | History of pelvic radiation | Long-segment hyperenhancement without asymmetry |
| NSAID enteropathy | History of NSAID ingestion | Focal 1- to 2-cm lesion, often multiple, with intervening normal-appearing bowel |
| Ischemia | Patient often older or had recent cardiac event | Symmetric involvement; concurrent closed loop obstruction, venous or arterial occlusion or thrombosis |
| Infection or neutropenic enteritis | Clinical history often suggestive | Symmetric involvement |
| Vasculitis | Symmetric involvement; vessel and solid organ involvement |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree