Key Points
- ▪
Cardiac CT is best suited to imaging the larger-caliber coronary segments, such as proximal coronary arteries, ostial lesions, and the left main stem coronary artery.
- ▪
The ambiguities in a subset of coronary ostial lesions as assessed by conventional angiography may potentially be resolvable by CCT.
- ▪
Aortic diseases associated with ostial coronary involvement are well assessed by CCT.
- ▪
Ostial compression by extrinsic structures also is potentially well evaluated by CCT.
Lesions of the coronary artery ostia and of the left main stem coronary artery are of higher clinical and angiographic risk.
Left main coronary ostial lesions are notable for being found more commonly among middle-aged women with fewer conventional coronary artery risk factors and for lower long-term patency of internal thoracic grafts.
Although conventional angiography is the gold standard for assessment of ostial and left main stem lesions, numerous aspects of conventional angiography for the assessment of these lesions may be challenging when trying to obtain definitive imaging characterization, especially to assess membrane-like lesions, complex courses, eccentric luminal geometry (slit-like orifices), and extremely proximal or ostial lesions. Intravascular ultrasound (IVUS), coronary CT angiography (CTA), and coronary MR angiography (MRA) have all been used in attempts to resolve ambiguous cases.
The left main stem coronary artery, when investigated by cardiac CT studies of 70 consecutive cases, was seen to be elliptical at its ostium in 94%, at its mid-portion in 73%, and at its distal portion in 77% of cases. The most common morphology is biconcave, followed by tapering, combined morphology, and, least commonly, funnel-shaped. In 72% of cases, the ostium of the left main stem arises from the middle one third of the aortic sinus, in 22% it arises from the posterior third of the sinus, and in 4% it arises from the anterior third. In men, significant correlation is found between left main stem cross-sectional area and body weight, height, and body surface, whereas there is no such correlation in women. Left main stem ostial angulation is a normal finding/variant associated with posterior position of the ostium.
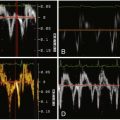
CCT is best suited to imaging the larger-caliber coronary segments—hence its suitability to assess the left main stem in particular, as has been advocated by some. In general, for the assessment of minimal lesional diameter ( r = 0.77, P < .01), minimal lumen area ( r = 0.93, P < .01), lumen area stenosis ( r = 0.83, P < .01) and plaque burden ( r = 0.94, P < .01), CCT findings correlate well with those of IVUS.
Among asymptomatic patients, an IVUS minimal luminal diameter and minimal luminal area of 2.8 mm and 5.9 mm 2 , respectively, strongly predict the physiologic significance of a left main stem coronary lesion; a fractional flow reserve of 0.75 is strongly associated with survival and event-free survival.
The prevalence of significant stenotic disease of the left main stem is influenced principally by the nature of the referral population, but has been suggested to be 2% by one series of 1000 consecutive cases. The dichotomization of left main stem disease into significant versus nonsignificant based on 50% stenosis has been queried by some.
The use of cardiac CTA to image the left main stem coronary artery post-stenting has shown some validation. However, numerous considerations still need resolution :
- □
Complex (distal) left main stem coronary artery stenting of LMCA bifurcation lesions that achieve overlapping or adjacent (“crush”) stents would be expected to yield greater artifacts and more potential for error, and have not been well described in the literature.
- □
Prominent calcium within left main stem lesions presents another difficulty for the assessment of LMCA lesions, as with all coronary lesions.
- □
A 1-mm error in ascertaining restenosis of the LMCA would incur, on average, quite a significant (∼20%) error of restenosis severity.
The relationship between LMCA IVUS determinations of stenosis severity with fractional flow reserve determinations of severity is presented in Figure 13-1 .

Etiology of Coronary Artery Ostial Lesions
- □
Congenital
- •
Isolated atresia of the coronary ostia
- •
Associated with supravalvular aortic stenosis
- •
- □
Aorto-arteritis
- □
Takayasu arteritis
- □
Relapsing polychondritis
- □
Syphilis
- □
Post-radiation
- □
Post–aortic valve replacement (AVR)
- □
Post-AVR/intracoronary cardioplegia perfusion
- □
Post–freestyle stentless bioprosthesis
- □
Trauma
- □
Stenting/restenosis
- □
Spasm
Etiology of Left Main Stem Lesions
- □
Intrinsic lesions
- •
Congenital
- •
Atresia
- •
Anomalous right-sided origin
- •
Anomalous origin from the pulmonary artery (ALCAPA)
- •
Anomalous fistula of the LMCA to the pulmonary artery
- •
Kinking
- •
- •
Atherosclerotic
- •
Thrombotic
- •
Stenting
- •
Restenotic
- •
Restenosis
- •
In-stent restenosis
- •
- •
Post-surgical
- •
Post-AVR
- •
Post-AVR/coronary perfusion
- •
Post–aortic root reconstruction
- •
Surgical gelatin-resorcin-formalin glue
- •
End-to-end anastomosis of the LMCA onto a Freestyle (Medtronic) or Toronto SPV (St. Jude Medical) aortic valve/root
- •
Post–aortic valve and root repair
- •
Post–Bentall repair aneurysms, stenosis, and dehiscence
- •
- •
Dissection
- •
Aortic dissection
- •
Spontaneous coronary dissection
- •
- •
Post-syphilitic
- •
Post-radiation
- •
Aortitis: Takayasu arteritis
- •
Spasm
- •
- □
Extrinsic lesions
- •
Pulmonary artery compression of the LMCA
- •
Pulmonary hypertension
- •
Eisenmenger syndrome
- •
Pulmonary artery dilation
- •
Main pulmonary artery aneurysm
- •
- •
Right pulmonary artery stenting
- •
Post-pericardiotomy syndrome
- •
Coronary CTA
Coronary CTA has documented:
- □
Left main stem patency, stenosis, restenosis, and ostial stent-related problems of the left main stem coronary artery ( Figs. 13-2 and 13-3 )

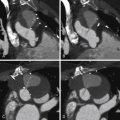
Figure 13-2
A 63-year-old man with symptoms of typical stable angina, CCS Class III, and a negative myocardial perfusion scan . A and B, CT coronary angiography demonstrating a mixed calcified/noncalcified plaque of significant stenosis at the ostium of the left main stem coronary artery. On the basis of these findings the patient was referred for coronary angiography. C and D, First angiogram, different views. The study was reported as “normal.” E and F, Second angiogram 2 weeks later, with intravascular ultrasound (IVUS). The IVUS image at the ostial left main stem reveals a significant (70–80%) stenosis from a large volume of plaque. Note the lucent internal elastic lamina layer that references the actual vessel wall. More careful angiography demonstrates the same finding, which was poorly represented on the initial angiogram, as the catheter tip was distal to the lesion and the back-blow never clearly outlined the stenosis.

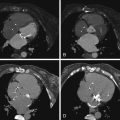
Figure 13-3
A 52-year-old man with a known history of coronary artery disease. The patient had previously undergone stenting of the proximal right coronary artery (RCA) and proximal circumflex arteries. At the time of the circumflex artery stenting, an iatrogenic dissection of the left main coronary artery occurred. The patient was treated conservatively for this. A follow-up cardiac CT study 1 year later ( A and C ) demonstrated a small amount of mixed plaque at the origin of the left main coronary artery, causing a mild 30% stenosis. There was no evidence of a residual dissection. A second follow-up study at 36 months ( B and D ) demonstrates interval worsening of left main coronary artery disease, with more extensive soft and calcified plaque, and the development of a significant 50% to 60% left main stenosis.
- □
In-stent restenosis of the left main stem coronary artery ( Figs. 13-4 through 13-6 )

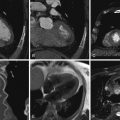
Figure 13-4
A 63-year-old man presented with an acute coronary syndrome. An initial angiogram ( A ) demonstrated thrombosis of the left main coronary artery, which led to immediate percutaneous intervention of the left main coronary artery ( B ). The patient presented with recurrent angina 4 months later. C, A repeat conventional angiogram showed mild in-stent restenosis involving the inferior potion of the left main stent. D, A subsequent cardiac CT study in follow-up confirms the mild in-stent restenosis of the left main coronary artery as subtle low attenuation along the inferior aspect of the stent, but no further progression.

Figure 13-5
Multiplanar reformations of a patient with a stented left main stem coronary artery. The proximal aspect of the stent sits 3 to 4 mm back in the aortic root. There is mild to moderate mixed disease of the proximal left anterior descending artery.

Figure 13-6
A 51-year-old man 1 year following a Bentall procedure with coronary button implantation using reinforcement with BioGlue (CryoLife), was experiencing atypical chest pains. A, CT axial oblique image showing ostial right coronary artery (RCA) stenosis after a Bentall procedure ( arrow ). The left main artery ( arrowhead ) also is seen. B, Right coronary angiogram in the left anterior oblique view shows ostial RCA stenosis ( arrow ). C, Right coronary angiogram after stenting of the RCA stenosis. D, RCA ostial stenosis was shown proximal to the previously deployed stent on CT axial oblique and cross-sectional images ( arrows ) and curved multiplanar reformatted images ( arrowhead ). E, Right coronary angiogram in left anterior oblique view shows ostial stenosis proximal to stent and mild in-stent restenosis. Ao, aorta; LAA, left atrial appendage; RV, right ventricle; ∗, coronary button markers.
(Reprinted with permission from Shenoda M, Barack BM, Toggart E, Chang DS. Use of coronary computed tomography angiography to detect coronary ostial stenosis after Bentall procedure. J Cardiovasc Comput Tomogr . 2009;3(5):340-343).
- □
Catheter-induced spasm of the left main stem coronary artery ( Figs. 13-7 through 13-9 )

Figure 13-7
A 65-year-old man with a prior ventricular fibrillation cardiac arrest and an implantable cardioverter defibrillator. Coronary angiography reveals occlusion of the proximal left circumflex; CT angiography demonstrates patency and no plaques within the left circumflex, establishing a strong case that the occlusion at the time of angiography was due to spasm.

Figure 13-8
A–C, Conventional coronary angiography showing left anterior descending (LAD) artery, left circumflex (LCX) artery, and right coronary artery (RCA) with isolated high-grade ostial stenosis of the left main coronary artery ( arrow ). D–F, Curved multiplanar reconstructions of LAD, LCX, and RCA in CT angiography showing a completely normal left main coronary artery ( arrow ), thereby proving catheter-induced spasm as the underlying reason for the false-positive lumen narrowing in invasive catheterization.
(Reprinted with permission from Pflederer T, Marwan M, Ropers D, et al. CT angiography unmasking catheter-induced spasm as a reason for left main coronary artery stenosis. J Cardiovasc Comput Tomogr . 2008;2(6):406-407.)

Figure 13-9
An angiographic case of left coronary artery spasm. A, On the first injection, the caliper of the left main stem is seen. Within a few seconds, the left main stem artery has occluded and the entire left coronary tree is also in severe spasm. Ventricular fibrillation ensued. C, After defibrillations, and intracoronary nitroglycerin, the examination was resumed. D, An eccentric lesion at the ostium to the left coronary artery is evident on the angiogram and on the intravascular ultrasound image.
- □
Left main stem ostial stenosis due to Takayasu arteritis ( Figs. 13-10 through 13-12 )

Figure 13-10
Occlusion of the left main coronary artery in Takaysu arteritis . Coronary CT and invasive angiography of a patient with Takayasu arteritis (Numano type IIb) with an ostial occlusion of the left main coronary artery and right coronary artery dilation as a compensatory mechanism. A, Coronary CT angiographic axial image at the origin of the left main coronary trunk shows lack of opacification of the vessel ( arrow ), associated with negative remodeling. The vessel reconstitutes distally. B, Conversely, the right coronary artery ( arrowheads ) is diffusely dilated as a compensatory mechanism, as demonstrated in this volume-rendered image, but its origin also shows mild tapering, as seen in the multiplanar reformat ( C; arrow ). Note the absence of contrast material within the lumen of the left main coronary trunk ( B; arrow ). D and E, Conventional coronary angiography confirms the right coronary ectasia and its tapering at the origin ( D; arrow ) and the occlusion of the left main trunk ( E; black arrow ). Multiple collaterals ( small white arrows ) to the left coronary circulation coming from the right coronary artery and reconstituting the mid-distal left anterior descending and left circumflex coronary arteries ( large white arrows ) also are seen. Most significant coronary lesions in patients with Takayasu arteritis are ostial and noncalcified, as they were in this case.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree