CHAPTER 18 CT colonography
Introduction
Colon cancer is the third most common malignancy in men and the second most common malignancy in women with 35 599 new cases reported in the UK in 2005 (Cancer Research UK, 2008). Most colon cancers arise from pre-existing adenomatous polyps where the incidence of malignancy increases with size. There are well known risk factors to developing colon cancer, such as hereditary polyposis syndromes and chronic inflammatory bowel disease, but most cancers occur in patients with no predisposing risk factors other than advancing age (Tolan et al., 2007).
Computed tomography colonography (CTC) was first described in 1994 as dual position, helical computed tomography (CT) of a cleansed, gas distended colon (Vining et al., 1994). Since that time, the examination has steadily evolved to the point where it is not only advocated for the investigation of patients with symptoms of colon cancer but also as a screening test for asymptomatic patients (Levin et al., 2008).
Meta-analysis of published data demonstrates that CTC has high sensitivity and specificity rates (using conventional colonoscopy as the reference standard) for large and medium size polyps and a high reported diagnostic accuracy for symptomatic cancer (Pickhardt et al., 2003; National Institute for Health and Clinical Excellence, 2005). However, diagnostic accuracy falls with polyp size and, as with conventional colonoscopy, there may be low sensitivity in detecting flat colonic lesions (Hoon et al., 2003). Studies have also shown that CTC is significantly more sensitive than barium enema at polyp detection and has the additional advantage of being able to demonstrate significant extracolonic pathology in approximately 9% of patients (Yee et al., 2005; Spreng et al., 2005; Taylor et al., 2006; Tolan et al., 2007).
Research shows that the diagnostic accuracy of CTC is highly dependent on the quality of the examination and that meticulous attention must be given to both examination and interpretation techniques in order to achieve acceptable diagnostic performance (Ho Park et al., 2007; Rockey et al., 2007).
CTC technique
Patient information
Patient compliance is vital to achieve an optimal CTC examination, therefore, it is most important that high quality, accurate written information should be available so that patients are aware of the bowel preparation, the dietary restrictions and their effect on the diagnostic outcome. A description of what the procedure involves must be included so patients are forewarned about what they will undergo, especially the need to distend the colon with gas and the administration of spasmolytic drugs and contrast media. The information leaflet should clearly explain the risks, benefits and alternatives to the procedure, including the risk of perforation, contrast reaction, side effects of the spasmolytic drugs and the radiation dose. The information should also include advice for patients with diabetes on where and how to obtain specific information on glycemic control and specialist information for those taking Metformin (Tolan et al., 2007).
Bowel preparation and fecal tagging
Reliable detection of small polyps at CTC is heavily dependent on optimum bowel preparation. The presence of fecal residue and retained fluid can cover colonic mucosa, hide pathology and reduce the sensitivity and specificity of the examination (Mang et al., 2007).
There is currently no consensus as to the optimum bowel preparation regime for CTC, although it is generally recognized that a clean, dry colon is required. Picolax (sodium picosulphate and magnesium citrate) is widely used in the UK for barium enema and colonoscopy bowel preparation (Box 18.1) and there is evidence to support its efficacy for CTC (Taylor et al., 2003a). Patient safety and tolerance should also be considered in the choice and administration of bowel preparation as vulnerable groups, such as the elderly and those with renal impairment, may be at risk of dehydration and electrolyte disturbance. Patients with diabetes should be advised to contact departments so they can be scheduled first on the list and to contact a diabetic nurse specialist to obtain advice regarding glycemic control for the period of dietary restriction (Tolan et al., 2007).
BOX 18.1 Standard bowel preparation
The day before the procedure
| 0800 | Take one sachet of Picolax |
| Then drink as much clear fluid as you can, including clear soups, Oxo, Bovril, jelly and sweet, fizzy drinks | |
| 1600 | Take the second sachet of Picolax |
| Continue to drink as much clear fluid as you can, including clear soups, Oxo, Bovril, jelly and sweet, fizzy drinks until your examination |
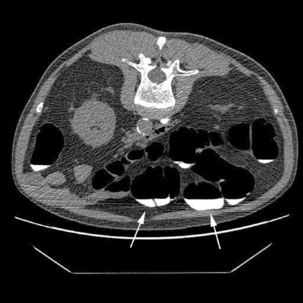
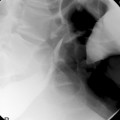
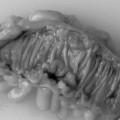
It is recognized that full bowel preparation does not always result in a completely clean colon. Techniques have been developed that allow residue and fluid to be labeled or tagged using oral contrast agents in order to avoid them being confused with pathology. There are a number of tagging protocols in use which include barium compounds, iodinated contrast media or a combination of the two (Box 18.2). It is suggested that barium is superior at tagging solid residue and iodinated contrast media is better at tagging fluid, although there is undoubtedly some overlap (Figure 18.1). The diagnostic accuracy of CTC can be further increased by using specialized computer software to perform ‘electronic cleansing’. This allows the opacified colonic fluid and barium tagged stool to be digitally removed at the post processing stage so that it does not obscure the visualization of polyps or significant pathology (Rockey et al., 2007; Mang et al., 2007).
BOX 18.2 Full purgation with stool/fluid tagging: University of Wisconsin (standard regimen)
| 24 h before | Clear liquids only |
| 18 h before | Fleet phosphosoda (45 ml) undiluted followed by 1–2 l of clear fluids |
| 15 h before | Barium 2.1% (250 ml) (plus 296 ml of magnesium citrate if bowel cleansing not commenced). Further 1–2 l of clear fluid |
| 12 h before | 60 ml gastrograffin (Bracco Diagnostics) with clear fluids |
| 8 h before | Nil by mouth until examination |
The development of fecal tagging protocols and ‘electronic cleansing’ has resulted in some centers reducing the laxative regime given to patients. This undoubtedly increases patient acceptability as the bowel preparation is often considered the most intolerable part of the examination. It also allows CTC to be performed with reduced or no laxation in those patients where it may be harmful, e.g. the elderly and those with significant comorbidity (O’Hare and Fenlon, 2006; Laudi et al., 2008).
Colonic insufflation
Good colonic distension is fundamental to obtaining a high quality examination and optimal mucosal visualization. Imaging under-distended or collapsed segments of bowel can render the examination non-diagnostic, necessitating a repeat examination or referral for colonoscopy. It may also result in pathologies being missed or lead to a false positive diagnosis. There are several strategies currently used to achieve good colonic distension which include the use of different catheters and insufflation devices, administering spasmolytics and obtaining images in the prone and supine position (Burling et al., 2006a; Mang et al., 2007).
Insufflation techniques vary between centers and a number of methods are currently used to distend the colon. These include manual insufflation of room air or carbon dioxide or the use of a commercially available automated carbon dioxide colon inflator. Inflating carbon dioxide is preferable to room air as it is more readily absorbed, resulting in less post-procedural discomfort and bloating (Burling et al., 2006a).
A small caliber (20F), soft flexible catheter is adequate to obtain optimal colonic distension and is well tolerated by the patient. Large, rigid rectal catheters of the type used for barium enema examinations have not been found to improve colonic distension and, in some cases, have led to rectal perforation and their use should be actively discouraged (Sosna et al., 2006).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree