Etiology
Cystic lesions of the pancreas encompass a wide spectrum of different pathologic entities, ranging from developmental, to inflammatory, to neoplastic cysts. Neoplastic cystic lesions, which are the most important, owing to their profound impact on patient prognosis and the frequent necessity of surgical treatment, are described in detail in this chapter.
Although every pancreatic tumor may undergo central necrosis and manifests predominantly cystic, the term cystic neoplasm properly refers to a cyst lined by a neoplastic epithelium, which identifies the tumor as a serous cystic neoplasm (SCN), mucinous cystic neoplasm (MCN), and intraductal papillary mucinous neoplasms (IPMNs). These lesions account for more than 90% of the whole spectrum of cystic neoplasms. The remaining 10% are represented by neoplasms undergoing cystic degeneration, such as solid and pseudopapillary epithelial neoplasms (SPENs), cystic pancreatic endocrine neoplasms (CPENs), acinar cell cystoadenocarcinomas, cystic metastases, and few other even rarer tumors.
Prevalence and Epidemiology
Owing to the increased awareness, improved diagnostic imaging technology, and intensive use of diagnostic imaging, neoplastic cystic lesions of the pancreas have been increasingly diagnosed recently, often at smaller size than in the past and in asymptomatic patients. Therefore, their actual prevalence and size at presentation are not accurately reflected in the literature.
Prevalence and Epidemiology
Owing to the increased awareness, improved diagnostic imaging technology, and intensive use of diagnostic imaging, neoplastic cystic lesions of the pancreas have been increasingly diagnosed recently, often at smaller size than in the past and in asymptomatic patients. Therefore, their actual prevalence and size at presentation are not accurately reflected in the literature.
Clinical Presentation
Most neoplastic pancreatic cysts are infrequently associated with any symptomatology, and several are incidentally discovered during imaging for an unrelated medical problem. However, few patients present with symptoms related to mass effect, such as abdominal pain, early satiety, vomiting, and jaundice or with symptoms secondary to obstruction or communication with the pancreatic duct, such as recurrent pancreatitis. Moreover, advanced cystic malignancies may manifest as pain, weight loss, and jaundice and be clinically indistinguishable from pancreatic adenocarcinoma.
Imaging
The most commonly encountered neoplastic cystic lesions of the pancreas are represented by SCNs, MCNs, intraductal papillary neoplasms, SPENs, and CPENs.
Computed Tomography
Because of the widespread availability of multidetector computed tomography (MDCT), its capability to image the whole abdomen and pelvis in a single breath-hold, the superb spatial and temporal resolutions, the nearly isotropic voxels obtainable with the current technology, and the robustness to breathing artifact, it is considered the mainstay of diagnostic imaging to assess patients with suspected pancreatic lesions. Moreover, aesthetically pleasing and clinically useful multiplanar reformatted images, including three-dimensional (3D) and angiographic reconstructions of the MDCT data and MDCT-pancreatographic images, facilitate accurate preoperative staging.
Magnetic Resonance Imaging
Although many of the new magnetic resonance imaging (MRI) sequences allow faster imaging acquisition, some factors continue to limit its use as a first-line diagnostic tool for imaging of the pancreas. These factors are mainly related to the need for patient cooperation to reduce motion and breathing artifacts, which can severely degrade and compromise the quality of the examination, the reduced availability of the technique, and inherent costs. MRI may be the preferable imaging modality in patients who are allergic to iodinated contrast agents or have renal insufficiency.
Because of an inherent high soft tissue contrast and resolution achieved with MRI, detection of subtle pancreatic lesions and evaluation of their internal details can be enhanced. Similar to CT, 3D contrast-enhanced dynamic MR angiography also can be performed to map the regional vascular anatomy to enable assessment of vascular involvement from the tumor. In addition, MR cholangiopancreatography (MRCP) allows excellent visualization of the entire extrahepatic biliary tract and the pancreatic duct. When MRCP is used in combination with a dynamic MR examination of the pancreas, comprehensive preoperative imaging can be accomplished to facilitate detection and preoperative staging of the pancreatic malignancy.
Ultrasonography
Transabdominal ultrasonography is highly operator dependent, nonreproducible, and limited by abdominal gas and patient body habitus. Endoscopic ultrasonography (EUS), on the other hand, provides high-resolution images of the pancreas and detailed assessment of cyst morphology. Moreover, EUS allows both aspiration of cystic fluid and sampling of cyst wall or mural nodules. Cystic fluid analysis can provide relevant insights into the nature of the cyst. Extracellular mucin or high viscosity usually favor mucinous neoplasms, whereas high amylase concentration, indicating a communication with the pancreatic duct, can be observed both in pseudocysts and in IPMNs, with very high levels usually found in the case of pseudocysts. Moreover, tumor markers in the cystic fluid can render the diagnosis of malignancy feasible in selected cases.
Nuclear Medicine
Nuclear medicine plays a role only in the case of functional endocrine tumors, whereby the use of a specific radiopharmaceutical can allow the diagnosis to be undertaken.
Positron Emission Tomography with Computed Tomography
The role of positron emission tomography with computed tomography (PET/CT) in the evaluation of cystic lesions of the pancreas is still under investigation. Usually, PET/CT is used as an additional imaging functional test to help differentiate benign from malignant neoplasms. In some studies it demonstrated a diagnostic accuracy as high as 83%, but larger studies are needed.
Imaging Algorithm
The most used diagnostic imaging technique for cystic neoplasms of the pancreas are MDCT and MRI/MRCP, which provide high spatial and contrast resolution images and allow dynamic acquisition and postprocessing image reconstruction. Cyst morphology, relationships with the pancreatic duct, and ancillary findings can be evaluated, providing clues for diagnosis, management planning, and preoperative strategy.
MDCT and MRI have been demonstrated to be equally accurate in establishing the diagnosis of malignancy and characterizing pancreatic cystic lesions.
Through MRCP and MDCT images and reconstructions, the entire course of the main pancreatic duct (MPD) can be displayed and its relationships with the cystic lesion can be evaluated. If the patient is considered a surgical candidate, multiplanar image reconstructions displaying the extent of cystic lesions and their anatomic relationships to surrounding structures can be useful.
For selected cases, when MRCP and MDCT pancreatograms have been unable to provide the required information regarding cystic lesion relationships with the MPD, invasive endoscopic retrograde cholangiopancreatography (ERCP) and/or EUS can be performed. The role of PET/CT is still under investigation.
An ideal algorithm is shown in Figure 47-1 ; see also Table 47-1 .
| Modality | Accuracy | Limitations | Pitfalls |
|---|---|---|---|
| Radiography | Poor |
| Unable to directly visualize soft tissue masses in the pancreas. |
| CT |
| Radiation exposure |
|
| MRI |
|
|
|
| Ultrasonography | Not assessed |
|
|
| Nuclear medicine | No utility unless functional PEN | Poor spatial resolution | |
| PET/CT |
|
| Difficult differentiation of benign from borderline neoplasms. |
The different types of cystic lesions of the pancreas are described in the corresponding sections; the most important features are shown in Table 47-2 .
- •
Cysts may be difficult to differentiate on morphology alone.
- •
Most important answers to be asked in the approach to cystic lesions in the pancreas are differentiation of:
- •
True cyst versus pseudocyst
- •
Mucinous versus nonmucinous
- •
Benign versus malignant lesions
- •
| Factor | SCN | MCN | IPMN | SPEN | PEN | Pseudocysts | Cystic Adenocarcinoma |
|---|---|---|---|---|---|---|---|
| Sex | F > M | F | M > F | F | F = M | M > F | M > F |
| Age * | 6th-7th decades | 4th-5th decades | 6th-7th decades | 2nd-3rd decades | 5th-6th decades | 4th-6th decades | 5th-7th decades |
| Location | Head/body/tail | Tail/body | Head/uncinate | Body/tail | No predilection | Head/tail/body | Head > body/tail |
| Shape and borders | Lobulated | Oval | Grape-like (branch) Focal or diffuse main pancreatic duct dilatation (main type) Both (combined) | Oval | Oval | Oval | Oval/irregular |
| Cystic appearance | Microcystic dense stroma | Macrocystic | Macrocystic or cystic with solid component | Cystic with solid component | Cystic with solid component | Unilocular | Cystic with solid component Septa uncommon |
| Size * | 5-11 cm | 6-10 cm | 1-4 cm (branch type) >5 mm (main type) | 5-9 cm | 2-10 cm | 4-8 cm | 4-9 cm |
| Main pancreatic duct | Normal or rarely compressed | Normal or rarely compressed | Dilated | Normal | Normal | Normal (acute pancreatitis) Dilated (chronic pancreatitis) | Obstructed Upstream dilatation |
| Calcification | Central stellate in 30% | Peripheral/septal | Intraductal (or in the case of mucin plug calcification) | Peripheral, nonlaminated | Sometimes | Parenchymal (chronic pancreatitis) | No |
| Signal intensity | T1 low T2 high | T1 high/low T2 high | T1 low/high T2 high | T1 high T2 high | T1 high/low T2 high | T1 low T2 high | T1 low T2 high |
| Wall | Occasionally thick | Uniformly thick, variable enhancement | Thick, variable enhancement | Thick, strongly enhancing | Thin, occasionally thick | Thick, variable enhancement | |
| Solid components | No Small (<3 cm) may appear solid | If malignant | If malignant | Yes | Yes | No | Yes |
| Clinical history | Noncontributory | Noncontributory | Noncontributory | Noncontributory | ± Endocrine syndromes | Pancreatitis | Weight loss Back and abdominal pain Jaundice (head) |
* Due to increased diagnostic imaging use and improved technology, lesions are currently discovered at a younger age and at a smaller size than reported in the literature.
Specific Lesions
Serous Cystic Neoplasms
Etiology
Biallelic inactivation of the von Hippel-Lindau (VHL) gene has been reported both for the sporadic and the VHL-associated form of SCNs.
Prevalence and Epidemiology
SCNs, which account for 30% to 39% of all pancreatic cystic neoplasms, are slowly growing, benign lesions with a very low malignant potential. They occur predominantly in women (75%), and patient mean age at presentation is 62 years.
Clinical Presentation
They are usually incidentally discovered, unless large enough to be responsible for compression of the surrounding organs.
Pathophysiology
SCNs are usually discovered in the head (42%) or body/tail (48%), less often in the proximal body (7%), or diffusely through the pancreas (3%).
Pathology
At gross pathologic examination, SCNs manifest as large (average diameter, 2 to 11 cm), well-circumscribed, lobulated cystic masses that lack a capsule or a definite wall. SCNs do not communicate with the pancreatic duct and are devoid of peripheral wall calcifications. Cystic fluid is watery, without mucin.
SCNs are usually microcystic and, less commonly (10%), macrocystic or oligocystic. The classic microcystic SCNs have a “sponge-like” or “honeycomb”-like morphology, characterized by innumerable small cysts of a few millimeters in size. Larger cysts, if present, are less than 2 cm in diameter and peripheral. Microcystic SCNs tend to exhibit a central stellate fibrous scar, a feature considered specific for SCNs; they can manifest as stellate calcifications in about 30% of cases. Macrocystic SCNs are composed of a countable number of larger cysts, between 2 and 7 cm, or even by a single large cyst and usually affect a younger population.
On histopathologic examination, SCNs are lined by a monomorphic epithelium, made up of cuboidal or flat cells, rich in glycogen, that stain with periodic acid–Schiff.
Imaging
SCNs are usually asymptomatic and incidentally discovered. Less often, in the case of large lesions, they may come to clinical attention because of mass-effect symptoms.
Computed Tomography.
On both MDCT and MRI, the appearance of SCNs is similar to that of gross pathologic findings. Fine external lobulations and a central fibrous scar ( Figure 47-2 ), with a stellate pattern of calcification ( Figure 47-3 ), are suggestive. Enhancement of septa, cyst wall, and the central fibrous scar is best appreciated in the portal and delayed phases of contrast enhancement, respectively. The MPD is normal unless compressed by a large SCN.
On MDCT, 20% of SCNs, as a result of a honeycombed microcystic composition, appear as well-defined, “spongy,” soft tissue or mixed density lesions, sharply demarcated from the adjacent structures and difficult to differentiate from a solid pancreatic mass on MDCT.
Magnetic Resonance Imaging.
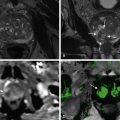
The features of SCNs on MRI are very similar to those on MDCT. The main differences are in better detection of calcification on MDCT and improved visualization of cyst internal architecture and contrast enhancement with MRI. In the case of SCNs appearing solid on MDCT, MRI may provide useful insights into the microcystic nature of the lesions, revealing numerous discrete hyperintense cysts with bright signal on T2-weighted sequences and allowing the correct diagnosis to be undertaken ( Figure 47-4 ).
Ultrasonography.
On transabdominal ultrasonography, owing to the innumerable acoustic interfaces of microcystic SCNs, they can manifest as hyperechoic, lobulated, sharply demarcated masses, lacking posterior acoustic enhancement. In the case of macrocystic and oligocystic SCNs, internal septa are visualized.
EUS, which can resolve the fine details of the internal structure of SCNs, is particularly useful in uncertain cases.
Positron Emission Tomography With Computed Tomography.
Although there is no currently established role for PET/CT in the characterization of SCNs, based on our experience these lesions do not take up fluorodeoxyglucose (FDG).
Imaging Algorithm.
An imaging algorithm is provided in Figure 47-1 ; see also Table 47-3 .
- •
External lobulations
- •
More than six loculations, each less than 2 cm
- •
Central scar, with/without stellate calcifications
- •
Absence of communication with the MPD
| Modality | Accuracy | Limitations | Pitfalls |
|---|---|---|---|
| Radiography | Poor | Insensitive Nonspecific | Unable to directly visualize soft tissue masses in the pancreas. |
| CT | 100% SCN from MCN 90% SCN from IPMN | Radiation exposure | 20% of SCNs, usually when <3 cm, may appear solid on CT. Characterization of small cysts may be difficult. Thin septa not easily appreciated. |
| MRI | Although data not available to specify accuracy, no relevant differences are expected between MRI and MDCT. | Patient cooperation High cost | Calcifications not well visualized. |
| Ultrasonography | Data not available to specify accuracy. | Poor performance in the case of obesity or overlying bowel gas Operator dependent Comprehensive imaging difficult | SCN may present as solid lesions in the case of “honeycomb pattern.” |
| Nuclear medicine | Data not available to specify accuracy; no current role in diagnosis. | Poor spatial resolution | |
| PET/CT | Data not available to specify accuracy. | Radiation exposure High cost |
Differential Diagnosis
Clinical data usually are not helpful in discriminating SCNs from other cystic neoplasms of the pancreas. Lobulations, microcysts, and a central scar are present in the classic form of SCN and suggest the diagnosis.
Macrocystic SCNs are usually difficult to differentiate from mucinous cystic tumors, with which they share many morphologic features. However, external lobulations or a central scar support the diagnosis of SCN ( Table 47-4 and Figure 47-5 ).
| Feature | Macrocystic SCN | MCN |
|---|---|---|
| Sex | Female/male (75%/25%) | Female almost exclusively |
| Age | 6th-7th decades | 4th-5th decades |
| Location | Head/body/tail | Body/tail 85% |
| Shape | Lobulated | Oval |
| Wall | Absent | Present, usually thick |
| Number of loculations | >6 | <6 |
| Coexisting microcysts (<2 cm) | Present | Absent |
| Central scar | May be found | Absent |
| Calcifications | Central if present | Peripheral |
More than six loculations, a loculation diameter less than 2 cm, a central scar, and absence of channel-like communication with the MPD are useful to differentiate SCNs from branch duct IPMNs ( Table 47-5 ).
| Feature | SCN | BD-IPMN |
|---|---|---|
| Sex | F/M (75%/25%) | M > F (60%/40%) |
| Age | 6th-7th decades | 6th-7th decades |
| Morphology | Lobulated | Uncommonly lobulated |
| Scar | Central | Absent |
| Loculations | Smaller | Larger |
| Main pancreatic duct communication | No | Present |
Treatment
Surgical Treatment.
SCNs are usually regarded as benign, slowly growing lesions, with an estimated growth of 4 to 12 mm per year. The decision to operate is often based on size at presentation, patient age, clinical presentation, and location. In younger patients, owing to the increase in size over time, lesions larger than 4 cm are usually resected. In other cases the cysts should be observed with imaging at 6-month intervals for the first year, then annually for a period of 3 years. If the cyst remains stable and the patient is symptom free, no further workup may be needed.
- •
SCNs are regarded as benign lesions, usually occurring in asymptomatic middle-aged women.
- •
They may grow over time; therefore, if detected in young patients with a size at presentation of 4 cm or greater, surgery is advocated; in other cases they should be observed by periodic imaging.
- •
MRI is the most accurate noninvasive diagnostic modality for SCNs.
- •
Macrocysts are difficult to characterize with imaging; therefore, if lobulations or a central scar is not detected, these patients should undergo EUS and/or biopsy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree