Air Space Disease
Acute Consolidation
Air space consolidation results when alveoli are filled with material that attenuates the x-ray beam more than the surrounding lung parenchyma. When encountered in the acute setting, air space consolidation on imaging has a differential diagnosis ( Table 14.1 ).
| DIAGNOSIS | COMMENTS |
|---|---|
| Infectious pneumonia | Alveoli are filled with pus or inflammatory fluid; fever and leukocytosis are often present |
| Aspiration of gastric contents | There is a predilection for gravity-dependent lung segments; Mendelson syndrome refers to chemical pneumonitis from aspiration during obstetric anesthesia |
| Alveolar pulmonary edema | Cardiomegaly and pleural effusions may be present; acute respiratory distress syndrome may be considered in severe cases |
| Pulmonary hemorrhage | Consider pulmonary contusion in the setting of trauma; there may be a history of anticoagulation or bleeding diathesis |
| Pulmonary infarction | Risk factors for pulmonary embolism are present; consolidation is peripheral and is more likely in the lower lobes |
Chronic Consolidation
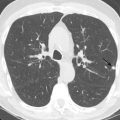
When air space consolidation persists on imaging studies over several weeks to months, the differential diagnosis shifts away from pneumonia, which typically has a more acute presentation, with subsequent resolution. Infectious, inflammatory, and neoplastic causes should be considered when chronic air space consolidation is encountered ( Fig. 14.1 ), and the most common entities are outlined in Table 14.2 .
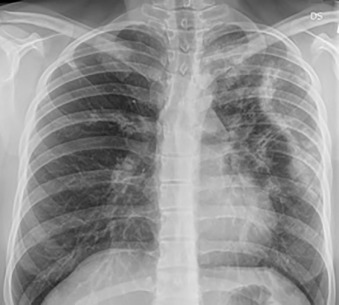
| DIAGNOSIS | COMMENTS |
|---|---|
| Organizing pneumonia | Reverse halo sign seen in up to 20%; consolidation typically responds to steroid therapy |
| Chronic eosinophilic pneumonia | Often elevated eosinophils in bronchoalveolar lavage fluid and peripheral blood; upper lobe predominant consolidation; typically responds to steroid therapy |
| Poorly treated infection | May be due to suboptimal antibiotic choice or resistance; also consider fungal and mycobacterial infection |
| Adenocarcinoma | Air bronchograms may be seen |
| Sarcoidosis | Confluent granulomatous nodules may mimic consolidation |
| Lymphoma | Usually derived from bronchus-associated lymphoid tissue; air bronchograms may be seen |
| Lipoid pneumonia | Demonstrates areas of fat attenuation; generally indolent course |
| Churg-Strauss syndrome | Nearly all patients have asthma and eosinophilia |
Pulmonary Nodules
Solitary Pulmonary Nodule
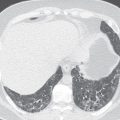
The differential diagnosis of a solitary pulmonary nodule on chest radiography or CT is broad, but more than 95% of the time the nodule is an infectious granuloma ( Fig. 14.2 ), malignant neoplasm (primary or metastatic), or benign tumor (e.g., a hamartoma). Nodule size, shape, density, calcification pattern, degree of contrast enhancement, fluorodeoxyglucose (FDG) avidity, and rate of growth are key imaging characteristics that when combined with clinical factors (e.g., patient age, immune status, clinical history) help narrow the differential diagnosis. Ultimately, this combination of factors may indicate a nodule’s likelihood of benignity versus malignancy or even provide a specific diagnosis. There are many possible causes of a solitary pulmonary nodule ( Table 14.3 ).
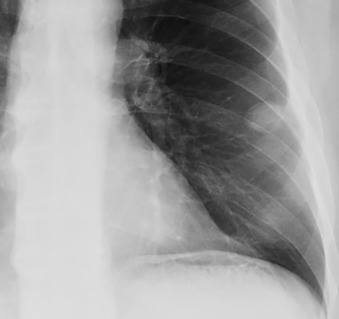
| DIAGNOSIS | COMMENTS |
|---|---|
| Granuloma | May be other calcified granulomas or calcified lymph nodes; likelihood dependent on where patient has lived (far more common in Midwestern United States) |
| Lung cancer | Calcification seen in 6%–7%; most lung cancers double in volume every 1–18 months |
| Metastasis | Patients with melanoma, testicular cancer, or sarcoma more likely to have a solitary metastasis than primary lung cancer |
| Intrapulmonary lymph node | Often found along fissures or in a subpleural location; morphology may be triangular, oval, round, or trapezoidal |
| Pulmonary nodule mimic | Consider clothing artifact (button), nipple shadow, skin or chest wall lesion, healing rib fracture, and enostosis in a rib |
| Hamartoma | Fat within a nodule nearly pathognomonic; popcorn calcification sometimes present |
| Pulmonary carcinoid | Location generally more central; can enhance avidly; high false-negative rate on FDG PET imaging |
| Infection (nongranulomatous) | Round pneumonia, septic embolus, or abscess |
| Mesenchymal or neural tumor | Consider fibroma, chondroma, lipoma, or neurofibroma |
| Arteriovenous malformation | Feeding artery and draining vein; multiple in hereditary hemorrhagic telangiectasia |
| Inflammatory nodule | Rheumatoid nodule or granulomatosis with polyangiitis |
Multiple Pulmonary Nodules
Centrilobular Distribution
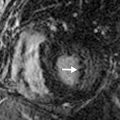
Diseases that involve the centrilobular bronchioles, pulmonary arterioles, or lymphatics may produce multiple centrilobular nodules. These nodules characteristically are evenly spaced from one another and spare the interlobular septa and pleural surfaces ( Fig. 14.3 ). Nodules in this distribution may be solid or of ground-glass attenuation, and the most common entities producing this pattern are outlined in Table 14.4 . If the nodules are branching, with a V or Y shape, it is termed a tree-in-bud pattern .
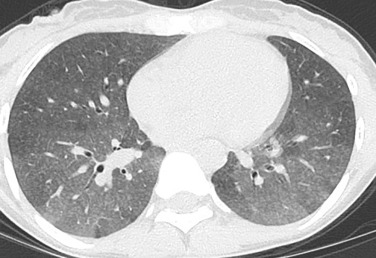
| DIAGNOSIS | COMMENTS |
|---|---|
| Hypersensitivity pneumonitis | Ground-glass nodules; exposure to organic antigen identified in 50%; air trapping usually present |
| Respiratory bronchiolitis | Upper lobe predominant ground-glass nodules; history of cigarette smoking |
| Pulmonary arterial hypertension; pulmonary capillary hemangiomatosis; pulmonary veno-occlusive disease | Ground-glass nodules; due to pulmonary edema and/or hemorrhage; dilated main pulmonary trunk |
| Metastatic calcification | Upper lobe ground-glass nodules ± calcification; hypercalcemia |
| Endobronchial infection, bronchopneumonia | Solid or ground-glass ± tree-in-bud opacities; bacterial, viral, or fungal causes |
| Follicular bronchiolitis | Solid or ground-glass nodules ± tree-in-bud opacities; immunosuppression or connective tissue disease |
| Pneumoconioses | Solid centrilobular nodules; especially siderosis and coal workers’ pneumoconiosis |
| Langerhans cell histiocytosis | Upper lobe predominant solid or ground-glass nodules; early in disease course; history of cigarette smoking |
Perilymphatic Distribution
On CT imaging, diseases that involve the perilymphatic channels are typically subpleural and are along bronchovascular bundles, interlobular septae, and fissures. Unlike centrilobular nodules, nodules with a perilymphatic distribution are generally unevenly spaced from one another. There are several diagnostic considerations for nodules with a perilymphatic distribution ( Table 14.5 ).
| DIAGNOSIS | COMMENTS |
|---|---|
| Pulmonary sarcoidosis | Concomitant symmetric hilar and mediastinal lymph nodes; patients commonly between 20 and 40 years of age |
| Silicosis and coal workers’ pneumoconiosis | Nodules range from 1–10 mm in diameter; nodules have an upper lobe and posterior distribution |
| Lymphangitic carcinomatosis | Adenocarcinoma (lung and breast) most common cause; associated pleural effusions are common |
| Nodular pulmonary amyloidosis | Patients usually in sixth decade of life and often asymptomatic; lower lobe and peripheral distribution often appreciated |
| Lymphocytic interstitial pneumonia | Associated with HIV (in children) and Sjögren syndrome; scattered thin-walled perivascular cysts often present |
Random Distribution
Multiple pulmonary nodules that are diffusely distributed and without architectural predominance—that is, lack a centrilobular or peribronchovascular distribution—are likely random. These nodules may be variable in number and density, and often the underlying pathophysiologic mechanism is hematogenous dissemination. There are several diagnoses to consider when pulmonary nodules are found to have a random distribution ( Table 14.6 ).
| DIAGNOSIS | COMMENTS |
|---|---|
| Hematogenous metastases | Lower lobe predominance; metastases may be small in size, as from thyroid cancer, or may be large (cannonball), as from seminoma |
| Fungal infection | Can be miliary or larger; histoplasmosis, blastomycosis, and coccidioidomycosis; aspergillosis or zygomycosis in the immunocompromised |
| Miliary tuberculosis | May be seen in primary or postprimary infection; miliary nodules imply active infection and poor prognosis |
| Septic emboli | Peripheral and lower lobe predominance ± cavitation |
| Adenocarcinoma of lung | Rarely presents as multiple nodules in a random distribution |
| Rheumatoid necrobiotic nodules | Nodules wax and wane with rheumatoid arthritis activity; miliary nodularity occurs early, prior to pulmonary fibrosis |
| Amyloidosis | Nodules may cavitate and/or show calcification |
| Granulomatosis with polyangiitis | Nodules ± cavitation |
| Benign metastasizing leiomyoma | Often a history of hysterectomy for uterine leiomyomas; nodules enhance with contrast and may cavitate |
Calcified Pulmonary Nodules
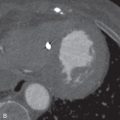
A variety of disease entities can present with multiple calcified pulmonary nodules, with causes that include neoplastic, infectious, metabolic, and inhalational factors ( Fig. 14.4 ). Clinical history and the size, appearance, and distribution of the nodules can add specificity. The most common entities producing multiple calcified pulmonary nodules are outlined in Table 14.7 .
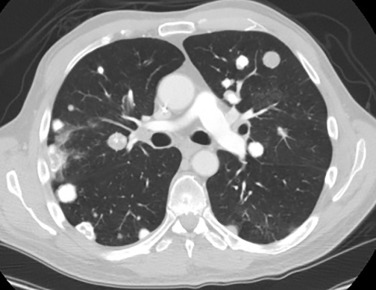
| DIAGNOSIS | COMMENTS |
|---|---|
| Chronic granulomatous disease | Tuberculosis and histoplasmosis most common; may be associated findings such as calcified lymph nodes |
| Metastatic disease | History of sarcoma or medullary thyroid cancer; mucinous adenocarcinoma metastases may calcify; treated choriocarcinoma metastases may calcify |
| Pneumoconioses | Particularly silicosis and coal workers’ pneumoconiosis |
| Healed varicella pneumonia | Generally 2–3 mm in size; diffuse distribution |
| Pulmonary alveolar microlithiasis | Sandlike calcifications, often ≈1 mm in size; lower lobe predominant |
| Metastatic calcification | Upper lobe predominant; centrilobular ground-glass nodules; hypercalcemia |
| Pulmonary amyloidosis | May be associated with tracheobronchial amyloidosis |
| Multiple chondromas | One component of the Carney triad—paragangliomas, gastrointestinal stromal tumors, pulmonary chondromas |
Cavitary Pulmonary Nodules
A cavitary lung nodule usually contains air, but may also contain blood, pus, or debris. A cavitary nodule has a definite wall with thickness of at least 4 mm and should not be confused with a cyst, which is a thin-walled, air-containing lesion. Chest radiography may identify cavitary nodules, although CT offers increased sensitivity and provides improved lesion localization and characterization. There are several entities to consider when cavity pulmonary nodules are encountered ( Table 14.8 ).
| DIAGNOSIS | COMMENTS |
|---|---|
| Pneumonia and lung abscess | Typically from Staphylococcus aureus, Pseudomonas aeruginosa , or Klebsiella pneumoniae; tuberculosis and fungal pneumonia also cavitate |
| Metastases | Usually a primary squamous or transitional cell carcinoma; nodules often have round, well-demarcated borders |
| Primary lung cancer | Squamous cell carcinoma most likely to cavitate; cavitation most likely when mass is >3 cm |
| Pulmonary emboli | Lower lobe and peripheral distribution; septic emboli cavitate more commonly than bland emboli |
| Granulomatosis with polyangiitis | Cavitation occurs in nearly 50% of patients with nodules; nodule size variable; may be associated with consolidation |
| Rheumatoid lung nodules | Occur in <1% of rheumatoid arthritis patients; more common in men and in upper and middle lung zones |
| Congenital lesions | Congenital pulmonary airway malformation; pulmonary sequestration |
Pulmonary Masses
Anterior Mediastinal Masses
Because the anterior mediastinum houses the thymus, lymph nodes, vessels, and fat, masses that arise in this space originate from these structures. These masses may be recognized as mediastinal widening on a frontal radiograph or as loss of the retrosternal clear space on a lateral radiograph, but they are best evaluated on cross-sectional imaging ( Fig. 14.5 ). The most common masses arising in the anterior mediastinum are outlined in Table 14.9 .
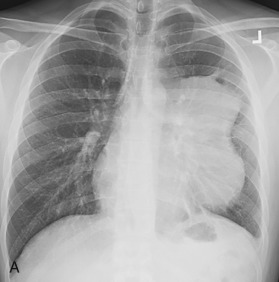
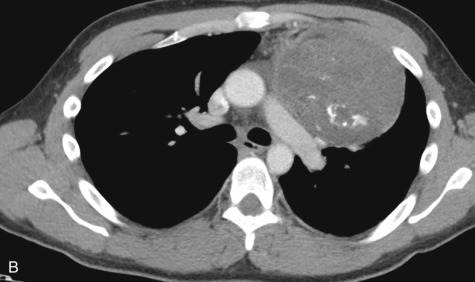
| DIAGNOSIS | COMMENTS |
|---|---|
| Thymic masses | Thymoma, thymic carcinoma, thymolipoma, thymic cyst; rarely, thymic carcinoid or thymic lymphoma |
| Lymphoma or lymphadenopathy | Lymphoma more common in younger patients; adenopathy from metastases or granulomatous disease |
| Germ cell tumors | More likely to contain fat and/or calcification; more likely to maintain a midline anatomic position |
| Thyroid masses | Substernal thyroid goiter usually continuous with thyroid; thyroid adenoma and carcinoma less common |
| Vascular masses | Ascending aortic aneurysm |
| Congenital cysts | Bronchogenic cyst or lymphangioma |
| Neural tumors | Arise from vagus or phrenic nerve |
| Foramen of Morgagni hernia | Sometimes demonstrated on fluoroscopic studies |
Middle Mediastinal Masses
The middle mediastinum is bounded by the thoracic inlet superiorly, the diaphragm inferiorly, the pericardium anteriorly, and the posterior tracheal wall (or, according to some authors, the esophagus) posteriorly. It contains the trachea, azygos vein, vena cavae, aortic arch, pulmonary vessels, lymph nodes, and (according to some authors) the esophagus. It also contains the phrenic, vagus, and left recurrent laryngeal nerves. Radiographic findings suggestive of a middle mediastinal mass include widening of the paratracheal stripe, convexity of the aortopulmonary window, displacement of the azygoesophageal recess to the right, and appearance of a left pseudoparavertebral line. Several masses arise in the middle mediastinum ( Table 14.10 ).
| DIAGNOSIS | COMMENTS |
|---|---|
| Foregut duplication cyst | Includes esophageal duplication and bronchogenic cysts; usually contains simple fluid |
| Lymphoma or lymphadenopathy | Lymphoma more common in younger patients; adenopathy from metastases or granulomatous disease |
| Vascular abnormality | Aortic aneurysm may present as a middle mediastinal mass; right-sided aortic arch may appear as a “mass” |
| Hiatus hernia | May be sliding, paraesophageal, or mixed hernia; may be retrocardiac air-fluid level on chest radiography |
| Pulmonary artery enlargement | Seen in pulmonic stenosis or pulmonary artery hypertension |
| Pericardial cyst | May change in shape with inspiration and patient position; may be congenital or result from prior pericarditis |
| Tracheal lesions | Intrinsic and extrinsic lesions occupy middle mediastinum |
| Germ cell tumors | Rarely in middle mediastinum (more common anteriorly); often contain fat and/or calcification |
| Neural tumors | Arise from vagus, phrenic, or left recurrent laryngeal nerve |
Posterior Mediastinal Masses
Neurogenic tumors are the most common cause of a posterior mediastinal mass ( Fig. 14.6 ), comprising 75% of primary posterior mediastinal neoplasms. Neurogenic tumors usually arise from peripheral nerves or sympathetic ganglia; the most likely tumor type is dependent on the patient’s age. Nonneoplastic masses in the posterior mediastinum include esophageal masses, duplication cysts, paraspinal abscess, and other types, the most common of which are presented in Table 14.11 .
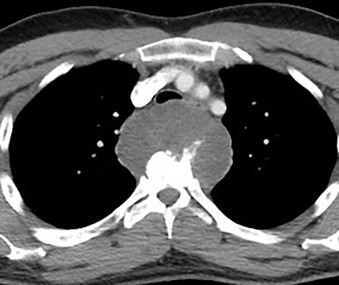
| DIAGNOSIS | COMMENTS |
|---|---|
| Neurogenic nerve sheath tumor | Schwannomas and neurofibromas are benign; malignant peripheral nerve sheath tumors in neurofibromatosis patients |
| Neurogenic sympathetic ganglion tumor | Neuroblastoma in young children; ganglioneuroma affects older children and young adults |
| Nonneurogenic tumor | Includes chordoma and pheochromocytoma |
| Descending aortic aneurysm | Vascular origin apparent on cross-sectional imaging |
| Esophageal mass | May be benign such as leiomyoma; malignancies include squamous cell carcinoma and adenocarcinoma |
| Paraspinal abscess | Bacterial, fungal, or tuberculous causes most common |
| Extramedullary hematopoiesis | History of hemoglobinopathy or bone marrow infiltration; smooth, lobulated, and often bilateral paraspinal masses |
| Lateral thoracic meningocele | 75% of cases associated with neurofibromatosis; continuity of cerebrospinal fluid between meningocele and thecal sac |
| Duplication cyst | Neurenteric cyst or esophageal duplication cyst; usually fluid attenuation on CT unless infected or hemorrhagic |
| Lymphoma or lymphadenopathy | Usually accompanied by enlarged lymph nodes elsewhere |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree