Multiple sclerosis (MS) is considered the most common inflammatory autoimmune neurologic disorder and the most frequent cause of nontraumatic neurologic disability in young and middle-age adults. This article reviews the basic features of its magnetic resonance (MR) imaging lesions and, primarily, the use of diffusion MR imaging, which is increasingly applied to assess patients with MS, not only to investigate plaques but also the normal-appearing white matter, gray matter, optic nerve, and spinal cord, because of its ability to detect and quantify disease-related pathologic conditions of the central nervous system.
Multiple sclerosis (MS) is considered the most common inflammatory autoimmune neurologic disorder, involving especially the white matter (WM), the main pathologic features of which include a primary perivascular inflammation, demyelination, gliosis, and axonal injury. MS is a chronic disease, estimated to affect 2.5 million people worldwide, and almost 400,000 persons in only the United States. Women are affected twice to 3 times as frequently as men, and it is uncommon in children, accounting for only about 0.3% to 0.4% of all cases. The symptoms begin most frequently during the third and fourth decades. However, MS can develop after age 50 years, accounting for 10% of cases. MS is the most frequent cause of nontraumatic neurologic disability in young and middle-age adults. Thus, early diagnosis is required to promptly begin a more effective treatment.
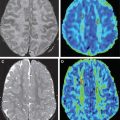
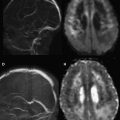
Conventional magnetic resonance (MR) imaging has become a primary tool for the investigation of MS and for clinical diagnosis over the past 3 decades, since its introduction. MR scans offer the most sensitive way to identify MS lesions and their changes, for example disease accumulation and disease activity. Therefore, the use of MR imaging has had a major effect on early and more precise diagnosis as well as on treatment management, predicting the prognosis of patients ( Fig. 1 ).
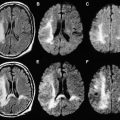
The role of MR imaging in the diagnosis of MS has been strengthened with the introduction of the McDonald criteria, because this modality is able to determine dissemination in both time and space of the demyelinating lesions. The McDonald criteria include the Barkhof-Tintore MR imaging criteria, because a spinal cord lesion can substitute for any of the brain lesions. Nevertheless, in the light of subsequent studies, new insights were added and the previous proposed 2001 criteria were revised at the reconvened international panel during 2005. The Barkhof-Tintore MR imaging criteria were kept in the new diagnostic MS criteria. However, the 2 criteria differ in the extent to which a spinal cord lesion can also assist with fulfillment of dissemination in space.
As mentioned earlier, conventional MR imaging offers the most sensitive way to detect demyelinating plaques and their histopathologic changes over time. It is also useful in helping to determine the diagnosis of clinically defined MS as well as to rule out other conditions that may resemble the disease. Although high sensitivity has been described, the MR specificity is low and these findings may resemble other pathologic conditions, such as those secondary to atherosclerosis. MR imaging contributes decisively to the management of patients with MS, adding useful information regarding conversion to clinically definitive MS, earlier diagnosis, disease prognosis, and treatment response. MR imaging is the most versatile technique for depicting MS lesions. More recently, some investigators have suggested that diffusion tensor imaging (DTI) could play an important role in the diagnosis of certain patients, who present with few lesions and more diffuse WM involvement depicted only with fractional anisotropy (FA) measurements.
Nevertheless, predicting the prognosis is not easy, and it depends on several factors. MR imaging could be used to provide certain information relevant for this purpose. Atrophy can be useful, but we also need measures of diffuse damage to the normal-appearing WM (NAWM), and even to the normal-appearing gray matter (NAGM), using DTI for instance.
Diffusion MR imaging
Diffusion MR imaging (diffusion-weighted imaging [DWI]) has been used in the clinical scenario for the last 2 decades. Since its clinical usefulness in detecting hyperacute infarcts was established, DWI has been studied extensively in the evaluation of other neurologic conditions, such as brain neoplasm, brain abscess, and degenerative diseases.
An increasing improvement in the assessment of MS has been seen in the last few years, leading to a better understanding of its immunopathogenesis, pathologic conditions, and genetics. Because of its ability to detect and quantify disease-related pathologic conditions of the central nervous system, DTI has increasingly been applied to assess patients with MS, not only to investigate plaques but also the NAWM and more recently the gray matter (GM), optic nerve, and spinal cord.
Physical basis
Diffusion Sensitization
DWI is based on the random or Brownian motion of water molecules. In the brain, the presence of some tissue structures restricts free water motion. MR imaging makes it possible to estimate the diffusivity of water molecules. Most diffusion measurements are made using a variant of the Stejskal and Tanner sequence, by applying the 2 equal and opposite gradient pulses (compensating for the effects of uniform gross movement like flow), to determine the amount of signal loss, as a function of the magnitude of diffusion. Echo-planar imaging spin-echo (SE) T2-weighted sequences are used, because of their ability to reduce motion artifacts and their rapid acquisition time. The signal intensity on DWI is influenced by both T2 tissue effect and the tissue diffusion characteristics. To exclude the T2 effect and to reflect solely the diffusion effect itself, an apparent diffusion coefficient (ADC) map is generated. Although the results of DWI seem to be promising, it is important to consider the multicenter variability of the method.
DTI
The magnitude of diffusion depends on the direction in which it is measured. A full characterization of diffusion can be obtained in terms of a tensor, a 3 × 3 matrix that accounts for the correlation existing between molecular displacements along orthogonal directions. From the quantitative parameters of DTI it is possible to derive ADC, mean diffusivity (MD), the average of the ADCs measured in the 3 orthogonal directions, as well as an anisotropy measure such as FA. FA is a quantitative measure of deviation from isotropy and reflects the degree of alignment of cellular structures and tissue coherence. Because interpreting a tensor representation can be nonintuitive, scalar metrics have been proposed to simplify DTI data. FA measures the fraction of the total magnitude of diffusion anisotropy, varying from completely isotropic diffusion, denoted as 0, up to complete anisotropic diffusion, denoted as 1.
MS Plaques
One can speculate that in the acute phase of the plaque formation, an expanded extracellular volume is caused by astrocyte proliferation, perivascular inflammation, and demyelination, all representing pathologic stigmata of the initial step in plaque formation, whereas in its chronic stage, a more pronounced increase in extracellular space represents axonal loss and tissue destruction.
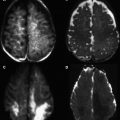
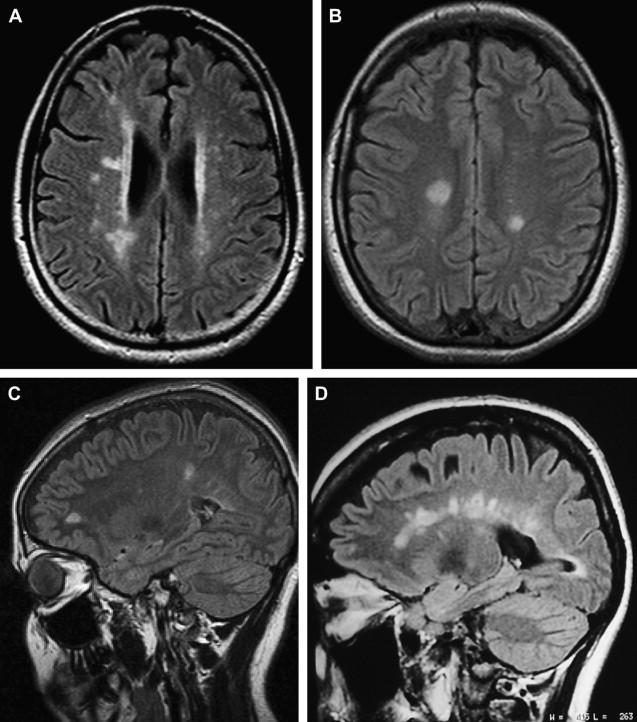
Although T2-weighted and fluid-attenuated inversion recovery (FLAIR) sequences are highly sensitive for MS lesions, they lack histopathologic specificity ( Fig. 2 ). This finding may be explained because inflammation, edema, demyelination, gliosis, and axonal loss are all represented as areas of high signal intensity on these sequences. Thus, they are not able to differentiate between different plaque subtypes. Mildly T1-weighted SE imaging can be more specific and can determine whether the lesion is acute or chronic. T1-weighted SE imaging seems to be more specific than T2-weighted imaging for identifying clinically relevant lesions. Hypointense lesions on T1-weighted SE, the so-called black holes, represent chronic plaques, which may correlate with disease progression and disability. Contrast-enhanced T1-weighted imaging is routinely used in clinical assessment of patients with MS to depict acute lesions. In the acute inflammatory phase, a disruption of the blood-brain barrier occurs, and an acute lesion appears first as a nodule-enhancing lesion and subsequently progresses to a ring-enhancing lesion. MR imaging can be 5 to 10 times more sensitive in depicting acute plaques, representing disease activity, than the clinical evaluation of relapses, which suggests that most of the enhancing lesions are clinically silent.
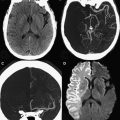
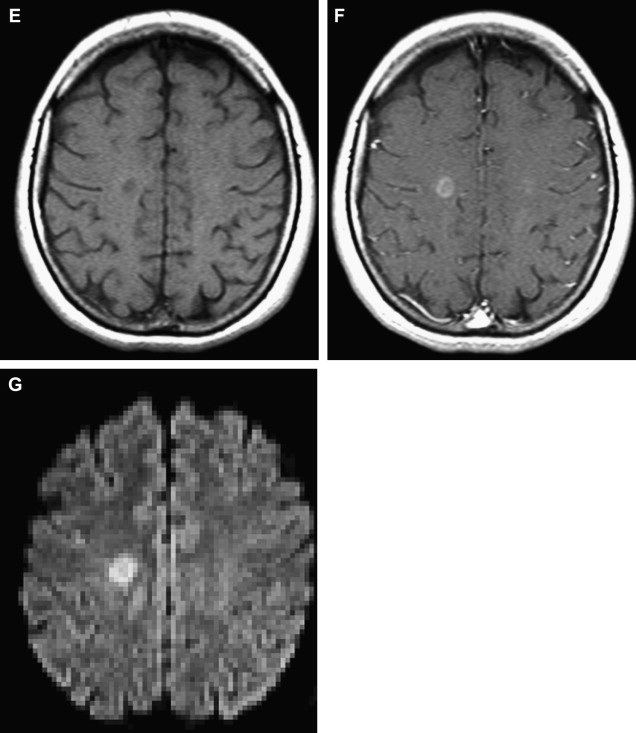
Diffusion MR imaging has been used to evaluate MS plaques. Some acute plaques may show restricted diffusion ( Fig. 3 ), mostly at their margin. This pattern of enhancement is not specific for demyelinating disease, but its diagnosis can be suspected, because it is frequently peripheral and discontinuous ( Fig. 4 ). This imaging characteristic may also be useful in the differential diagnosis with other expansive lesions, such as gliomas, which can present restricted diffusion within the solid portion of the lesion, secondary to high cellularity.
Diffusion MR imaging has been used to differentiate acute from chronic MS plaques, using ADC and FA. Although some investigators have used DWI and DTI to try to make such a distinction, the results are inconsistent. Initial papers suggested that ADC is increased within MS plaques compared with normal WM. Current evidence indicates that the pathologic substrate of MS lesions is different among MS subgroups and that disease progression may be associated with neuronal and axonal loss. Therefore the increase in diffusivity possibly reflects expanded free water content within the lesion, although the relative contribution from edema, demyelination, and axonal loss is difficult to determine. The highest diffusion values seem to be found in hypointense lesions, the so-called black hole, compared with enhancing lesions and isointense lesions. However, hypointensities on T1-weighted images do not solely represent tissue destruction of the late stages of the disease but may also be associated with acute lesions, resulting in enlargement of the extracellular space as a result of edema and inflammation, a process that is potentially reversible at follow-up. Acute edematous lesions were found to sometimes have even higher ADC values than chronic lesions. Thus, predicting activity of the disease based on the DWI findings, measured through ADC maps, is unreliable.
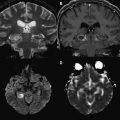
In general, MS lesions have decreased FA values on DTI when compared with the contralateral NAWM and normal control subjects ( Fig. 5 ). The reduction in the FA values within the plaques indicates the disruption of myelin and axonal structures that leads to disorganization and increased extracellular space. Some investigators reported that reduction in FA values observed in enhancing plaques is lower than in nonenhancing plaques, suggesting that DTI can indicate disease activity. FA values can also be found to be reduced in ring-enhancing plaques compared with homogenous enhancing lesions. However, conflicting results have been reported when FA values are used to differentiate acute from chronic plaques. Some reports were not able to depict any statistical difference between these 2 histopathologic MS plaque subtypes.
In an acute lesion tissue damage can be permanent, related to neurodegeneration, and on the other hand it can be transient, as observed in edema, demyelination, and remyelination. The postprocessing of DTI data may add new information regarding the cytoarchitecture and histopathologic alterations related to MS. The tensor model provides, besides the FA and MD, other measures of the diffusion direction. For each vector, a value known as an eigenvalue is attributed (λ 1 , λ 2 , and λ 3 ). Axial diffusivity (eigenvalue λ 1 ) is defined as the diffusion along the long axis and is higher than across the main fibers. It indicates a rate for the diffusion of water parallel to a bundle of axons or tract. Radial diffusivity is the one observed at the other 2 perpendicular plans, represented by the average of eigenvalue λ 2 and λ 3 , and represents the rate of the diffusion of water perpendicular to the axons. Demyelination in general leads to reduction in radial diffusivity values, whereas axonal damage leads to axial diffusivity alterations. Increased radial diffusivity relates to demyelination, and decreased radial diffusivity is seen during remyelination. Based on these findings regarding axial and radial diffusivities alterations related to histopathologic intrinsic characteristics of MS plaques, a recent study suggests that DTI can determine the plaque subtype: acute, subacute, or chronic. Such a distinction is hard to make using FA values and MD. Although these are preliminary results, the investigators suggest that eigenvalues may be more sensible than FA and MD measurement when the demyelinating process is the most important damage. In the acute plaque, the demyelinating process is predominant, with altered radial diffusivity. A mixed pattern of histopathologic damage is observed in chronic plaques, including demyelination, altering radial diffusivity, and axonal loss, altering axial diffusivity.
Clinical Correlation
Conventional MR imaging findings do not correlate well with clinical manifestations of disease activity. MS lesions on T2-weighted images are spatially and histopathologically nonspecific. Lesion load provides useful information in monitoring the disease and its treatment response. However, lesion load may not adequately account for the patient’s functional state. Nonetheless, brain atrophy correlates strongly with clinical deficiency, although the lesion load does not show such a correlation. Thus, destructive process underlying MS-related brain atrophy may not merely depend on the severity of T2 lesion load.
The increased use of MR imaging in the assessment of MS has disclosed that the extent of WM abnormalities measured using conventional MR imaging contributes only partially to the clinical manifestations of the disease, including accumulation of irreversible disability. The advent of new advanced MR imaging sequences may play an important role in the assessment of patients with MS and may therefore contribute to explaining the discordance between conventional MR imaging findings and clinical status, especially analyzing the NAWM and GM. Axonal damage is a key feature in MS lesions, occurring within both acute and chronic plaques, as well as in the NAWM. There is increasing evidence that axonal damage has a strong effect on permanent neurologic deficits. Increased GM disease might be an additional factor contributing to the presence and severity of cognitive impairment in patients with progressive MS. Some investigators have suggested that GM diffusivity changes may be viewed as a hallmark of the more disabling and progressive stages of the disease, because the accumulation of damage in focal lesions and NAWM occurs even during the earliest nondisabling phases of MS. GM diffusivity changes have also been related to neuropsychologic impairment in MS.
Despite disappointing preliminary data, diffusion imaging seems to present a better correlation with clinical disability. The pathologic damage detected by DTI in clinically eloquent NAWM regions is a significant factor contributing to MS disability. Overall, diffusion studies indicate that the severity of damage within T2-visible lesions and in the NAGM, as well as in clinically eloquent WM tracts, has a significant effect on neurologic disability. DTI measures may thus contribute to composite MR-based scores, explaining otherwise unexplained variance in MS-related disability.
NAWM
Conventional MR imaging has limitations in assessing lesion burden and delineating tissue damage, which are known to occur beyond the visible margins of MS plaques. However, the findings at conventional MR imaging do not correlate well with the specific pathologic or clinical course of MS. Recent evidence from histopathologic studies has shown that the disease processes of MS involve not only the plaques seen on conventional MR images but also the WM regions that seem to be normal. The extension of disease beyond the boundaries of plaques has important ramifications in disease assessment, because the quantification of disease burden is an important surrogate marker for disability progression. An imaging examination that depicts greater disease burden than that seen at conventional MR imaging could provide valuable additional information for both drug trials and patient treatment. With the advent of diffusion MR imaging as a quantitative MR technique, the ability to better detect and characterize the disease burden, including occult microscopic disease changes, has improved greatly. Although these results seem to be promising, it is also important to consider the multicenter variability in DTI measurements, which can be influenced by the type of scanner, the magnetic field strength, and the number of diffusion gradient pulse directions applied.
Several studies have shown diffusion changes in the NAWM in patients with MS, suggesting that the histopathologic abnormalities may be widespread. These diffusion abnormalities can be characterized as an increase in MD and ADC values, as well as a decrease in FA values, which is in agreement with findings from magnetization transfer ratio studies. Such alterations in the NAWM tend to be more severe in perilesional regions and in sites where visible MS plaques are usually located, suggesting subtle microstructural changes. Local alterations in DTI parameters may herald the future appearance of new focal lesions.
Although most of the studies show increased diffusivity and reduced anisotropy in the NAWM, some studies did not find any significant difference in these analyses. These disagreements can possibly be explained by the different forms of postprocessing of data obtained by DTI. A subsequent study with the same patient cohort using histogram analysis did report subtle abnormalities.
However, NAWM damage is only partially correlated with the extent of focal lesions and the severity of intrinsic damage, suggesting that diffusivity changes in NAWM are not entirely dependent on retrograde degeneration of axons transected in MR conventional visible plaques.
Similar alterations can perhaps already be observed in patients presenting with clinically isolated syndrome (CIS), suggestive of MS, using histogram analysis of segmented diffusion maps of the brain.
Some reports also showed DTI alterations in the normal-appearing corpus callosum, which may precede abnormalities in other NAWM regions, suggesting preferential occult injury to this location. The cumulative bridging effects of the corpus callosum resulting in Wallerian degenerative changes from the connecting distal WM plaques may play an important role in the relationship between the corpus callosum diffusion abnormalities and the cerebral lesion load. In agreement with these imaging findings, a postmortem MS study showed quantitative reduction in total number and attenuation of axons passing through the corpus callosum that grossly appeared normal.
GM
Recently, new insights have been added in GM assessment with the advent of modern scanners and advanced MR techniques. Postmortem studies have shown that cerebral GM is also pathologically involved in patients with MS. Cerebral GM lesions occur in many patients with MS. In addition to axonal injury, the presence of cortical plaques has been identified in several different types, according to their topography within the cortex. Microglial activation predominates in the characterization of cortical plaques, instead of infiltrating lymphocytes, which occurs to a lesser extent. Moreover, neuronal apoptosis, loss of dendritic arborization, and transected and demyelinated axons represent the involvement of neuroaxonal structures in the pathophysiology of the cortical plaques. The loss of dendrites and axons contributes to reducing the diffusion anisotropy even more. Retrograde degeneration of GM neurons secondary to WM damage is also likely to occur, contributing to the GM damage. The discrepancy regarding patient clinical status and lesion burden assessed by conventional MR sequences can be partially explained by the lack of depicting NAWM abnormalities and GM lesions as well.
Although histopathologic studies show cortical lesions, reliable detection of these lesions with conventional MR sequences is challenging. There is a low sensitivity in the detection of GM lesion because of their small size, partial volume effects with cerebrospinal fluid (CSF) and relaxation characteristics similar to the GM on T2-weighted and FLAIR images. Advanced MR imaging techniques, such as the double inversion recovery (DIR) sequence, are superior to conventional T2-weighted and FLAIR images ( Fig. 6 ), like other sequences (phase-sensitive inversion recovery) that better depict the cortical-WM junction. Both CSF and WM are selectively suppressed. DIR sequence has some limitations regarding its inherent sensitivity to noise, and flow-related artifacts, which can result in false-positive findings.
Using the DTI sequence to assess cortical lesions, geometric distortions may be observed, especially if these sequences use echo-planar imaging. To minimize this problem, DTI analysis of cortical lesions requires accurate coregistration of DW images to the images used for lesion detection. Advanced nonlinear image registration methods have the potential to reduce this artifact. DTI has shown GM involvement, which seems to be greater in the progressive form of MS. DTI may reflect the severity of cortical dysfunction, associated with memory and attention deficits in patients with MS, because a correlation between GM MD and cognitive impairment could be shown. Discordant results were reported in the FA value measurement of NAGM. Using bulk histogram analysis, FA values were reduced in NAGM. A more recent report showed an increase in mean FA values in cortical lesions in patients with MS when compared with healthy control subjects. However, results from the same patient cohort using histogram analysis of the entire GM showed a reduction in FA values, in agreement with previous reports, probably because of a widespread microstructural alteration. Conflicting results were described when GM damage in different MS phenotypes was assessed with DTI. Although a significant reduction in GM diffusivity was observed in patients with secondary progressive (SP) MS, no difference was noted in patients with relapsing remitting (RR) MS. These findings may point to a more extensive pathologic abnormality in progressive forms of MS than in patients with RRMS, supporting the concept that in part GM MS lesions are secondary to WM damage. This report is consisted with the more profound cognitive impairment found in patients with SPMS and with accelerating cortical atrophy in progressive MS.
Optic Nerve
Acute demyelinating optic neuritis is often the initial clinical manifestation of MS, and MS is the most common cause of optic neuritis. A clinical challenge arises when predicting which patients presenting with optic neuritis will develop MS. In absence of other clinical manifestations, optic neuritis is referred to as one of the CISs suggestive of MS. Typical findings on conventional MR imaging include a swollen nerve and hyperintensity on T2-weighted and short-tau inversion recovery images, which may enhance on gadolinium-enhanced T1-weighted images. Although these findings are typically related to optic neuritis, they are nonspecific because ischemia and infection can present similarly.
DTI is a surrogate marker of the structural integrity of the optic nerve, with increases in MD and reductions in FA in case of neuritis. DTI can also be applied to further detection of early optic nerve alterations, before abnormalities detected on conventional MR imaging. Despite the potential usefulness of DTI in the clinical scenario in the assessment of the optic nerve, its acquisition is still a technical challenge. This structure is small, mobile, and has a surface area susceptible to volume ratio, leading to increased partial volume contamination to bone and intraconal fat. Applying the gradient pulse in fewer directions, the diffusion sequence can be performed with a shorter acquisition time. Nonetheless, most previous studies concentrated on ADC measurement. ADC is increased in the diseased optic nerve and has a close correlation with visual activity and clinical measures of visual functioning. DTI measures the integrity of the axons in the optic nerve and is related to physiologic findings, like evoked potentials. Special DTI techniques may be required for examination of this structure. Reduction in the connectivity of the optic radiations was also shown in patients with optic neuritis, suggesting a mechanism of transsynaptic degeneration secondary to the damage.
Pediatric MS
MS is an increasingly recognized condition in the pediatric population. Although MS typically affects young adults in the second and third decades, it has been increasingly recognized to occur in children. Clinical symptoms related to MS can occur before the age of 18 years in about 10% of cases. Although the underlying pathophysiology of disease does not vary with age, it seems that MS in the pediatric population has a more inflammatory course. Children have accrued physical disability more slowly than adults. Nonetheless, they have significant cognitive dysfunctions.
In a previous report, only a slight increase of ADC in the NAWM was found. In another study, an increased ADC and decreased FA values in the NAWM of pediatric patients with MS compared with healthy controls was reported. A more recent study concluded that more diffuse damage is present in the early stages of pediatric MS, because DTI measures indicate abnormalities in the WM-containing lesions or not in the principal pathways. The absence or mildness of DTI abnormalities in the NAWM may explain a favorable clinical status in the pediatric MS population. Because DTI has an exquisite ability to depict subtle alterations in the NAWM during the early course of the disease, it can contribute to the management of patients and serve as an outcome in clinical trials. Moreover, DTI may also be useful to assess the effect of MS on specific neurologic functions.
Diffusion Tractography
Recently, DTI tractography has been used to better depict more subtle abnormalities related to MS in main fiber tracts. DTI tractography may increase the specificity of MR imaging findings, thereby improving the strength of their relationship with clinical findings. DTI postprocessing techniques, such as tract-based analysis, provide an opportunity for further studies of the connectivity and function of the main fiber tracts. However, streamline algorithms have difficulty in passing through the GM, areas where fibers cross, and through damaged WM, because of uncertainty in the principal diffusion direction within the voxel when FA decreases. Fiber tract disruption caused by the transection of lesions can be directly visualized on fiber tractography. Such lesions may be associated with significantly fewer fiber tracts and lower FA. Fiber tract loss observed in the corticospinal tract according with lesion load supports the concept of Wallerian degeneration and axonal transection and correlates with clinical measures of locomotor disability. Moreover, DTI tractography may show that patients presenting with CIS have increased diffusivity and lesion load in the corticospinal tract. DTI tractography can detect subtle pathologic abnormalities beyond macroscopic lesions in WM main fiber tracts in MS and in addition may also increase the specificity in the assessment of specific neurologic deficits. Tract-based DTI analysis uses spatial normalization with subsequent group comparison. Thus, results obtained from this technique should be interpreted conservatively.
High field
High-field MR systems provide a higher signal-to-noise ratio (SNR), allowing the acquisition of thinner and volumetric sections, with higher spatial resolution as well as increasing contrast-to-noise. These findings may contribute to increasing the sensitivity for detection of MS plaques ( Fig. 7 ). Moreover, the susceptibility effects in high-field strength scanners are boosted, allowing an improvement in the appreciation of microvascularity and its relationship with MS plaques.
These new insights, regarding the assessment of patients with clinically suspected MS, increase the sensitivity for lesion detection, and may allow an earlier definite diagnosis and initiation of appropriate treatment, which may in turn influence clinical management. Thus, high-field MR imaging further strengthens the role of MR imaging in the study of MS.
The higher SNR provided by high-field MR systems can achieve higher-quality DTI. Although high-field MR systems have contributed to the evaluation of patients with MS, the susceptibility-induced signal intensity loss and distortion near the skull base and air sinuses is also prominent and should be carefully considered in clinical applications. Nonetheless, the improvement in radiofrequency coils and sequence acquisition has the advantage of performing diffusion imaging at higher field strengths, achieving increased SNR for more detailed compounds that might be obscured in lower field scanners.
Technical Developments
The best acquisition and postprocessing strategies for diffusion MR imaging studies in patients with MS remain a matter of debate. Some drawbacks are associated with DTI in the assessment of multiple fibers crossing within a single voxel. The interscanner variability and issues related to high-field MR system acquisition may also alter the results regarding the analysis of specific areas in the brain. Moreover, DTI tractography still has some technical limitations, which may lead to erroneous fiber tracking and FA analysis. Higher-strength MR scanners and multiple-receiver coils with parallel imaging data acquisition techniques have the potential to improve DTI acquisition because they can provide a better SNR, enhancing spatial resolution. A parallel imaging approach can reduce the distortions seen in echo-planar imaging, but has the disadvantage that the SNR is poorer. Periodically rotated overlapping parallel lines with enhanced reconstruction also allows the acquisition of diffusion imaging at high spatial resolution with minimal distortion.
Diffusion spectrum imaging is a novel approach developed to minimize the adverse effects in the crossing fibers within a single voxel. With this technique, it is possible to more accurately detect variations in diffusion along different directions, measuring the diffusion coefficient at high angular resolution.
There are other approaches that do not require any assumptions about diffusion within a voxel, such as q-space formalism, which measures the displacement profile. It has been used to estimate the three-dimensional displacement distribution of water molecules, allowing the characterization of the slow restricted diffusion component, which is probably mainly dependent on axonal membranes and modulated by myelin layers. In principle, this approach improves the ability to differentiate between demyelination, axonal loss, and inflammation in MS.
Other approaches measure the diffusion along different directions with a high angular resolution. The resulting three-dimensional representation of diffusivity can be broken down into a set of orthogonal three-dimensional functions, the so-called spherical harmonics.
The knowledge of the underlying anatomy is a prerequisite to a better analysis of pathologic changes in tissue microstructures, to avoid misinterpretation of the postprocessed DTI data. When comparing DTI data in patients with those in healthy controls, therefore, it is imperative to consider regions that match anatomically. Recently, several methods have been described for handling this uncertainty in a probabilistic fashion. The subjectivity of placing a region-of-interest approach to collect information for analyzing DTI data can be replaced by histogram analysis of MD and FA maps. A more elegant approach is to perform nonlinear spatial normalization of DTI data in an anatomic reference frame to facilitate the analysis and allow a voxel-based assessment that does not require a priori hypothesis. Registration may also be improved by matching based on the whole of the DTI, rather than just a simple image intensity.
The contribution of such new DTI technical acquisition in the analysis of patients with MS is promising but still needs to be validated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree