Introduction
Color and pulsed Doppler evaluation of the mesenteric arteries is performed to assess for compromise of intestinal blood flow in patients presenting with chronic, unexplained, and atypical abdominal pain. This examination includes evaluation of the abdominal aorta and the celiac, superior mesenteric (SMA), and inferior mesenteric (IMA) arteries. Doppler ultrasonography can accurately assess anatomic and physiologic features that are important for patient management including vessel patency, atherosclerotic burden, degree of arterial stenosis, and the number of arteries affected. Similar to assessment of the renal arteries, these studies are technically challenging and rely on operator experience and expertise. This chapter provides a review of mesenteric anatomy and physiology, technical factors, diagnostic criteria, and insights that allow for the successful evaluation of the mesenteric arteries.
Anatomy, Physiology, and Natural History of Bowel Ischemia
Anatomy
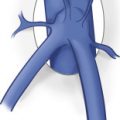
A thorough understanding of the anatomy of the mesenteric circulation provides a foundation for the interpretation of the sonographic examination. The mesenteric arterial circulation is comprised of the celiac, superior mesenteric, and inferior mesenteric arteries ( Fig. 26.1 ), all of which arise from the abdominal aorta. The abdominal aorta is a continuation of the thoracic aorta and extends from the aortic hiatus of the diaphragm to the aortic bifurcation. The average male abdominal aorta measures approximately 13 cm in length, up to 27 mm in anteroposterior diameter in its proximal portion, and tapers down to 13 mm in anteroposterior diameter at the level of the bifurcation. The celiac artery is the first major branch of the abdominal aorta and arises from the ventral surface of the aorta at the level of the T12 and L1 vertebral bodies. It courses anteroinferiorly before branching into the common hepatic, splenic, and left gastric arteries. The celiac artery supplies blood to the solid visceral organs (liver, pancreas, and spleen), the stomach, and proximal small bowel. The SMA originates approximately 1 cm inferior to the celiac artery, at the L1 vertebral level, and supplies blood to the bowel from the duodenum to the splenic flexure. The IMA is the smallest mesenteric vessel and originates from the left anterolateral aspect of the abdominal aorta, approximately 4 cm above the aortic bifurcation at the L4 vertebral level. It supplies the descending and rectosigmoid colon. Note that both the right and left renal arteries arise from the lateral aspects of the abdominal aorta between the SMA and the IMA. The mesenteric arteries may have variant anatomy in approximately 20% of the population, which may lead to misinterpretation of Doppler examination findings. The most common variant is a replaced right hepatic artery, in which the right hepatic artery originates from the SMA rather than the common hepatic artery (a branch of the celiac artery). This anomaly is seen in up to 17% of the general population. On gray-scale and color Doppler imaging, the replaced right hepatic artery may be seen arising from the SMA and coursing into the liver ( Fig. 26.2 ). On pulsed Doppler interrogation, the examiner may observe a low-resistance blood flow pattern in the SMA, a feature that is more characteristic of the celiac artery. Other common anatomic variants include an anomalous common hepatic artery that originates from the SMA (2% to 3% of cases) or from the aorta (1% to 2% of cases); and a common origin of the celiac artery and the SMA, termed the celiacomesenteric artery, which originates from the abdominal aorta and is seen in less than 1% of the population ( Fig. 26.3 ).






A rich collateral network exists between the mesenteric arteries via the mesenteric arcades and the marginal artery of Drummond, which ensures continuous perfusion of the organs that these vessels supply. Additional vascular protection is provided from communicating pathways between the three mesenteric arteries. For example, communication between the celiac artery and the SMA occurs by way of the gastroduodenal artery (also known as the pancreatoduodenal arcade; Fig. 26.4 ). The superior and inferior mesenteric arteries are connected by the arc of Riolan and the marginal artery of Drummond. The arc of Riolan, also known as the “meandering mesenteric artery” connects the middle colic artery, a branch of the SMA, with the left colic artery, a branch of the IMA ( Fig. 26.5 ). The marginal artery of Drummond represents a continuous arterial arcade along the inner margin of the colon, formed by anastomoses of the terminal branches of the SMA (ileocolic, right colic, and middle colic arteries) and IMA (left colic and sigmoid arteries) ( Fig. 26.6 ). Additional anastomoses exist between the IMA and branches of the internal iliac arteries, specifically via the superior rectal artery (a distal continuation of the IMA), the middle rectal artery (a branch of the internal iliac artery), and the inferior rectal artery (a branch of the internal pudendal artery) ( Fig. 26.7 ).




Given this extensive collateral circulation, patients may remain asymptomatic despite the presence of underlying mesenteric arterial disease. Mesenteric stenosis or occlusion of a single vessel may not produce symptoms in the setting of a patent collateral network. In general, severe compromise (≥70% stenosis or occlusion) of at least two of the three mesenteric arteries is required for symptoms of mesenteric ischemia to be present. This “two-vessel rule” holds in most patients and is utilized clinically for the diagnosis of chronic mesenteric ischemia.
- •
The mesenteric arterial circulation is comprised of the celiac, superior mesenteric, and inferior mesenteric arteries.
- •
The mesenteric arteries may have variant anatomy in up to 20% of the population, which may lead to misinterpretation of Doppler examination findings. The most common variant is a replaced right hepatic artery, in which the right hepatic artery originates from the SMA rather than the common hepatic artery.
- •
The rich collateral network between the mesenteric arteries via the mesenteric arcades and the marginal artery of Drummond, and direct pathways between the three mesenteric arteries ensure continuous perfusion of the organs that these vessels supply. Patients may remain asymptomatic despite the presence of underlying mesenteric arterial disease.
- •
Vascular compromise of at least two of the three mesenteric arteries is required for symptoms of chronic mesenteric ischemia to be present.
Physiology
Normal blood flow patterns differ between the celiac artery and the mesenteric arteries ( Fig. 26.8 ). The celiac artery supplies blood to the low-resistance vascular beds of the liver and spleen. The waveforms of the celiac artery and its branches demonstrate a low-resistance pattern with high end-diastolic velocities seen on pulsed Doppler imaging ( Fig. 26.9 ). This low-resistance flow pattern is because of the continuous forward flow of blood during both systole and diastole necessary to meet the high oxygen demands of the liver and spleen. The low-resistance blood flow pattern in the celiac artery is independent of food intake; therefore there is no significant change in peak systolic or end-diastolic velocities in the celiac artery following a meal.


The superior and inferior mesenteric arteries supply the high-resistance vascular beds of the small intestine and colon. Pulsed Doppler evaluation reveals high-impedance flow with low diastolic velocities in the fasting state ( Fig. 26.10A ). This is because of the relative vasoconstriction of the mesenteric branch vessels before a meal, when the bowel is empty and quiescent. After a meal, however, there is an increase in mesenteric arterial blood flow in order to assist digestion. This is accomplished by vasodilatation of the mesenteric branches, which in turn allows for increased blood flow to the intestines (see Fig. 26.10B ). Moneta and colleagues showed that both peak systolic and end-diastolic velocities increase after a meal, with at least doubling of the end-diastolic velocity (EDV) in the SMA after eating. They found the greatest increases in blood flow velocity following meals that included fat, carbohydrate, and protein, and concluded that providing a patient with a mixed meal could be used as a provocative test to assess the reactivity of the mesenteric circulation. The presence of increased flow velocities after a meal infers the patency of splanchnic blood supply in their studies.


Although duplex ultrasound examinations of the mesenteric arteries may be performed before and after a meal to identify physiologic changes in blood flow, there appears to be significant variability in the response to food. Healy and associates found that postprandial duplex studies were not dependable and did not improve diagnostic accuracy in their series of patients. Because of the inconsistency and unreliability of the preprandial and postprandial examination results, this technique has fallen out of favor and is not commonly utilized in clinical practice.
- •
The celiac artery and the mesenteric arteries demonstrate distinct blood flow patterns.
- •
The celiac artery waveforms demonstrate a low-resistance pattern with high end-diastolic velocities as it supplies the low-resistance vascular beds of the liver and spleen.
- •
In the fasting state, the superior and inferior mesenteric artery waveforms demonstrate a high-impedance flow pattern with low diastolic velocities as they supply the high-resistance vascular beds of the small intestine and colon.
- •
After a meal, there is an increase in superior and inferior mesenteric arterial blood flow because of vasodilatation of the mesenteric branches in order to assist digestion.
Natural history of bowel ischemia
Mesenteric ischemia is an uncommon disorder that is associated with high mortality and morbidity, with mortality rates ranging between 30% and 90%. Factors that are related to the high mortality include insidious onset of symptoms, age at presentation greater than 60 years, and delay in diagnosis. Most cases of mesenteric ischemia are attributable to an acute event such as embolization or thrombosis of an artery or vein, which leads to decreased blood supply to the splanchnic vasculature. Mesenteric ischemia can be classified into acute and chronic forms, with chronic mesenteric ischemia accounting for less than 5% of all cases.
Acute mesenteric ischemia
Acute mesenteric ischemia is a life-threatening condition with reported mortality rates ranging from 59% to 93%. It is estimated that approximately 33% of cases of acute mesenteric ischemia are caused by arterial embolism, 33% are caused by arterial thrombosis, between 20% and 30% are caused by nonocclusive ischemia (hypotension), and the few remaining cases are caused by venous thrombosis.
Acute Arterial Occlusive Disease.
Acute arterial occlusive disease is caused by embolus, thrombosis, dissection, or external compression of a vessel. Doppler sonography is generally less useful in this situation because of the rapid time course of the disease and necessity for emergent intervention. Computed tomography (CT) angiography is the examination of choice. Duplex Doppler ultrasound may demonstrate acute thrombosis of a mesenteric artery as an echogenic focus within the lumen of a mildly dilated artery. High-resistance waveforms may be detected just proximal to the level of occlusion, and tardus-parvus waveforms may be seen distal to the site of occlusion ( Fig. 26.11 ). The treatment of choice depends on the size and location of the thrombus. Anticoagulation with thrombolytics and continuous papaverine infusion is considered acceptable in the case of a small thrombus that is located within a proximal vessel, prior to the branching of collateral arteries. A large thrombus that is located distal to branching collaterals is typically treated with embolectomy and/or resection, particularly in the setting of positive peritoneal findings ( Fig. 26.12 ).






Nonobstructive Mesenteric Arterial Insufficiency.
Nonobstructive mesenteric arterial insufficiency is usually attributable to hypotensive shock, blood loss, or sepsis that leads to insufficient blood supply to the bowel. Sonographically, one may find low peak systolic velocities (PSVs) in the aorta and the major aortic branches, without identifiable stenosis or thrombosis. Treatment of the underlying condition is key to the management of this type of bowel ischemia.
Mesenteric Venoocclusive Disease.
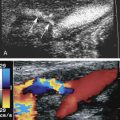
A third etiology of bowel ischemia is related to venoocclusive disease. Doppler sonography plays a lesser role in the investigation of this type of bowel ischemia because of its limited ability to detect slow venous blood flow or occlusive thrombus in the mesenteric vein and its branches . Absence of flow within the superior mesenteric vein (SMV) and splenic vein/portal vein confluence in association with the presence of mesenteric edema and ascites can imply this diagnosis. On gray-scale imaging, the occluded veins are usually distended, and may contain echogenic intraluminal material. Power and color Doppler imaging are utilized for the assessment of residual flow or visualization of venous collaterals ( Fig. 26.13 ). Similar to the other causes of acute mesenteric ischemia, ultrasound is not the first-line imaging modality, and clinically suspected cases usually are assessed with contrast-enhanced CT or magnetic resonance imaging (MRI), both of which have proven to be more valuable in the assessment of venous thrombosis.



Chronic mesenteric ischemia
The fourth type of mesenteric ischemia is chronic mesenteric ischemia (CMI), which in 95% of cases is attributable to long-standing atherosclerotic disease that results in stenosis or occlusion of two or more of the main mesenteric arteries. Nonatheromatous causes of CMI include vasculitides such as Takayasu arteritis, thromboangiitis obliterans, and radiation-induced vasculitis. CMI accounts for less than 5% of all cases of mesenteric ischemia and generally occurs in patients older than 60 years. Most studies have shown it to be more prevalent in females.
Atherosclerosis is a chronic progressive disease that can lead to near complete or complete occlusion of the mesenteric arteries. Progressive plaque formation primarily results from damage to the endothelial cells of the intima, with deposition of lipids within the vessel wall that lead to the development of cellular hypoxia. Increasing plaque burden at the level of the ostia of the mesenteric arteries may lead to significant stenosis, thus compromising the blood supply of the bowel and potentially leading to bowel ischemia. The rich collateral circulation between the celiac artery, the SMA, and the IMA can often compensate for stenosis within a single mesenteric vessel, and usually at least two of the three mesenteric vessels must be occluded or significantly narrowed for symptoms of CMI to develop. CMI is not commonly seen in clinical practice, but when it is encountered it represents a serious complex vascular disorder. Doppler sonography can play a major role in screening patients with suspected CMI.
Patients with suspected chronic mesenteric ischemia classically present with abdominal pain related to recent ingestion of a meal. Patients typically complain of postprandial pain, bloating, weight loss, and diarrhea. They may describe a “fear of food,” as they experience pain after meals. They may change their diet or alter their eating habits by consuming smaller more frequent meals in order to avoid discomfort. In some cases, this change of eating habits is not recognized by the patient, who may only note weight loss. Other patients have a more confusing clinical presentation, with vague symptoms of pain that may or may not be related to meals. CMI should always be considered in older adult patients with unexplained abdominal pain and weight loss, and therefore evaluation of the mesenteric arteries may prove helpful when working through the differential diagnosis in this group of patients. In the past, conventional angiography was the primary diagnostic modality used in the assessment of patients with suspected CMI. With continuous advances in technology, Doppler ultrasound, MR, and CT angiography have proven to be accurate noninvasive alternatives to conventional arteriography. Advantages of Doppler ultrasonography include direct evaluation of all three mesenteric vessels, assessment of the hemodynamics of blood flow, and determination of the significance of lesions involving the visceral vessels. It is an inexpensive, noninvasive method that requires no radiation or contrast material, can be performed portably, and does not have any of the inherent risks associated with other angiographic studies. Doppler ultrasound determines the hemodynamic significance of stenotic lesions by demonstrating alterations in blood flow including prestenotic and poststenotic waveform changes. These findings can influence the decision to intervene in the appropriate clinical setting.
- •
Acute mesenteric ischemia is a life-threatening condition that in about 33% of cases is caused by arterial embolism, 33% by arterial thrombosis, 20% to 30% by nonocclusive ischemia (hypotension), and the few remaining cases by venous thrombosis. CT angiography is the examination of choice.
- •
CMI is attributable to long-standing atherosclerotic disease that results in stenosis or occlusion of two or more of the main mesenteric arteries.
- •
CMI accounts for less than 5% of all cases of mesenteric ischemia and generally occurs in patients older than 60 years.
- •
Patients with suspected chronic mesenteric ischemia classically present with abdominal pain related to recent ingestion of a meal, and complain of postprandial pain, bloating, weight loss, and diarrhea.
- •
Doppler ultrasound determines the hemodynamic significance of stenotic lesions by demonstrating alterations in blood flow including prestenotic and poststenotic waveform changes.
Technique
Duplex and color Doppler examination of the mesenteric arteries includes evaluation of the proximal abdominal aorta and the origins and proximal segments of the celiac artery, SMA, and IMA. The distal segments of the mesenteric arteries are usually not well-imaged with ultrasound. Most atherosclerotic lesions occur at the ostia of these vessels, and these segments are the main focus of investigation.
Imaging in the fasting state is preferred in order to reduce the amount of scatter and attenuation from overlying bowel gas. Examination in the fasting state also avoids misinterpretation of the elevated PSVs seen in the postprandial state from stenotic flow. There is no need for premedication prior to the examination. Mesenteric Doppler studies should be performed using modern ultrasound equipment, which combines high-resolution gray-scale imaging with high-quality color and power Doppler imaging and sensitive pulsed Doppler waveform acquisition. Given that the mesenteric vessels are situated deep in the abdomen, a low-frequency 2 to 5 MHz convex (curvilinear) transducer is used. This allows for adequate visualization and resolution over a large field of view.
Examinations are usually performed with patients in the supine position by way of an anterior approach. Bowel gas can be displaced by the gradual advancement of the ultrasound transducer, resulting in compression of abdominal structures and displacement of the gas-containing bowel loops. If visualization of the vessels is limited by bowel gas despite a continuous compression technique, patients may be turned to the decubitus or oblique positions for better visualization of the vessels of interest. During the study, the patient is asked to breath-hold or breathe quietly in order to obtain adequate spectral Doppler samples.
The abdominal aorta is usually evaluated first via an anterior approach by placing the transducer just below the xiphoid process of the sternum. Transverse and sagittal planes are used for evaluation of aortic diameter and velocity. The abdominal aorta can be found just anterior to the spine, slightly to the left of midline on the transverse view, adjacent to the IVC. The abdominal aorta is included in the examination to assess for aortic aneurysm, dissection, and underlying atherosclerotic disease.
When examining the mesenteric arteries, several landmarks are helpful to ensure correct identification of the mesenteric vascular anatomy. The celiac artery, in the transverse view, has a distinctive appearance, with a characteristic T-shaped bifurcation (the “seagull sign” Fig. 26.14 ). On the transverse view, the SMA is surrounded by a prominent ring of peritoneal fat that separates it from the pancreas. In patients with normal bowel rotation, the SMA lies to the left of the SMV, posterior to the splenic vein and pancreas, and anterior to the left renal vein ( Fig. 26.15A ). On the longitudinal view, the SMA is found directly below the celiac artery and can be followed inferiorly along its long axis (see Fig. 26.15B ). Visualization of these structures improves detection of the mesenteric vessels and helps to avoid possible errors in diagnosis in patients with variant vascular anatomy. The IMA can be found on the transverse view arising from the left anterolateral aspect of the aorta, just below the renal arteries, and approximately 4 cm above the aortic bifurcation ( Fig. 26.16 ).




It has been noted in the literature that studies performed by experienced vascular technologists, sonographers, or sonologists provide the best results. Indeed, we have noted that our highest quality abdominal vascular studies are performed by sonographers and physicians with at least 1 year of experience with abdominal Doppler. The learning curve for these examinations depends on the technical ability, motivation, and patience of the examiner. It is clear that a steady volume of abdominal Doppler studies is required in order to gain proficiency and speed with these examinations. It is also clear that experience with these studies allows the examiner to determine, in short order, whether a successful study will be obtained in any given patient. It should be obvious in most cases whether excessive bowel gas, shortness of breath, large body size, or severe atherosclerotic disease will preclude a complete study. This is usually determined within the first 10 minutes of the examination.
The experienced examiner uses a number of techniques to improve visualization of the abdominal vessels and the detection of significant lesions. Optimization of gray-scale and color Doppler parameters assists in the visualization of vessel walls, the detection of atherosclerotic plaque, and assessment of the residual lumen. Harmonic imaging is routinely used to improve resolution and decrease noise in the image. The color Doppler gain, pulse repetition frequency, and wall filter are adjusted so that laminar flow has a homogeneous color-flow pattern in the normal segments of the aorta and branch vessels ( Fig. 26.17 ). These adjustments should be tailored to each patient, as mesenteric arterial blood flow velocities vary widely. Proper adjustment of the color Doppler parameters aids in the assessment of vessel patency and the visualization of normal blood flow in a vessel. These adjustments permit the examiner to screen a vessel quickly for potential flow abnormalities and to detect color aliasing and color bruit artifacts that occur in areas with significant flow disturbance ( Fig. 26.18 ). The ability to demonstrate these color flow changes increases the sensitivity for stenosis detection and decreases the time of examination.


Pulsed Doppler waveforms are obtained with a small sample volume (1.5 to 3 mm) to ensure that velocity information is derived from the vessel of interest and not from adjacent structures. Angle correction is always used for accurate Doppler analysis. Most studies are performed with a Doppler angle of insonation of 60 degrees or less in order to provide true velocity estimation. Higher Doppler angles of greater than 60 degrees may lead to overestimation of the measured velocity in a given region of interest. Rizzo and coworkers showed that varying the angle of insonation from 0 to 80 degrees produced marked increases in SMA PSV in normal volunteers. They found that for the SMA, 70-degree and 80-degree angles produced 16% and 120% increases, respectively, in PSVs. Although many examiners prefer to obtain Doppler velocity measurements at 60 degrees (similar to carotid artery studies), a 60-degree angle of insonation may be difficult to obtain in the celiac artery because the artery projects toward the probe at a 0- to 30-degree angle. In these cases, a lower Doppler angle is used for celiac artery velocity measurement. This should be noted in the report or patient’s chart, so that the same angle can be used on follow-up evaluations in order to ensure that the error in the estimation of PSVs is consistent among all studies. Most spectral samples are obtained from a sagittal (longitudinal) projection of the aorta and mesenteric vessels . The direction of blood flow in the celiac and SMA is also more easily determined in the sagittal plane. An oblique plane is used for Doppler evaluation of the IMA along the direction of blood flow.
Examination Protocol
Our protocol includes an initial gray-scale evaluation of the abdominal aorta in order to assess for the presence or absence of atherosclerotic plaque, luminal narrowing, dissection, and aneurysm ( Figs. 26.19 and 26.20 ). The presence of significant atherosclerotic disease in the abdominal aorta should raise suspicion for potential mesenteric arterial disease. Once the initial gray-scale assessment is completed, color Doppler is utilized to search for potential abnormalities within the vessel lumen, which may manifest as color flow abnormalities ( Fig. 26.21 ). Pulsed Doppler samples are also obtained from the abdominal aorta at the level of the mesenteric arteries. These measurements provide baseline velocities for comparison with the mesenteric artery PSVs and may be used to calculate the mesenteric artery to aortic PSV ratios. PSV measurements are also obtained from the origin and visualized segments of the celiac artery, SMA, and IMA. The sample volume is passed through the abdominal aorta into the ostium and visualized segments of each artery, in search of the highest PSV and signs of poststenotic turbulence and bruit. It is important to keep in mind that the highest velocity measurements will be obtained in an area of stenosis.
- •
Duplex examination of the mesenteric arteries includes evaluation of the proximal abdominal aorta and the origins and proximal segments of the celiac artery, SMA, and IMA.
- •
Imaging in the fasting state reduces the amount of scatter and attenuation from overlying bowel gas. It also avoids mistaking an elevated PSV as a stenosis rather than a normal postprandial response.
- •
Gray-scale evaluation of the abdominal aorta and branch vessels is performed to assess for the presence of atherosclerotic plaque, luminal narrowing, dissection, and aneurysm.
- •
Optimization of the color Doppler parameters (color Doppler gain, pulse repetition frequency, and wall filter) is required to ensure that normal vessel segments demonstrate a homogeneous color flow pattern and to detect color aliasing and color bruit artifacts in areas with significant stenosis.
- •
Most spectral samples are obtained from a longitudinal projection of the aorta and mesenteric vessels with angle correction of 60 degrees or less.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree