Introduction
Duplex and color Doppler imaging play an important role in the ultrasound evaluation of the female pelvis. Doppler is indispensable for the workup of many common problems. These techniques can be applied to both transabdominal and endovaginal examinations. The combination of Doppler ultrasound with endovaginal scanning is particularly valuable for gynecologic investigations because there is improved resolution and increased sensitivity to blood flow. The term endovaginal color flow Doppler imaging (EVCF) is used to describe this pairing of techniques. We routinely utilize Doppler ultrasound in a number of different applications:
- 1.
Identification of the dominant follicle or corpus luteal cyst in patients with pelvic pain or suspected ectopic pregnancy
- 2.
Evaluation of vaginal bleeding in nongravid and pregnant patients
- 3.
Detection of placental tissue in abnormal intrauterine pregnancy, ectopic pregnancy, and retained products of conception
- 4.
Diagnosis of ovarian torsion
- 5.
Characterization of ovarian and adnexal masses
- 6.
Detection of a number of uterine abnormalities, including fibroids, polyps, and tumors, and vascular abnormalities such as arteriovenous malformation, enhanced myometrial vascularity, and the pelvic congestion syndrome
Technical Issues
Because the technical aspects of color flow imaging are covered in Chapter 3 , this chapter emphasizes the key points pertinent to female pelvic sonography. Similar to other color and pulsed Doppler examinations, Doppler evaluation of the uterus and ovaries should be considered a dynamic process, requiring adjustment of the color flow parameters according to the type of examination. Using the manufacturer’s settings (presets) is a good starting point for any examination. They serve as a general guide and can be adjusted to improve visualization of blood flow. Presets are helpful to novice or beginner sonographers and sonologists, particularly when the initial demonstration of color flow is suboptimal but critical for diagnosis.
It is important to remember that color, power, and pulsed Doppler images are based on similar physical principles but display different information. Color Doppler images are based on the mean velocity display of reflected frequency shifts. In other words, the frequencies reflected from the moving red blood cells are averaged over time and presented on the image. Color Doppler displays a range of velocities on the image but does not provide absolute or peak systolic velocity (PSV) information. Pulsed Doppler is required to determine the peak velocity at a particular location.
Power (amplitude) Doppler images are determined by the strength or amplitude of the returning Doppler shifts. The frequency shifts are amplified, assigned a color, and displayed along with the gray-scale information. Power Doppler provides three to five times increased sensitivity to blood flow compared with color flow imaging. There is less dependence on the angle of insonation, so that flow signals can be detected at angles close to 90 degrees. Thus very weak Doppler shifts are presented on the power Doppler image. Advantages of power Doppler include improved vascular detail, faster localization of blood flow for pulsed Doppler sampling, and assessment of global tissue perfusion.
Power Doppler images do not demonstrate color aliasing or direction of flow. This is not a significant limitation, as determination of flow direction or velocity is achieved with color flow or pulsed Doppler sampling. Power Doppler is also susceptible to similar pitfalls related to incorrect color flow settings that occur with color Doppler imaging. In general, there are three Doppler parameters that should always be checked to ensure optimal color and power Doppler imaging: (1) color velocity range or pulse repetition frequency (PRF), (2) color gain, and (3) wall filter.
These settings should be adjusted to improve the sensitivity of color flow for each study, as they are fundamental to the detection of low-flow states. Decreasing the PRF (color velocity scale), increasing the color gain, and/or decreasing the wall filter settings will facilitate the detection of low-velocity Doppler signals. When flow velocities are high, and we want to reduce the degree of color noise or aliasing in the image, we increase the PRF or color velocity scale, decrease the color gain, and/or increase the wall filter. Experience and practice with different parameter settings will increase understanding of the interrelationships between these settings and lead to improved detection of blood flow. As always, the focal zone should be placed near the region of interest.
Most studies are performed with transducers in the 2- to 5-MHz range for transabdominal studies. Endovaginal scans utilize frequencies in the 5- to 10-MHz range. For pulsed Doppler evaluation, angle correction is performed when the direction of blood flow can be ascertained. We use no angle correction (0 degrees) for tiny or tortuous vessels in the pelvis when the direction of flow cannot be determined. A small sample volume is used to interrogate small vessels for Doppler spectral analysis and to avoid obtaining signals from multiple sources.
- •
Doppler is indispensable for the workup of many obstetric and gynecologic conditions, including pelvic pain, vaginal bleeding, and pelvic mass.
- •
Doppler allows for a specific diagnosis, frequently eliminating the need for computed tomographic or magnetic resonance imaging correlation.
- •
Color, power, and pulsed Doppler display different information, but rely on similar physical principles.
- •
Color Doppler images are based on the mean velocity display, whereas power Doppler images are determined by the amplitude of the returning Doppler shifts.
- •
Doppler parameters that should always be checked to ensure optimal color and power Doppler imaging are color velocity range or PRF, color gain, and wall filter.
Normal Anatomy and Hemodynamics
A thorough gray-scale sonographic evaluation of the uterus and ovaries is usually performed in conjunction with Doppler assessment for blood flow. We usually begin our evaluation with the uterus and then turn our attention to the adnexa and evaluation of the ovaries. The uterus is a pear-shaped, midline structure that is usually easy to identify ( Fig. 29.1 ). Measurements of the length, width, and anteroposterior diameter of the uterus should be obtained. We also evaluate the thickness of the endometrium and cervix and the presence and location of any uterine masses. The ovaries are variable in shape and location. Although the ovaries can be identified with either transabdominal or endovaginal scanning, a combination of both techniques may be necessary for complete evaluation. The ovaries are also measured in three dimensions. The presence of cysts or masses is noted and correlated with the menstrual cycle.
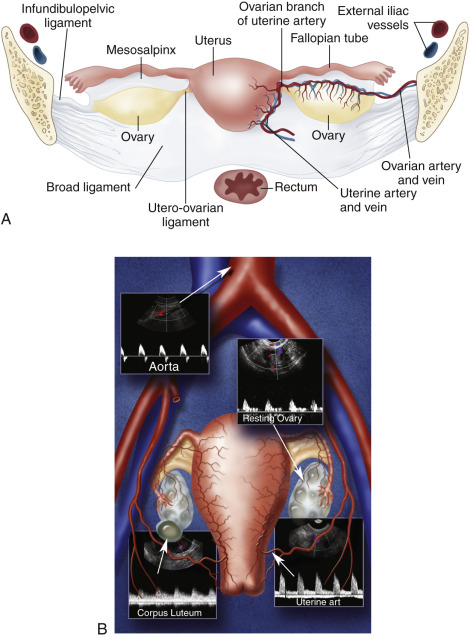
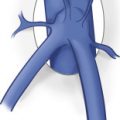
Color and pulsed Doppler examination requires knowledge of the vascular anatomy and hemodynamic changes of the female pelvis. The vessels most frequently examined in the pelvis include the iliac, uterine, and ovarian arteries and veins (see Fig. 29.1 ). These vessels may be identified with both transabdominal and endovaginal imaging. EVCF affords improved resolution and vascular detail compared with the transabdominal approach.
The uterine artery is a branch of the internal iliac artery and penetrates the uterus at the lower uterine segment (see Fig. 29.1B ). Uterine artery branches course toward the uterine fundus and cervix and toward the ovaries in the broad ligament. Color and pulsed Doppler sampling of the uterine artery reveals high-impedance, low diastolic flow in the nongravid state. A characteristic diastolic “peak” is usually noted ( Fig. 29.2 ). Identification of this peak is helpful in characterizing waveforms obtained during pelvic examination as originating from the uterine artery. There is a gradual decrease in resistance to flow in the spectral samples obtained from the uterine arteries during the second trimester of pregnancy. The decrease in resistance and resistivity index is related to increased blood flow, particularly in diastole, necessary for normal placental and fetal growth. This is recognized as continuous low-resistance blood flow on the waveforms obtained from the placenta and umbilical arteries supplying the fetus.

Each ovary receives a dual blood supply, as shown in Fig. 29.1A . The ovarian arteries originate from the abdominal aorta and descend to the pelvis to supply each ovary. The ovaries also receive branches from the uterine arteries that course along the broad ligament. Blood flow patterns observed during color and pulsed Doppler sampling of the ovary vary depending on the phase of the ovulatory cycle. Low-velocity, high-impedance (low diastolic flow) waveforms are usually noted early in menses into the follicular phase. ( Fig. 29.3 ). This is seen during the first menstrual week when the ovaries are dormant and before the formation of the dominant follicle and corpus luteal cyst.

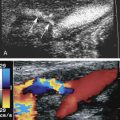
The luteal phase coincides with the extrusion of the mature egg or oocyte and formation of the corpus luteal cyst. Thickening of the cyst walls is seen with gray-scale imaging. Color Doppler demonstrates a ring of vascularity around the luteal cyst, related to formation of tiny vessels in the walls of the cyst. Pulsed Doppler shows an increase in peak systolic and end-diastolic velocities ( Fig. 29.4 ). The increased velocities are related to neovascularization of the corpus luteum, required for oocyte maturation and hormonal activity.



I originally described the “ring of fire” color flow pattern to represent the increased vascularity noted around the periphery of an ectopic pregnancy. Subsequently, we became aware that a similar pattern of peripheral vascularity occurs with the formation of a corpus luteal cyst. In fact, we look for the ring of increased vascularity in the ovary to locate and characterize luteal cysts. It should be clear that the ring of fire color flow pattern alone cannot distinguish between ectopic pregnancies and luteal cysts. Investigators have tried to identify discriminating Doppler parameters to distinguish luteal cysts from ectopic flow. This is difficult due to an overlap in peak systolic velocity and resistivity index measurements between luteal cysts and ectopic pregnancies. The origin of the Doppler signal, from within the ovary or from an adnexal mass, allows more accurate characterization of a corpus luteal cyst or an ectopic pregnancy than velocity measurement or resistivity index alone. Therefore we do not use Doppler to distinguish luteal from ectopic flow but to localize the site of origin of the signals to determine their significance.
Color and pulsed Doppler signals obtained from a postmenopausal ovary have low peak systolic velocities similar to ovaries in the follicular phase ( Fig. 29.5 ). This is typical of ovaries in the resting state. Because postmenopausal ovarian vessels demonstrate low-velocity flow, this vascularity may be very difficult to visualize with conventional color Doppler flow settings. Low color velocity scale and color wall filter adjustments may be necessary to detect postmenopausal ovarian blood flow. Power Doppler imaging improves the visualization of ovarian flow, particularly in postmenopausal women. Because postmenopausal ovaries no longer ovulate, they remain relatively quiescent and are associated with little or no diastolic flow.
- •
A combination of transabdominal and endovaginal scanning may be necessary for complete evaluation of the female pelvis.
- •
Doppler sampling of the uterine artery reveals high-impedance, low diastolic flow in the nongravid state with a characteristic diastolic “peak.”
- •
Blood flow patterns observed during Doppler sampling of the ovaries depend on the phase of the ovulatory cycle.
- •
During the early follicular phase or in the postmenopausal state, low-velocity ovarian flow is noted.
- •
During the luteal phase, there is a marked increase in peak systolic and end-diastolic velocities with a “ring of fire” pattern around the ovarian cyst.

Current Applications
Current applications of transabdominal scanning and EVCF include identification of the corpus luteal cyst, detection of intrauterine placental flow, diagnosis of ectopic pregnancy and retained products of conception, evaluation for ovarian torsion, and characterization of adnexal masses and uterine abnormalities.
Corpus luteal cyst
Identification of the dominant follicle or corpus luteal cyst has proven extremely helpful in patients who present with pelvic pain, adnexal mass, or ectopic pregnancy. Ovarian cysts are the most common cause of acute pelvic pain in premenopausal patients. The pain is usually associated with enlargement of the cyst during the midportion of the menstrual cycle and precedes cyst rupture and the release of fluid.
Simple ovarian cysts are easily characterized by their lack of internal echoes, smooth and thin walls; and posterior sound enhancement. Complex hemorrhagic ovarian cysts can be much harder to characterize as benign ovarian cysts. Hemorrhagic cysts may be filled with low-level echoes and can become hypoechoic or isoechoic to the ovarian parenchyma. These cysts may also contain solid areas and septations and may resemble ovarian neoplasms.
Color and pulsed Doppler are important tools for the characterization of complex ovarian cysts. Color Doppler can demonstrate the ring of increased vascularity in the wall of the corpus luteal cyst ( Fig. 29.6 ). As mentioned earlier, the peripheral vascularity is related to neovascularization associated with the formation of the corpus luteal cyst. The increased blood flow identified with color Doppler is associated with elevated peak systolic velocities and low-resistance flow. Dillon and colleagues demonstrated a peak systolic velocity of 27 ±10 cm/s and a resistivity index of 0.44 ±0.09 for corpus luteal cysts. The demonstration of peripheral vascularity with color or power Doppler increases the conspicuity of the corpus luteal cyst, even when it is filled with blood and is isoechoic to ovarian tissue on gray-scale images. The lack of vascularity within the central part of the lesion suggests it is a hemorrhagic cyst. This is particularly helpful when gray-scale evaluation demonstrates wall thickening, nodules, or septations within the cyst cavity. The absence of flow within the cyst cavity suggests that any solid material within the cyst is likely related to hematoma or a retracting clot and not a tumor ( Fig. 29.7 ). A follow-up ultrasound study 6 to 8 weeks later, during the first week of a subsequent menstrual cycle, can be performed to assess for complete resolution of the complex cyst and to exclude the possibility of a tumor.



The recognition of the corpus luteal cyst also aids in the diagnosis of ectopic pregnancy. Approximately 85% to 90% of ectopic pregnancies occur on the same side as the corpus luteal cyst. Identification of the corpus luteum determines the side of ovulation and directs the examiner to the expected site of the ectopic pregnancy ( Fig. 29.8 ). Color and pulsed Doppler also play a role in the identification and follow-up of ectopic pregnancy, which is our next topic of discussion.




Ectopic Pregnancy
Ectopic pregnancy occurs in approximately 2% of pregnancies and is the leading cause of pregnancy-related deaths during the first trimester. There is a rising incidence of ectopic pregnancy that is related to: (1) an increased number of patients at risk; (2) new techniques that allow earlier diagnosis; (3) improved treatment for salpingitis and ectopic pregnancy; and (4) increased utilization of ovulation induction and assisted reproduction techniques.
The vast majority of ectopic pregnancies occur in the fallopian tube, usually in the ampullary region. Less commonly, ectopic pregnancies will be seen in the interstitial portion of the fallopian tube, in the cervical canal, or within a cesarean defect. Abdominal and intraovarian pregnancies are rare.
It is important to understand and solicit risk factors from patients with suspected ectopic pregnancy. Any process that produces scarring or obstruction of the fallopian tube predisposes an individual to ectopic pregnancy. The obstruction may be related to prior pelvic surgery or tubal ligation, prior ectopic pregnancy, or a history of pelvic inflammatory disease or salpingitis. In vitro fertilization also increases the rate of ectopic pregnancy because of multiple risk factors, including infertility, ovulation induction, and embryo transfer with retrograde migration of the embryo into the fallopian tube. Infertility is associated with multiple anatomic and physiologic conditions that increase the risk of ectopic pregnancy. Other risk factors include prior cesarean section, in utero exposure to diethylstilbestrol (DES), and sterilization.
The clinical presentation of ectopic pregnancy is variable; however, a positive pregnancy test, pelvic pain, an adnexal mass, and/or vaginal bleeding raise clinical suspicion for this condition. The classic triad of pelvic pain, adnexal mass, and vaginal bleeding occurs only in approximately 45% of patients. Patients may be asymptomatic or have focal or generalized pelvic or abdominal pain.
Earlier diagnosis of ectopic pregnancy is possible due to improved sonographic techniques and increased awareness of the disease. Early detection reduces the risk for tubal rupture and significant hemorrhage. The evaluation for ectopic pregnancy usually includes a combination of endovaginal sonography and serum human chorionic gonadotropin (hCG) titers. Diagnostic laparoscopy is considered the gold standard and is usually reserved for difficult cases.
The sonographic findings associated with ectopic pregnancy include absence of a normal intrauterine pregnancy, a pseudogestational sac (described later), a live extrauterine embryo, pelvic fluid, and an adnexal mass. A classic sonographic feature is the extrauterine gestational sac or “tubal ring.” This appears as a ring-shaped adnexal mass with a thick wall, similar in appearance to a doughnut or life preserver. Commonly, a complex or solid adnexal mass is identified, related to pelvic hematoma or hematosalpinx. The complex appearance of the mass is usually related to bleeding or rupture of the ectopic pregnancy. A live embryo is seen less commonly but provides the highest positive predictive value for the diagnosis of ectopic pregnancy.
Placental flow
When a normal intrauterine pregnancy is not identified, color and pulsed Doppler imaging can be used to demonstrate features of placental flow in the uterus or adnexa. The presence or absence of placental flow in the uterus is an extremely valuable finding in the evaluation for suspected ectopic pregnancy, for when it is present in the uterus, the focus of examination shifts toward an abnormal intrauterine pregnancy rather than an extrauterine gestation. Placental flow is related to invasion of the endometrium by growing trophoblastic (placental) tissue. As the trophoblast grows into the uterine tissue, the maternal spiral arteries will shunt arterial blood into the intervillous sinusoids. This results in relatively high-velocity, low-resistance blood flow that is readily detected with color Doppler imaging ( Fig. 29.9 ). Pulsed Doppler examination of intrauterine placental flow reveals a peak systolic velocity greater than 21 cm/s, and the impedance decreases to a mean resistivity index of 0.44 ± 0.09. The increased vascularity related to trophoblastic implantation is detected 36 to 50 days after the last menstrual period.


Detection of the flow characteristics related to placentation is invaluable in the identification of gestational tissue in the uterus or in the adnexa. A peak systolic velocity cutoff at 21 cm/s or more is utilized to characterize placental flow in the uterus. We utilize no (0-degree) angle correction for the pulsed Doppler examination, as the vessels are too small to determine the direction of flow. Despite the 0-degree angle correction, Doppler is remarkably accurate for the detection of placental flow when the peak systolic velocity is greater than 21 cm/s. The detection of placental flow in the uterus confirms the presence of a normal or abnormal intrauterine pregnancy. Pulsed Doppler interrogation of a normal embryo is not performed due to potential bioeffects related to cavitation and heating.
Pseudogestation
Color and pulsed Doppler can also distinguish between an abnormal intrauterine pregnancy and a pseudogestational sac related to ectopic pregnancy. The sonographic appearance of “pseudosacs” ranges from endometrial thickening to a fluid collection in the endometrial canal. Unlike normal intrauterine gestational sacs, pseudosacs tend to be oval and located centrally in the endometrial cavity rather than eccentrically placed in the endometrium. They do not demonstrate a double decidual reaction, yolk sac, or embryo. They also do not exhibit placental flow, an important discriminatory factor ( Fig. 29.10 ). Doppler sampling of the area surrounding the pseudosac will demonstrate velocities less than 21 cm/s. Dillon and colleagues showed that Doppler findings were 100% specific in the identification of pseudogestational sacs.

Value of Doppler for Evaluation of Ectopic Pregnancy
The diagnosis of ectopic pregnancy is based on the finding of a cystic, complex, or solid mass, separate from the uterus and ovaries, in a pregnant patient. In a study of 155 patients with suspected ectopic pregnancy, my colleagues and I found that placental flow was observed in 85% (55 of 65) of patients with ectopic pregnancy. Color and pulsed Doppler showed a sensitivity of 95% and a specificity of 98% for the detection of ectopic pregnancy. We originally described the appearance of increased vascularity around the extrauterine gestational sac as a “ring of fire” pattern on color Doppler imaging. This is associated with a low-resistance flow pattern on pulsed Doppler interrogation ( Fig. 29.11 ). The velocity cutoff of 21 cm/s that is used to identify intrauterine placental flow is frequently not achieved with pulsed Doppler evaluation of ectopic pregnancy. Thus it is not a discriminating feature for the identification of placental flow in ectopic pregnancy.