Definition and Etiology
Emphysema is defined as a “condition of the lung characterized by abnormal, permanent enlargement of the airspaces distal to the terminal bronchiole, accompanied by destruction of their walls.” Because emphysema decreases the elastic recoil force that drives air out of the lung and thereby reduces maximal expiratory airflow, the disease is clinically classified as one of the chronic obstructive pulmonary diseases (COPDs). In morphologic appearance, two main subtypes of emphysema exist. The centrilobular (or centriacinar) form of emphysema results from dilatation or destruction of the respiratory bronchioles and is the type of emphysema most closely associated with cigarette smoking. The panlobular, or panacinar, form of emphysema is associated with α1-antitrypsin deficiency and results in an even dilatation and destruction of the entire acinus. It has been suggested that one or the other of these two subtypes predominates in severe disease and that the centrilobular subtype is associated with more severe small airways obstruction. There is a relation between the severity of emphysema and the pack-years of cigarette smoking, but this relation is weak. Indeed, only 40% of heavy smokers develop substantial lung destruction resulting from emphysema. On the other hand, emphysema can occasionally be found in individuals with normal lung function who have never smoked.
Prevalence and Epidemiology
The overall prevalence and epidemiology of emphysema are almost impossible to determine for three major reasons. First, the prevalence of emphysema strongly depends on regional factors, such as smoking habits, social standards, and environmental air pollution. Given that these factors largely vary, the prevalence of emphysema will show equally varying features, even in relatively small geographic areas. Second, emphysema becomes clinically evident in advanced disease, whereas mild or moderate disease can remain clinically silent. There are no screening programs dedicated to emphysema, although lung cancer screening with low-dose computed tomography (CT) may incidentally detect it, and a substantial number of individuals with emphysema will remain undiagnosed during their lifetime if no comorbidity exists that can bring to light emphysema as an incidental finding. Third, emphysema is clinically classified as a chronic obstructive lung disease. In this group of diseases the clinical findings may overlap with airways disorders. Furthermore, epidemiologic data exist for COPD as a group of diseases but not for the individual diseases such as emphysema. According to the Centers for Disease Control and Prevention, as of 2015 there are 36.5 million people who smoke cigarettes in the United States (1.1 billion smoke worldwide). Smoking is the leading cause of preventable death in the United States, accounting for more than 480,000 deaths per year.
Clinical Presentation
In many cases the clinical manifestations of emphysema are entirely nonspecific. In early stages, patients are often asymptomatic, and emphysema may be detected as an incidental finding on a CT examination performed for other purposes. In more advanced cases symptoms may overlap with symptoms caused by coexisting airway abnormalities and can therefore be difficult to attribute to the existence of emphysema. Patients with moderate to advanced disease, however, often complain of cough, either dry or productive, with increased frequency in the morning hours. Depending on the severity of the disease, breathlessness can occur either under exertion or at rest. Patients with severe emphysema can be susceptible to pulmonary infections that can occur at increased frequency or heal with increased delay.
Pathophysiology
Pathology
The definition of emphysema clearly refers to the acinus as a basic lung structure. The acinus is defined as the lung parenchyma that subtends from the terminal membranous bronchiole and consists of three generations of respiratory bronchioles, alveolar ducts, saccules, and alveoli. As opposed to the secondary pulmonary lobule, the acinus is not grossly identifiable. The terms centrilobular and panlobular are derived from their gross distributions within the secondary pulmonary lobule as defined by Miller. Because of the central location of the terminal bronchioles, the terms centriacinar, centrilobular, panacinar, and panlobular are roughly equivalent, and both terms are commonly used interchangeably.
The use of animal models and, particularly, genetically modified animals has produced extensive information about the pathogenesis of emphysema. The concept of a protease-antiprotease imbalance has been expanded but continues to include the inflammatory cascade, with involvement of the interleukins with Th1 cytokines and both serine proteases and metalloproteases. Neutrophils and macrophages have been joined by CD4-positive and CD8-positive T lymphocytes as important effector cells. The presence of apoptosis in emphysematous lungs has introduced a concept of disordered lung maintenance and repair, and there has been a suggestion of an immune basis for lung destruction.
Pathologic Subtypes of Emphysema
Centrilobular Emphysema
Centrilobular emphysema is characteristically found in cigarette smokers. Causes of centrilobular emphysema or bullae besides cigarette smoking include human immunodeficiency virus (HIV), Salla disease, Marfan syndrome, and Menke syndrome. On gross specimen, centrilobular emphysema is usually more common and more severe in the upper lung zones. In the upper lobe the posterior and apical segments are commonly affected; in the lower lobe the superior segment is more involved. Assessment of the secondary pulmonary lobule will demonstrate the central position of destruction, with sharply demarcated emphysematous areas separated from the acinar periphery by intact alveolar ducts and sacs of normal size ( Fig. 60.1 ). In more severe lesions the destruction will advance toward the periphery of the lobule, which can make the differentiation between centrilobular and panlobular emphysema difficult.
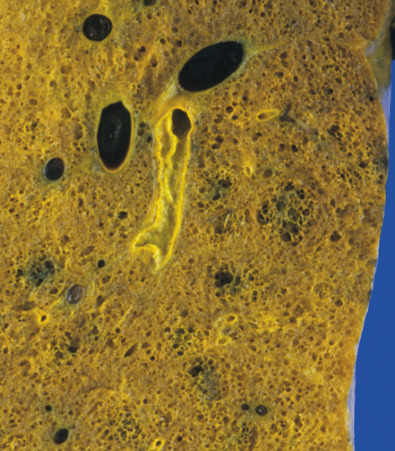
On microscopy airspace enlargement can be associated with a distorted respiratory bronchiole to form the classic centrilobular emphysema lesion. With increasing severity, isolated strands of alveoli can be seen. They are a useful indicator of the presence of emphysema. Collections of macrophages within the airspaces or adjacent to the bronchiole are common (representing respiratory bronchiolitis; see Chapter 34 ), and pigment can be seen both within the macrophages and in the bronchiolar fibrous tissue.
Panlobular Emphysema
On gross specimen, panlobular emphysema can be difficult to detect. In normal lungs the smaller alveoli can be clearly distinguished from the alveolar ducts and respiratory bronchioles; in panlobular emphysema, this distinction becomes lost because alveoli lose their sharp angles, enlarge, and eventually lose their contrast in size and in shape with the ducts. The lung architecture thus can appear simplified, with formation of small box-like structures. In more severe disease the abnormal enlargement becomes more obvious, even though the destruction is relatively uniform within the individual lobules ( Fig. 60.2 ). On microscopic examination the uniformity of the enlargement throughout the lobules persists (see Fig. 60.2 ). Airspaces adjacent to the venous septa are similar in size to those adjacent to the airways. Subtle signs of inflammation can be present.
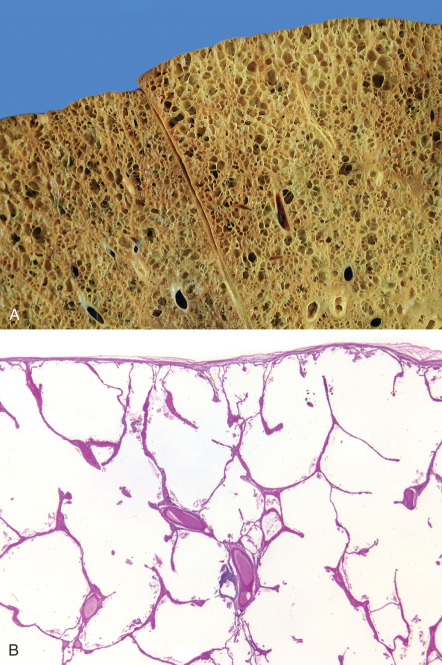
Panlobular emphysema is highly associated with α1-antitrypsin deficiency. Less likely causes of this pattern include hypocomplementemic urticarial vasculitis syndrome, intravenous methylphenidate abuse (so-called Ritalin lung), and some elastin abnormalities, such as cutis laxa and Ehlers-Danlos.
Other “Subtypes” of Emphysema
Paraseptal Emphysema.
This emphysematous destruction pattern is located in the periphery of the lung adjacent to the pleura or along interlobular septa. It is thus mainly subpleural in location and bound by the interlobular septa. It may occasionally occur as an isolated finding. However, it is usually seen in association with either severe centrilobular or panlobular emphysema. Paraseptal emphysema can be one of the many causes of spontaneous pneumothorax. Although the exact pathogenesis is unclear, the relationship between paraseptal emphysema and thin and tall body habitus has led to the suggestion that this subtype of emphysema is due to the effects of gravitational pull on the lungs, with a greater negative pleural pressure at the lung apices.
There is some evidence that smoking of marijuana cigarettes may be more highly associated with paraseptal emphysema than regular cigarettes. Some malnutrition syndromes can also cause paraseptal emphysema related to underlying elastase injury. Vanishing lung syndrome ( Fig. 60.3 ), also referred to as giant bullous emphysema, is a rare syndrome characterized by severe paraseptal emphysema and large bullae formation, with the bullae occupying at least one-third of a hemithorax and compressing the adjacent parenchyma. Disease can be unilateral but is more frequently bilateral, and spontaneous pneumothorax is frequent. The disease classically affects young male smokers, but there are few case reports with a possible hereditary component and some possible additional associations with marijuana use and HIV. Bullectomy can result in significant improvements in pulmonary function, but further decline 3 to 4 years after surgery is typical.
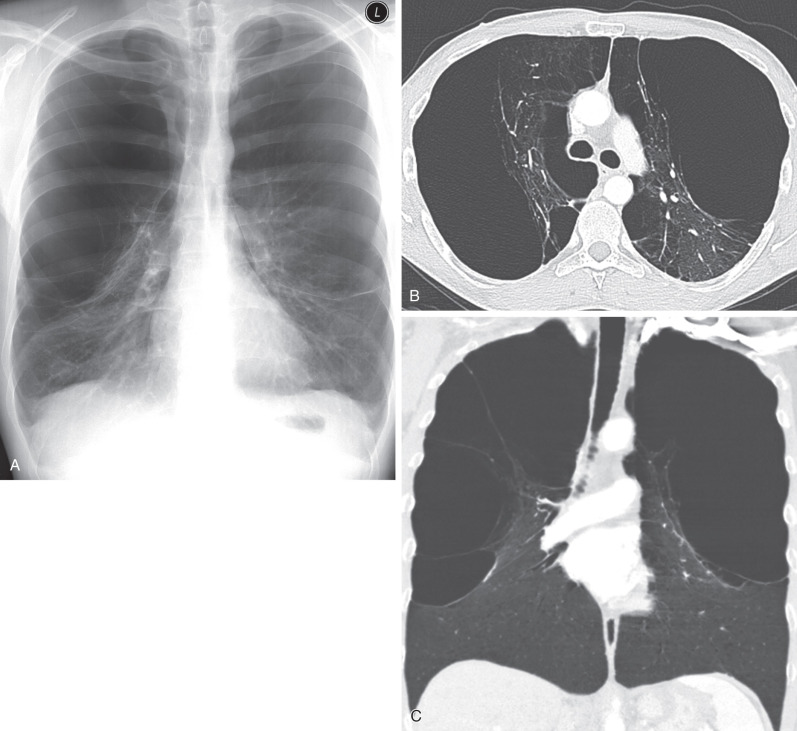
Paracicatricial Emphysema.
Lung destruction, and therefore emphysema, is commonly found adjacent to areas of scarring, which explains the term attributed to this alteration. Because the destruction has no particular position within the lobule, it was also termed irregular emphysema. Per definition, it is limited in extent and of little clinical relevance, with patient symptomatology generally attributed to the primary pulmonary diagnosis causing the emphysema, such as pulmonary fibrosis or sarcoidosis.
Physiology
The destruction of pulmonary parenchyma by emphysema creates a decreased mass of functioning lung tissue and thereby decreases the amount of gas exchange that can take place. As lung tissue is destroyed, it loses its elastic recoil and its volume expands. When destruction and expansion occur in a nonuniform manner, the most affected lung tissue can crowd the relatively spared lung tissue and prevent adequate ventilation of the latter. Eventually, obstruction of the small airways can occur, with obstruction being caused by a combination of reversible bronchospasm and irreversible loss of elastic recoil by adjacent lung parenchyma. The suitability of a patient for a given treatment will largely depend on the relative contributions of lung destruction, lung recoil, and small airways obstruction to the overall physiologic and clinical impairment of the patient.
In patients with emphysema, the forced expiratory volume in 1 second (FEV1), the forced vital capacity (FVC), the forced expiratory volume as a percentage of vital capacity (FEV/FVC%), the forced expiratory flow (FEF25%–75%), and the maximum expiratory flow at 50% and 75%, respectively, of exhaled vital capacity (Vmax50% and Vmax75%, respectively) will all be reduced. All of these parameters reflect functional obstruction, whether this is caused by alteration of the airway itself or by loss of radial traction resulting from emphysema. The FVC is reduced because the airways close prematurely at an abnormally high lung volume, which is at the source of an increased residual volume.
On the other hand, the total lung capacity, the functional residual capacity, and the residual volume are typically increased. In severe disease the expiratory flow-volume curve is grossly abnormal. Flow is strikingly reduced as the airways collapse, and flow limitation by dynamic compression occurs. A scooped-out appearance of the curve is often seen. Flow is greatly reduced in relation to lung volume and ceases at high lung volume because of premature airway closure. Simultaneously, the inspiratory flow-volume curve may be nearly normal. As elastic recoil of the lung is reduced in emphysema, the pressure-volume curve is displaced up and to the left. This probably reflects the disorganization and perhaps loss of elastic tissue as a result of destruction of alveolar walls.
Manifestations of the Disease
Radiography
The only direct sign of emphysema on radiographs is the presence of bullae (see Fig. 60.3 ). However, because of the limited contrast resolution of the chest radiograph, these focal areas of increased lucency can be difficult to detect. Indirect signs of lung destruction caused by emphysema include the focal absence of pulmonary vessels and the reduction of vessel caliber with tapering toward the lung periphery. Abnormalities of the vascular pattern are indeed highly suggestive of emphysema, but their sensitivity is low. These findings have a sensitivity of only 40% in detecting emphysema. Findings related to hyperinflation of the lungs include flattening of the diaphragm and an increased retrosternal space on the lateral view ( Figs. 60.4 and 60.5 ). These findings are more common than abnormalities of the vascular pattern, but their specificity is also low. The combined signs of hyperinflation and vascular alterations have been shown to allow the diagnosis of emphysema in 29 of 30 autopsy-proven, symptomatic patients but in only 8 of 17 autopsy-proven, asymptomatic patients.
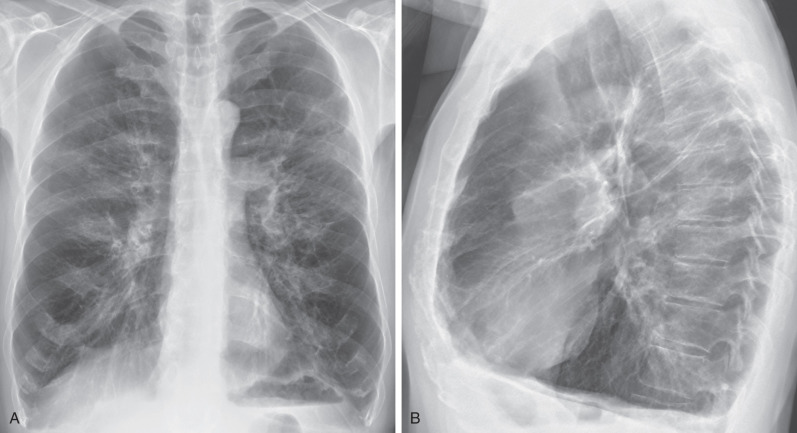
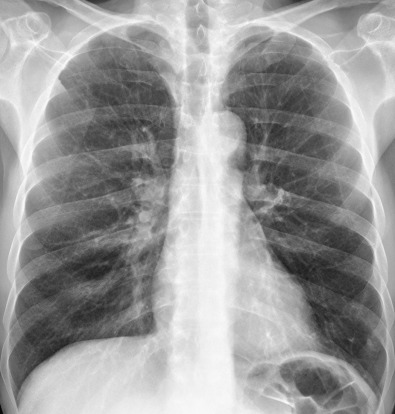
Taking the above into consideration, limitations of radiography in the assessment of emphysema include its low specificity, its low sensitivity in the evaluation of mild disease, its considerable interobserver variability in the interpretation of findings, and its inability to quantify the severity of emphysema.
Computed Tomography
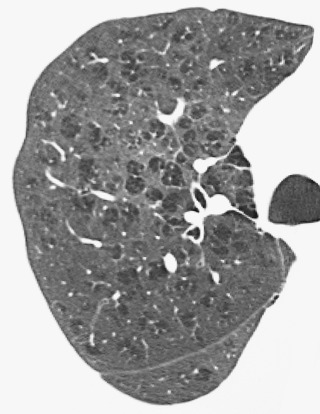
Computed tomography is superior to chest radiography in the detection of emphysema and in the assessment of its distribution and extent. On CT emphysema is characterized by the presence of areas of low attenuation that contrast with the surrounding lung parenchyma with normal attenuation ( Fig. 60.6 ). Mild to moderate centrilobular emphysema is characterized by the presence of multiple rounded and small areas of low attenuation that have diameters of several millimeters and usually have upper lung zone predominance ( Fig. 60.7 ). The lesions have no walls, as they are limited by the surrounding lung parenchyma. Sometimes, the lesions may appear to be grouped around the center of secondary pulmonary lobules ( Figs. 60.8 ). Panlobular emphysema is characterized by a uniform destruction of the secondary pulmonary lobule. This leads to widespread and relatively homogeneous patterns of low attenuation. Panlobular emphysema can involve the entire lung in a homogeneous manner or show lower lung zone predominance ( Figs. 60.9 and 60.10 ). On CT, paraseptal emphysema is seen as single or multiple bullae adjacent to the pleura or along interlobular septa ( Fig. 60.11 ). It may be an isolated finding or be associated with centrilobular or panlobular emphysema ( Fig. 60.12 ).