Gastroesophageal reflux disease (GERD) is the most common inflammatory disease involving the esophagus, with a prevalence of 10% to 20% in the West. In the past, barium studies were advocated for patients with reflux symptoms primarily to show the presence of a hiatal hernia or gastroesophageal reflux (GER), detect complications such as deep ulcers or strictures, and rule out other organic or motor abnormalities in the esophagus that can mimic reflux disease. By permitting a more detailed assessment of the esophageal mucosa, however, double-contrast radiographic techniques have made it possible to detect superficial ulceration and other changes of mild or moderate esophagitis before the development of deep ulcers or strictures. Double-contrast esophagography is also a useful screening examination for Barrett’s esophagus. With double-contrast techniques, barium studies therefore have a major role in the evaluation of patients with GERD.
Reflux Esophagitis
Pathogenesis
Reflux esophagitis is thought to be a multifactorial process related to the frequency and duration of reflux episodes, content of the refluxed material, and resistance of the esophageal mucosa. GER occurs when lower esophageal sphincter (LES) pressure is decreased or absent, so the major barrier to reflux is lost. In most patients, however, these reflux episodes result not from a sustained decrease in resting sphincter pressure but from multiple transient LES relaxations that frequently occur at night.
The severity of GERD depends not only on the frequency of reflux episodes but also on their duration. Because the duration of reflux is related to the efficacy of esophageal clearance by peristalsis, dysmotility exacerbates reflux disease and increases the risk of developing esophagitis by prolonging exposure to refluxed acid. As a result, esophageal involvement by scleroderma often leads to severe esophagitis because of absent peristalsis and extremely poor clearance of peptic acid from the esophagus after reflux has occurred. In one study, 60% of patients with scleroderma who underwent endoscopy had evidence of reflux esophagitis.
The severity of reflux disease also depends on the content of the refluxed material. Hydrochloric acid and pepsin are the noxious agents most responsible for injuring esophageal mucosa. These agents appear to have a synergistic effect, so reflux of acid and pepsin produces greater mucosal injury than reflux of acid alone. The concentration of refluxed acid is another important determinant of the degree of injury. Patients with Zollinger-Ellison syndrome are therefore more likely to develop severe esophagitis or strictures because of the high acidity of refluxed peptic juices into the esophagus.
Finally, the severity of reflux esophagitis depends on the intrinsic resistance of the esophageal mucosa. Because mucosal resistance and esophageal motor function deteriorate with age, older patients are at greater risk for developing reflux esophagitis.
Relationship Among Hiatal Hernia, Gastroesophageal Reflux, and Reflux Esophagitis
Axial hiatal hernias occur more frequently in older patients because of progressive weakening of the ligaments that anchor the gastroesophageal junction to the surrounding esophageal hiatus of the diaphragm. There is considerable controversy about the relationship between a hiatal hernia and the subsequent development of GER or reflux esophagitis. Because most patients with clinically significant GERD have evidence of a hiatal hernia, it has been postulated that the presence of a hernia predisposes patients to the development of GER and reflux esophagitis. Nevertheless, many patients with a hiatal hernia have no evidence of GER, and many patients with GER have no evidence of a hernia. Thus, intrinsic LES dysfunction is probably the major factor in the development of GER, independent of the anatomic location of the sphincter above or below the diaphragm.
Although a hiatal hernia by itself is a poor predictor of GERD, most patients with severe reflux esophagitis or reflux-induced (peptic) strictures have hiatal hernias. There is evidence that marked acid reflux causes longitudinal esophageal shortening, disrupting the ligaments surrounding the gastroesophageal junction. Thus, the hernia may represent an effect rather than a cause of esophagitis in these patients.
Similarly, GER is a poor predictor of reflux esophagitis because reflux may be demonstrated in some asymptomatic individuals but not in others with proven reflux esophagitis. Thus, the diagnosis of reflux esophagitis should be based not on the presence or absence of a hiatal hernia or GER but on specific morphologic evidence of inflammatory changes in the esophagus.
Clinical Findings
Patients with GERD classically present with heartburn—defined as retrosternal pain and burning that are worse after eating—and, less frequently, regurgitation. However, some patients may present with angina-like chest pain rather than heartburn. In one study, 43% of patients with chest pain of noncardiac origin were found to have GERD as the cause of their pain. Others may present with epigastric pain or dyspepsia that is erroneously attributed to peptic ulcer disease. Still others may have upper gastrointestinal (GI) bleeding, manifested by melena or guaiac-positive stool. However, major hemorrhage from reflux esophagitis is extremely uncommon.
An association between GERD and pulmonary problems has been well documented. Abnormal reflux has been reported in more than 80% of adult patients with asthma. These patients typically present with nocturnal coughing or wheezing and do not have an allergic component to their asthma. It has been postulated that this condition is caused by aspiration of refluxed acid into the airway or by vagally mediated bronchoconstriction secondary to reflux-induced irritation of the esophageal mucosa. Esophagopharyngeal reflux of peptic acid may also cause pharyngitis or laryngitis, manifested by a globus sensation, chronic cough, or hoarseness.
The perception of reflux symptoms depends on the degree and duration of exposure of the esophagus to refluxed acid. Nevertheless, the severity of symptoms correlates poorly with the severity of reflux esophagitis on endoscopy. Some patients with marked reflux symptoms have normal endoscopic findings, whereas others with unequivocal endoscopic findings of esophagitis are asymptomatic. It has therefore been postulated that the development of reflux symptoms is sometimes related to an esophageal visceral hypersensitivity and inappropriately heightened perception because of underlying neuronal dysfunction rather than the actual degree of esophagitis in these patients.
The development of a peptic stricture is typically manifested by slowly progressive dysphagia for solids (followed by liquids) superimposed on a history of long-standing reflux symptoms. Nevertheless, 25% of patients with peptic strictures present with dysphagia as the initial manifestation of their disease. Weight loss is usually minimal because patients with peptic strictures modify their diets to compensate for their dysphagia. Substantial weight loss should therefore raise concern about the possibility of a malignant esophageal stricture (see Chapter 23 ).
Diagnosis
Patients with reflux symptoms may undergo various clinical tests to determine whether the symptoms are esophageal in origin and whether there are objective findings of GER or reflux esophagitis. GER may be assessed by radiologic, scintigraphic, manometric, or esophageal pH monitoring techniques. Esophagography or endoscopy is required to establish a diagnosis of reflux esophagitis. Barium studies are particularly useful for evaluating patients with GERD when surgical treatment is planned.
Gastroesophageal Reflux
Spontaneous GER is detected on barium studies in only 20% to 35% of patients with reflux esophagitis. Because GER often results from transient LES relaxations rather than from a sustained decrease in sphincter tone, intermittent episodes of reflux are easily missed during the brief period of fluoroscopic observation. Some radiologists advocate the use of the water siphon test (see Chapter 17 ), which increases the radiographic sensitivity for GER to 70%. However, others do not favor the water siphon test, arguing that it is a physiologic technique with a low specificity for GER. Despite its limitations, I am a strong advocate of the water siphon test, which frequently permits detection of GER in patients in whom no reflux is observed spontaneously or with a Valsalva maneuver.
Gastroesophageal scintigraphy is an alternative technique for detecting and quantifying GER. Esophageal manometry can also be used to assess LES pressures, but many patients with normal resting sphincter pressures have intermittent GER secondary to transient LES relaxations. Intraesophageal pH monitoring is thought to be the most accurate diagnostic test for GER, but this test measures the acidity rather than the volume of refluxed material in the esophagus. Also, it has been shown that almost all patients with massive reflux on barium studies (defined as reflux of barium to or above the thoracic inlet with the patient in a recumbent position) have pathologic acid reflux on 24-hour esophageal pH monitoring. Patients with massive GER on barium studies can therefore be further evaluated and treated for their reflux disease without need for pH monitoring.
Because the severity of GERD depends not only on the frequency of reflux episodes but also on their duration, esophageal clearance may be evaluated after reflux has occurred. Fluoroscopy, scintigraphy, intraesophageal pH monitoring, and manometry are tests used to evaluate esophageal clearance or motility.
Reflux Esophagitis
Conventional single-contrast esophagography has been considered to be an unreliable technique for detecting reflux esophagitis, with an overall sensitivity of only 50% to 75%. With the use of double-contrast technique, however, the radiographic sensitivity approaches 90%. A major advantage of double-contrast esophagography is that it permits a detailed assessment of the esophageal mucosa for superficial ulceration or other changes of mild or moderate esophagitis that cannot be detected with conventional barium studies. At the same time, single-contrast technique (with the patient in the prone position) is best for demonstrating areas of decreased distensibility in the distal esophagus caused by strictures or rings. A biphasic examination with upright double-contrast and prone single-contrast views of the esophagus therefore appears to be the best radiologic technique for evaluating patients with suspected reflux disease.
Endoscopy is generally advocated as the most definitive diagnostic test for reflux esophagitis. Various grading systems have been used to estimate the severity of esophagitis based on the endoscopic findings of erythema, friability, exudates, ulcers, and strictures. Investigators have particularly focused on the importance of differentiating erosive esophagitis from nonerosive reflux disease. Nevertheless, the use of endoscopy as the gold standard for reflux esophagitis is problematic because of controversies about the endoscopic definition of esophagitis and interobserver reliability. In two studies, there was 50% or less agreement between the endoscopic and histologic findings of esophagitis. Thus, endoscopy is by no means an infallible technique for detecting this condition.
A definitive histologic diagnosis of reflux esophagitis can be made when endoscopic biopsy specimens reveal acute inflammatory changes with accumulation of neutrophils and eosinophils in the lamina propria. Basal cell hyperplasia of the squamous epithelium has also been recognized as an important sign of reflux disease, resulting from mucosal damage by refluxed acid and accelerated epithelial turnover in the esophagus. Nevertheless, the histologic diagnosis of reflux esophagitis can be unreliable because of the patchy distribution of disease. In one study, 30% of patients with reflux symptoms had normal and abnormal biopsy specimens obtained from the same regions of the esophagus. Some investigators have even questioned whether endoscopic biopsy specimens should be used as the gold standard for reflux esophagitis.
Radiographic Findings
Abnormal Motility
Between 25% and 50% of patients with reflux esophagitis have abnormal esophageal motility on manometry, manifested by weakening or disruption of primary peristalsis in the esophagus. In one study, esophagography revealed a distinct form of esophageal dysmotility in patients with GER in which there was intermittently weakened or absent peristalsis in the mid or lower esophagus without associated nonperistaltic contractions (NPCs). In contrast, esophageal dysmotility associated with aging (also known as presbyesophagus) is characterized by a combination of weakened peristalsis and multiple repetitive NPCs of varying severity. The presence of intermittently weakened or absent peristalsis without NPCs should therefore suggest underlying GERD on barium studies.
Much less frequently, esophageal aperistalsis may be the only radiographic finding in patients with reflux esophagitis. In such cases, abnormal motility may be secondary to neuronal damage in Auerbach’s plexus caused by the inflammatory process in the esophageal wall. Conversely, preexisting esophageal dysmotility (e.g., that associated with scleroderma) may predispose patients to the development of reflux esophagitis by impairing clearance of refluxed acid from the esophagus. In either case, the combination of abnormal motility and GER results in a vicious cycle, sometimes leading to progressively severe esophagitis and stricture formation.
Mucosal Nodularity
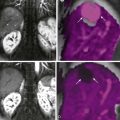
In the early stages of reflux esophagitis, mucosal edema and inflammation may be manifested on double-contrast images by a finely nodular or granular appearance in the distal third of the thoracic esophagus ( Fig. 19-1 ). In one study, mucosal granularity was the most frequent and reliable sign of reflux esophagitis on double-contrast esophagograms, with a specificity and positive predictive value of about 90%. This granularity is characterized by poorly defined lucencies that fade peripherally into the adjacent mucosa. Less frequently, reflux esophagitis may produce coarse nodularity of the mucosa. In almost all cases, this granularity or nodularity extends proximally from the gastroesophageal junction as a continuous area of disease.

More advanced reflux esophagitis may occasionally be associated with inflammatory exudates or pseudomembranes that resemble the plaquelike lesions of Candida esophagitis ( Fig. 19-2 ), but these patients usually present with reflux symptoms rather than odynophagia. A single large pseudomembrane can also be mistaken for a plaquelike carcinoma, particularly an adenocarcinoma arising in Barrett’s mucosa. However, pseudomembrane formation may be suggested by the presence of other satellite lesions or by changes in the size or shape of the lesions at fluoroscopy. When the radiographic findings are equivocal, endoscopy and biopsy should be performed for a definitive diagnosis.

Ulceration
Shallow ulcers and erosions associated with reflux esophagitis may appear on double-contrast barium studies as one or more tiny collections of barium in the distal esophagus at or near the gastroesophageal junction ( Fig. 19-3 ). The ulcers can have a punctate, linear, stellate, or serpiginous configuration and are often associated with surrounding mounds of edematous mucosa, radiating folds, and puckering or sacculation of the adjacent esophageal wall ( Fig. 19-4 ). Some patients may have diffuse ulceration of the distal third or even half of the thoracic esophagus. However, ulceration in reflux esophagitis tends to occur as a continuous area of disease extending proximally from the gastroesophageal junction, so the presence of one or more ulcers in the upper or middle thirds of the esophagus with sparing of the distal third should suggest another cause for the patient’s disease.


Reflux esophagitis may also be manifested by a solitary ulcer in the distal esophagus at or near the gastroesophageal junction. These marginal ulcers may be recognized en face as discrete collections of barium ( Fig. 19-5A ) but are best visualized when the ulcers are viewed in profile ( Fig. 19-5B ). In one study, about 70% of these solitary reflux-induced ulcers were found to be located on the posterior wall of the esophagus (see Fig. 19-5B ). Because GER often occurs during sleep, it has been postulated that patients who sleep primarily in the supine position are more likely to develop posterior wall ulcers as a result of prolonged exposure to refluxed acid that pools by gravity on the dependent (posterior) esophageal wall, causing maximal injury in this location.

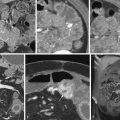
In advanced reflux esophagitis, the esophagus may have a grossly irregular contour with serrated or spiculated margins, wall thickening, and decreased distensibility secondary to extensive ulceration, edema, and spasm ( Fig. 19-6 ). Occasionally, the narrowing and deformity associated with severe esophagitis can even mimic the appearance of an infiltrating carcinoma ( Fig. 19-7 ). In such cases, endoscopy and biopsy are required for a definitive diagnosis.


Thickened Folds
In some patients with reflux esophagitis, submucosal edema and inflammation may lead to the development of thickened longitudinal folds ( Fig. 19-8 ). Thickened folds are best seen on mucosal relief views of the collapsed or partially collapsed esophagus in which folds wider than 3 mm are thought to be abnormal. These thickened folds may have a smooth, nodular, scalloped, or crenulated appearance. Occasionally, they may be tortuous or serpiginous, mimicking the appearance of esophageal varices.

Multiple delicate transverse folds may also be found in patients with GERD ( Fig. 19-9 ). In the past, this appearance has been described as the feline esophagus because transverse esophageal folds are normally found in cats. These delicate transverse striations are only 1 to 2 mm wide and extend completely across the esophagus without interruption. The folds occur as a transient phenomenon resulting from contraction of the longitudinally oriented muscularis mucosae, so they may be seen on only one of a number of spot images obtained during the radiologic examination (see Fig. 19-9 ). It has been shown that almost 100% of patients with a feline esophagus have associated GER. In fact, these transverse folds are most often observed at fluoroscopy during reflux of barium from the stomach rather than during swallowing of barium. Occasionally, the transverse folds may be thickened in patients with reflux esophagitis ( Fig. 19-10 ).


Inflammatory Esophagogastric Polyps
Other patients with reflux esophagitis may develop inflammatory esophagogastric polyps consisting of inflammatory and granulation tissue. The polyps usually appear on barium studies as smooth, ovoid, or club-shaped protuberances in the lower esophagus atop a single prominent fold that tapers distally at the gastroesophageal junction ( Fig. 19-11 ). Inflammatory esophagogastric polyps frequently straddle a hiatal hernia and may be associated with other radiographic findings of reflux esophagitis. Because these lesions have no malignant potential, endoscopy is not warranted when a typical inflammatory esophagogastric polyp is found on barium study (see Fig. 19-11A ). If the lesions have a lobulated or irregular appearance (see Fig. 19-11B ), however, endoscopy and biopsy should be performed to rule out malignant tumor.

Scarring and Strictures
Scarring from reflux esophagitis may be manifested by a variety of findings on esophagography. It is sometimes possible to detect slight flattening or puckering of the esophageal wall, radiating folds, or both in the absence of an actual stricture ( Fig. 19-12 ). Asymmetric scarring from reflux esophagitis may also lead to focal outpouching or sacculation of the distal esophagus as a result of outward ballooning of the wall between areas of fibrosis ( Fig. 19-13 ). Although these sacculations may resemble ulcer craters, they can usually be differentiated from ulcers by their more rounded appearance and changeable configuration at fluoroscopy. Sacculations are particularly likely to develop in patients with scleroderma involving the esophagus, presumably because of the severe esophagitis that occurs in these individuals. Much less frequently, patients with esophageal involvement by scleroderma may develop wide-mouthed outpouchings or sacculations in the absence of strictures as a result of asymmetric smooth muscle atrophy and fibrosis ( Fig. 19-14 ).



Scarring from reflux esophagitis may also be manifested by fixed transverse folds in the distal esophagus, producing a characteristic stepladder appearance as a result of pooling of barium between the folds ( Fig. 19-15 ). These transverse folds are usually 2 to 5 mm wide and do not extend more than halfway across the esophagus. They tend to be few in number and cannot be obliterated with esophageal distention. In most cases, there is other evidence of scarring from reflux esophagitis, and these transverse folds extend proximally a variable distance from the site of a distal stricture or scar. The folds probably represent areas of heaped-up or crinkled mucosa caused by simultaneous longitudinal scarring from reflux esophagitis. These fixed transverse folds should be distinguished from the thin transverse striations that are sometimes observed as a transient finding in patients with a feline esophagus (see Fig. 19-9 ).

Between 10% and 20% of patients with reflux esophagitis develop peptic strictures as a result of circumferential scarring of the distal esophagus. Accurate radiographic diagnosis of these strictures requires continuous drinking of low-density barium in the prone position to distend the distal esophagus and optimally demonstrate mild or even moderate strictures that are not visible on upright double-contrast images ( Fig. 19-16 ). With careful biphasic technique, barium studies have a sensitivity of almost 95% in detecting peptic strictures and may sometimes reveal strictures that are missed at endoscopy.

The vast majority of peptic strictures are located in the distal esophagus above an axial hiatal hernia ( Fig. 19-17 ). Because many patients with GER or mild reflux esophagitis do not have a concomitant hernia, it has been postulated that scarring from reflux esophagitis leads not only to circumferential narrowing of the distal esophagus but also to longitudinal shortening and subsequent hernia formation. Whatever the explanation, a hiatal hernia is found on barium studies in more than 95% of patients with peptic strictures. If the hernia fails to reduce below the diaphragm in the upright position and is a persistent finding at fluoroscopy, the patient is said to have developed a short esophagus. The latter finding has important implications for performing antireflux surgery because patients with a short esophagus may require an esophageal lengthening procedure such as a Collis gastroplasty at the time of Nissen fundoplication. Conversely, when a hiatal hernia is not present in patients with distal esophageal strictures, the possibility of malignant tumor should be considered as a possible cause of these strictures because it is unlikely for a benign stricture to develop in the absence of a hernia.

The classic appearance of a smooth, tapered area of concentric narrowing in the distal esophagus above an axial hiatal hernia should be almost pathognomonic of a peptic stricture (see Fig. 19-17A ). Many peptic strictures have an asymmetric appearance, however, with puckering, deformity, or sacculation of one wall of the stricture caused by asymmetric scarring from reflux esophagitis (see Fig. 19-17B ). Other strictures may involve a longer segment of the distal esophagus and may have irregular margins because of associated reflux esophagitis (see Fig. 19-17C ).
Most peptic strictures range from 1 to 4 cm in length and from 0.2 to 2.0 cm in width. These strictures rarely cause esophageal obstruction, but some patients may develop intermittent food impactions above the proximal end of the stricture. As many as 40% of radiographically diagnosed peptic strictures appear as ringlike areas of narrowing at the gastroesophageal junction with slightly tapered margins and a length of only 0.4 to 1 cm ( Fig. 19-18 ). Schatzki rings may produce similar radiographic findings, but they usually range from 2 to 4 mm in length and have more abrupt, symmetric margins (see Chapter 26 ). Despite these subtle distinctions, there probably is overlap between ringlike peptic strictures and Schatzki rings detected on barium studies or endoscopy.