The neoplastic lesions that can be found in the sinonasal space are numerous and varied. This article describes a general approach to narrowing down the differential diagnosis and provides the treating physician sufficient information to choose and deliver the best treatment modality. Computed tomography and MR imaging together provide complete radiologic assessment of sinonasal neoplasms.
Key points
- •
Imaging of sinonasal neoplasms is critical to map the entire extent of the lesion for management.
- •
A systematic approach is required to evaluate involvement of the critical areas related to the sinonasal space as well as nerves.
- •
Computed tomography and MR imaging complement each other in a complete evaluation of a sinonasal neoplasm.
Introduction
Sinonasal neoplasms are rare and account for 3% of all head and neck cancers. A large variety of neoplasms are possible, mainly of epithelial and mesenchymal origin; malignant lesions are more common than benign ones. Squamous cell carcinoma is by far the commonest, accounting for 80% of all the neoplasms in this region, and the maxillary sinus most frequently involved.
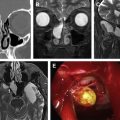
The closely apposed and communicating air-filled spaces allow for clinically silent tumor progression within the sinonasal tract. The clinical symptoms (commonly nasal congestion, epistaxis, and nasal obstruction) are so akin to the ubiquitous inflammatory and infective conditions, that the treating physician is often caught unawares. Occasionally, the neoplasm coexists with the inflammatory pathology, leading to misdiagnosis. Hence, most lesions are large at presentation and palliative options can only be offered. Many of the pathologies in this region are very aggressive, further contributing to poor prognosis. The small, restricted sinonasal space with its rich lymphatic supply as well as proximity to vital anatomic structures like orbits, skull base, palate and pterygopalatine fossa (PPF) account for early extracompartmental disease extension. There is a paucity of pain in sinonasal neoplasms until late in the disease, another important factor that leads to delayed diagnosis ( Fig. 1 ). Bony destruction, which is an important imaging feature, rarely causes pain. When there is pain, it usually is a sign of nerve or skull base involvement and heralds a poor prognosis.
The optimal assessment and treatment of sinonasal neoplasms require a multidisciplinary approach. Both surgery and radiotherapy have contributed to the management of these conditions. When curative surgery is possible, there is significant cosmetic deformity and functional morbidity. Plastic surgical reconstruction procedures are often undertaken for cosmesis. An often ignored feature of these advanced lesions and potentially morbid management options is the patient’s inability to eat, leading to early onset of cancer cachexia, which significantly contributes to poor outcome.
Both computed tomography (CT) and MR imaging have complimentary roles in the management of sinonasal tumors. The radiologist is called upon not only to detect the disease, but also to map the entire extent of surgical margins as well as radiotherapy planning.
Computed tomography and MR imaging
Only the superficial extent of a sinonasal neoplasm is visible on clinical examination and nasal endoscopy. The complete investigation of a sinonasal neoplasm mandates cross-sectional imaging. Both CT and MR imaging have played synergistic roles in the assessment of neoplasms of the sinonasal space.
Computed tomography
CT is usually the first radiologic modality to investigate a sinonasal neoplasm. It provides excellent anatomic detail of the sinonasal skeleton. CT is superior to MR imaging in evaluating matrix of fibroosseous lesions, which can aid in specific histologic diagnosis. Bony destruction of sinuses and orbital walls, as well as involvement of the anterior and middle skull base are precisely depicted in the newer multidetector CT scanners, with reconstruction in different planes. Neural foraminal widening owing to perineural spread (PNS) can be observed on thin sections and reconstructions. Fat is easily depicted on CT. Hence, for lesions that are invading the orbits, coronal CT images are reliable. All these features are of paramount surgical consideration and CT is usually a mandatory requirement from the surgical stand point. CT sections are generally reconstructed at 1- to 1.25-mm sections in the axial, coronal, and sagittal planes. When contrast is administered, the sections are taken 80 to 90 seconds after the initiation of the contrast bolus to achieve good tumor to nontumor interface as well as good visualization of the vessels of the neck. Rate of contrast injection generally is 1.5 to 2.0 mL/s. Both narrow and wide window settings need review for the soft tissue lesion as well as bone changes.
Radiation issues for CT continue to stimulate debate around the world. The new, sensitive multidetecter scanners are significantly dose efficient, with resultant low mAs scans reducing the radiation dose delivered, yet maintaining spatial resolution. Because the coronal reconstructions from axial scans in multidetector CT allow the required quality, there is no need for the direct coronal scans, thereby reducing radiation dose further. Of particular concern has been the dose delivered to the lens and thyroid gland. It has been shown conclusively that, even after multiple scans, patients have a negligible risk for premature cataract formation and thyroid cancer.
MR imaging
MR imaging has excellent soft tissue resolution and is accepted widely as the best radiologic modality to evaluate the extent of soft tissue component of tumors in the sinonasal space. The multiparametric capabilities, contrast administration, and multiplanar sequences make MR imaging a potent tool. The extension of tumors along the nerves contiguously or as skip lesions are best evaluated on contrast MR imaging. The orbital fat involvement, dural involvement, reactive changes in dura, and brain involvement are some of the areas where MR imaging plays a decisive role in formulating management options.
The protocol for MR imaging of the sinonasal space includes high resolution T1-weighted, T2-weighted, and diffusion-weighted images as well as T1-weighted, fat-suppressed, contrast-enhanced images. The sections are generally taken at 3-mm thickness with interslice gap of 0.3 mm. Axial and coronal planes are routine, but sagittal sequences are preferred when anterior skull base needs evaluation. The plane of imaging in axial sequences should be parallel to hard palate. Sagittal T1- and T2-weighted images are also indicated when evaluation of anterior skull base is required.
For evaluating PNS, high-resolution images with a small field of view of 16 to 18 cm are preferred. Thin section T1- and T2-weighted axial and coronal images with and without fat saturation are required that cover the entire course of the nerve. Contrast images are acquired in axial and coronal planes, both with and without fat suppression. MR imaging contrast (gadolinium gadopentetic acid) is administered at a dose of 0.1 to 0.2 mmol/kg body weight.
At the authors’ institution, the radiologic evaluation of sinonasal tumors consist of contrast enhanced MR imaging and multidetector CT with reconstruction in bone algorithm.
Radiologic features of a malignant sinonasal neoplasm
The radiologic features of a malignant sinonasal neoplasm can be varied. Enough evidence cannot be derived from imaging to arrive at a conclusive pathologic diagnosis in most cases. A structured approach narrows down the imaging differential. The following radiologic points raise the suspicion of a malignant tumor in the sinonasal space:
- 1.
Single site/compartment of involvement.
- 2.
Large soft tissue mass with heterogeneous enhancement and necrosis. Intermediate signal on T2-weighted images generally.
- 3.
Bone erosion/destruction.
- 4.
Contiguous multicompartmental disease with destruction of intervening boundary, be it bone or cartilage. Extracompartmental invasion to adjacent structures.
- 5.
PNS.
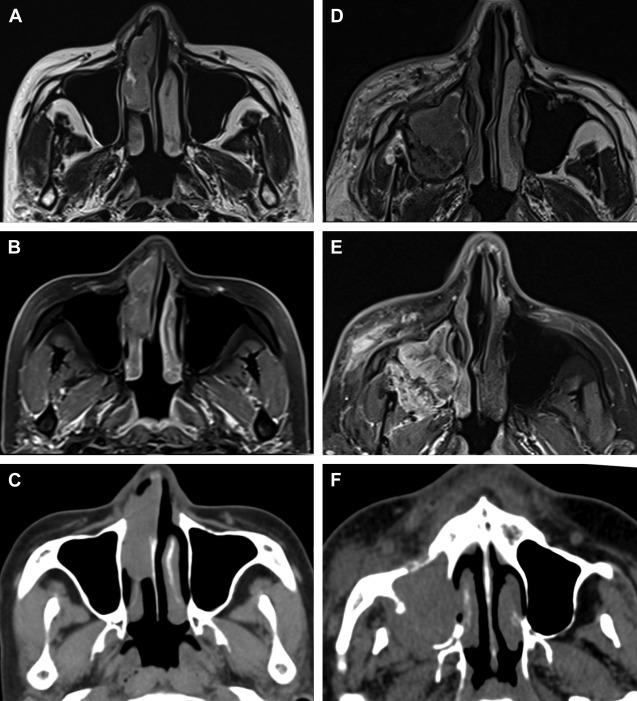
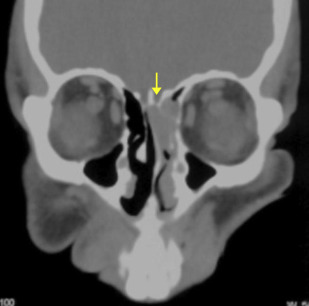
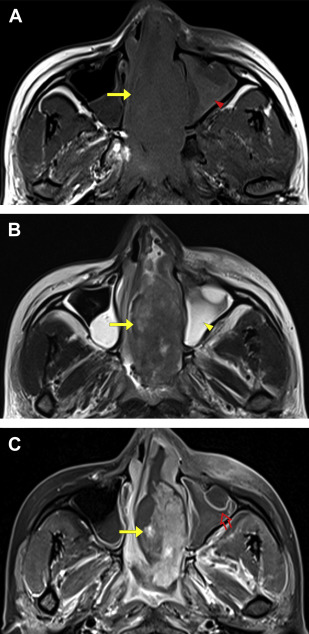
The sinonasal inflammatory diseases usually involve all or several sinuses on both sides. The changes are usually not confined to 1 side or 1 sinus only, suggesting generalized hypersensitivity of the mucosa of the entire sinonasal region. Malignant sinonasal diseases arise from a single compartment and grow into another in a contiguous manner with destruction of the intervening bone or boundary. A single site of involvement with no other sign of disease in the other compartments may suggest neoplastic disease ( Fig. 2 ). Again, large soft tissue lesions involving contiguous sites with destruction of the intervening boundary raise the suspicion of a malignant lesion ( Fig. 3 ).
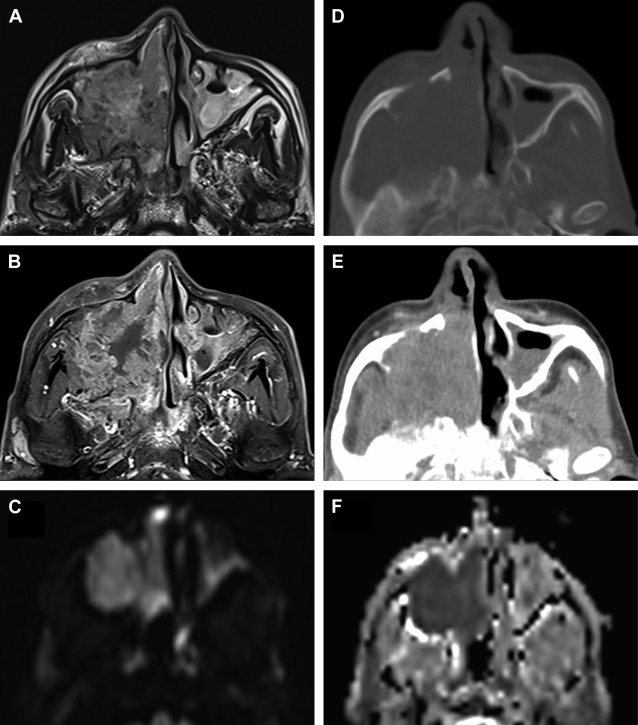
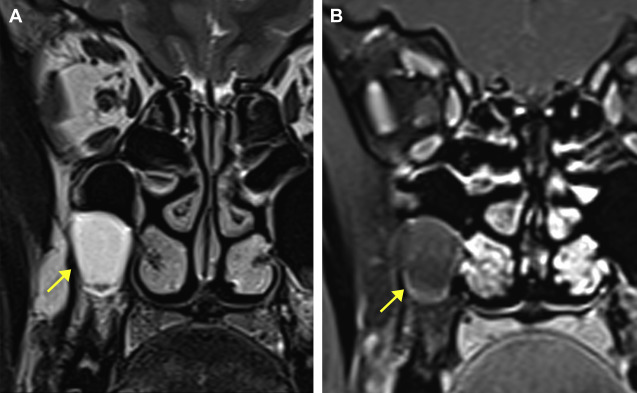
The soft tissue component of a malignant disease is large at the time of diagnosis. Areas of necrosis and heterogeneous enhancement are observed both on CT and MR imaging. The differentiation between an inflammatory polyp and a neoplastic lesion can be made on the T2-weighted images where intermediate signal of the neoplastic lesion makes the diagnosis evident. The neoplastic lesions are typically of intermediate signal intensity on T2-weighted images owing to low water content and high cellularity. Polyps are hyperintense on T2-weighted images, reflecting their higher water content, and show uniform linear enhancement of the margins ( Fig. 4 ).
Although most neoplastic lesions seem to be of intermediate signal on T2-wieghted imaging, there are certain exceptions, as in the case of low-grade adenoid cystic carcinoma, which can show high T2 signals owing to paucicellularity ( Box 1 ).
- 1.
Low-grade adenoid cystic carcinoma
- 2.
Other low-grade minor salivary gland tumors, pleomorphic adenoma
- 3.
Nerve sheath tumors
- 4.
Inverted papilloma
- 5.
Hemangioma
- 6.
Chondrosarcoma
Inspissated secretions can lead to ambiguity in diagnosis of a neoplastic lesion or delineating the exact margins of the tumor. MR imaging is the best modality to resolve this problem, because the secretions have an increased water content and show high signal intensity on T2-weighted images ( Fig. 5 ).
Chronic inspissated secretions from inflammatory conditions can produce a variety of signals on T1- and T2-weighted images. Secretions with high protein content render them hyperintense in T1-weighted images. But if the protein content is more than 28%, both T1 and T2 signals are hypointense with appearance of a pseudoaerated sinus on MR imaging. CT can be helpful in such cases. Smooth, marked peripheral enhancement is observed in inflammatory conditions. Most malignant tumors, especially large ones, are pathologically heterogeneous in nature, and this is represented on imaging. Contrast administration amplifies the heterogeneous nature of the tumor, with moderate enhancement at some parts and poor enhancement at other areas, representing necrosis. Necrosis is the hallmark of tumors, because the lesions grow rapidly, outstripping their own blood supply. Necrosis of a malignant tumor is a key differentiating feature from benign and inflammatory conditions. Hemorrhage may also be encountered in malignant lesions, appearing as focal bright areas on T1-weighted imaging and further contributing to the heterogeneous nature.
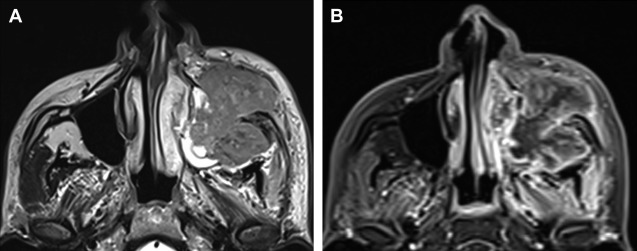
T2-weighted MR imaging is the most helpful sequence for delineating tumor boundaries. Even though contrast enhancement of the lesion may be prominent, the adjacent soft tissue and muscles can enhance significantly owing to inflammatory and desmoplastic changes induced by the tumor ( Fig. 6 ).
Diffusion-weighted MR imaging of neoplastic lesions (see Fig. 3 ) show restriction and low apparent diffusion coefficient values. This is owing to tightly packed cells in the neoplastic lesions with resultant restricted diffusion of water molecules in the intercellular space. Perfusion MR imaging has also been studied to differentiate a benign from malignant lesion. Time intensity curves plotted from dynamic contrast-enhanced MR imaging have been proven to be reliable when used together with apparent diffusion coefficient values in establishing the malignant nature of lesions.
Several bone changes are possible in a malignant lesion and they are best depicted on CT. Bone erosion and destruction are encountered in most aggressive sinonasal carcinomas. Once the bony boundary is breached, the lesions are free to involve critical areas such as the PPF, infratemporal fossa, orbits, palate, and anterior cranial fossa. Such changes, however, can be seen in certain benign conditions such as Wegener’s granulomatosis, rhinocerebral mucormycosis, and giant cell reparative granuloma. Bone remodeling is seen in several neoplasms of the sinonasal space like minor salivary gland neoplasm, lymphoma and melanoma. Natural killer–T-cell lymphoma can, however, cause aggressive bone destruction. Melanoma, despite being very aggressive, causes only bone remodeling ( Fig. 7 ). New bone formation is seen in osteosarcoma where there may be a typical aggressive periosteal reaction of the sunburst type. A chondroid type of matrix calcification is observed in chondrosarcoma. Both of these lesions usually have a large, heterogeneously enhancing soft tissue mass with necrosis. Chondroid calcification shows low signal on T1-weighted images and high signal on T2-weighted images.