Chapter Outline
Gastrointestinal Contrast Agents
Cholangiographic Contrast Agents
Magnetic Resonance Imaging Contrast Agents
Only a basic introduction and overview about contrast agent use in medical imaging are presented in this introductory chapter. These agents can be subdivided into intravascular contrast agents for computed tomography (CT) and angiography, intraluminal gastrointestinal (GI) tract agents, cholangiographic agents and a unique group of agents useful in magnetic resonance imaging (MRI). The currently active research field of contrast agent–assisted, image-guided therapy will be discussed briefly. In addition, there is a section on pharmacologic agents useful in GI radiology.
Intravascular Contrast Agents
Iodinated Water-Soluble Agents
Basic Properties
With the exception of MRI, all current intravascular contrast agents use iodine for x-ray absorption. Theoretically, sodium iodide is ideal, but its toxicity and iodism preclude its use. The complex delivery molecules developed over the years represent an attempt to deliver the greatest iodine concentration with the least toxicity. From a simplistic viewpoint, the intravascular contrast agents can be viewed merely as vehicles for delivering iodine to a blood vessel or structure. These water-soluble intravascular contrast agents can be subdivided into the following categories: (1) ionic, high osmolality, roughly five times the osmolality of blood; (2) nonionic, low osmolality, roughly twice or slightly more than the osmolality of blood; and (3) isotonic agents—nonionic dimers. The basic structures and physicochemical characteristics of available contrast agents are not covered here; these topics are discussed in appropriate specialized publications.
At the x-ray energies used in CT, the mass attenuation coefficient for iodine is considerably greater than that of surrounding soft tissues and blood. After the intravascular injection of iodinated contrast, initial CT images reveal aortic and major arterial enhancement, followed by a capillary or parenchymal “blush” and eventual venous opacification. The rate of contrast injection and timing of subsequent CT scans determine the structures enhanced on any one image. Compared with earlier scanners, multidetector CT (MDCT) scanners require shorter injection rates because of the short scanning times; as a result, faster injection rates of more concentrated contrast agents are necessary, and a contrast’s viscosity has a dominant role. Various techniques of intravascular contrast agent administration are discussed elsewhere in this text.
A number of drugs, especially more acidic ones, are incompatible to mixing with contrast agents, an incompatibility less evident with nonionic agents. Nevertheless, as a general safety precaution, a drug probably should not be mixed with a contrast agent, and a catheter should be flushed if used for both drug and contrast injection.
Ionic Agents
Acetylation of aminotriiodobenzoic acid and further structural changes led to the development of ionic contrast agents. These agents are formulated as salts and consist of a cation and anion. Two commonly used cations are sodium and meglumine. The anion portion of the molecule consists of a benzene ring containing iodine substituted at positions 2, 4, and 6, plus a number of other side chains. These side chains determine water solubility and indirectly affect resultant toxicity. The benzene ring can be viewed as a scaffold for attaching iodine and side chains. When the molecule dissociates, three iodine atoms are available for every two particles in solution, or a ratio of 1.5 : 1.
Further refinements of ionic contrast media consist of the attachment of two monomer triiodinated benzene rings at one of the side groups. Such a dimer, containing two benzene rings, with each having three iodine atoms and only one cation particle, has six iodine atoms per two particles, or a ratio of 3 : 1.
Ionic contrast agents are hypertonic at the concentrations used for vascular opacification; considerable effort has been spent in an attempt to decrease their osmolality. In general, the viscosity of a sodium salt is less than that of a corresponding meglumine salt, but the sodium salt tends to be more toxic. Toxicity and viscosity limitations during intra-arterial injections are not as relevant for the intravenous (IV) injections used with CT.
Nonionic Agents
If the carboxyl group in position 1 on the benzene ring is replaced with a stable side group, the molecule no longer dissociates when in solution, and each particle in solution has three iodine atoms, or a ratio of 3 : 1. A dimer structure can also be achieved by linking two triiodobenzoic acid molecules (ioxaglic acid), formulated as a meglumine sodium salt, another contrast agent with a ratio of 3 : 1. A contrast agent with a ratio of 6 : 1 has also been developed (iodixanol) and is often referred to as being iso-osmolar.
Various manufacturers have taken different approaches to the type of side chains used with ionic and nonionic contrast agents. As a result, these compounds differ in their viscosities and other properties. The interaction with other molecules also differs between ionic and nonionic agents and is affected by the type of side branches present. Within limits, however, for each group of contrast agents, viscosity varies directly with iodine concentration.
Commercial contrast agents also contain chelating agents, usually calcium edetate disodium, to chelate impurities and buffering agents to achieve an acceptable pH. Their action is more important during contrast agent manufacture than during clinical use.
The American College of Radiology (ACR) has published criteria for situations in which nonionic agents are preferred, although in many practices this is a nonissue because nonionic agents are used almost exclusively. The nonionic agents are associated with less patient discomfort and thus result in less motion artifacts; this is an evident advantage, especially with complex examinations such as three-dimensional (3D) reconstruction.
Pharmacokinetics
After a bolus intravascular injection, the initial plasma iodine concentration is determined by contrast agent iodine concentration and injected volume. Ionic and nonionic contrast agents are eventually distributed throughout the extravascular, extracellular space, with intravascular and extravascular equilibrium achieved within 10 minutes after intravascular injection. They are excreted mostly by renal glomerular filtration.
After injection, the relative plasma iodine concentration in a particular vessel depends on dilution by blood, extravascular diffusion, and renal excretion; the first factor is most important during arterial and venous phase imaging, with extravascular diffusion playing a larger role during the parenchymal phase. In theory, a contrast agent can be designed to have fast or slow extravascular diffusion and rapid or slow renal clearance; an ideal blood pool agent should have slow extravascular diffusion. In practice, with an equivalent iodine dose, nonionic agents achieve greater initial peak vascular enhancement than ionic agents, but subsequent blood iodine concentrations and parenchymal opacification are similar for the two types of agents (except renal visualization). Ionic and nonionic agents have similar extravascular diffusion rates. Extensive literature is available on relative time-dependent blood iodine concentrations and renal excretion of various contrast agents.
Dynamic CT scanning after a single bolus injection relies on the enhancement of vascular structures above baseline. Correct arterial phase timing is obtained by injecting an initial test dose or using automatic bolus tracking. One mg iodine per gram of tissue corresponds roughly to an increase of 30 Hounsfield units (HU), which is about the limit for detection. In general, it is desirable to have sufficient iodine concentration in the vascular structures of interest to elevate them above baseline by up to 100 HU. With this degree of enhancement, major vessel thrombi are detected and vascular fistulas and related conditions evaluated. Whether early dynamic scanning (arterial phase) is superior to portal venous phase or even delayed scanning after contrast equilibration depends on the organ in question and information sought. For the liver, these arterial and portal phase time window are roughly 20 to 30 seconds. These short time intervals are readily achieved with MDCT.
A typical CT examination consists of a precontrast scan, followed by scanning after the initial bolus reaches the structure of interest. A relatively large-caliber venous catheter and power injector provide reproducible injection rates, keeping in mind that prediction of bolus arrival is somewhat empiric because, among other factors, decreased cardiac output can prolong vascular flow times.
Intravascular contrast agents cross the placenta, are excreted in breast milk, and affect fetal and infant thyroid function. If feasible, alternate studies should be considered during pregnancy. Breast-feeding should be stopped for 1 to 2 days after contrast injection.
Acute Adverse Reactions
Only a brief summary of contrast reactions and their therapy are provided here. More detailed information is available from ACR and European Society of Urogenital Radiology (ESUR) publications.
The nonionic contrast agents have a considerably lower osmolality than the ionic agents; adverse reactions caused by hyperosmolality are therefore reduced with the nonionic agents. Hyperosmolality is also related to vasodilation of the involved capillaries. Nonionic contrast agents induce less hypotension than ionic agents.
Risk factors for acute renal failure include diabetes mellitus with decreased renal function, renal insufficiency, dehydration, and use of a high dose. Atopy confers an increased risk to contrast reactions, but allergies to shellfish appear not to increase the risk of reaction. Iodinated contrast agents are contraindicated in patients with obvious hyperthyroidism. Also, these agents should be avoided for 2 months prior to thyroid isotope imaging or radioactive thyroid therapy.
Types of Reactions.
Acute reactions vary from minor effects to severe and life-threatening. Sensations of warmth, nausea, and vomiting appear to be a direct side effect to contrast. Reactions such as mild changes in blood pressure or mild wheezing are often self-limiting or may progress to more severe reactions. An arbitrary but useful grading of contrast reactions is mild, moderate, severe, and fatal. Compilations of reactions to ionic contrast agents in the 1970s and 1980s revealed a risk of severe reaction to be 1 in 1000 to 4000 studies. Types of reactions are similar with ionic and nonionic agents. In general, the risk of adverse reactions varies with contrast osmolality, so fewer reactions occur with nonionic agents than with ionic agents. In particular, the risk of severe adverse reactions is lower with nonionic contrast agents. Deaths have occurred with both ionic and nonionic agents.
At times, urticaria and even more severe reactions do not represent a classic antigen-antibody reaction but are secondary to histamine or serotonin release induced directly by the contrast agent. However, histamine release is probably not the only factor involved in serious contrast reactions. Among other effects, contrast agents activate the complement system, which acts as a host defense, and is related to coagulation abnormalities and bradykinin release. Overall, only a minority of unpredictable reactions mimics immunoglobulin E (IgE) hypersensitivity, probably secondary to an antigen-antibody reaction. How to classify the rare bowel wall edema is not clear. Iodide mumps is a rare delayed reaction to iodine-containing contrast media.
Some reactions are disease specific. IV contrast agents in the presence of a pheochromocytoma can lead to catecholamine release and acute hypertension. In this setting, the onset of such hypertension should suggest a pheochromocytoma.
Over the years, many radiologists have avoided the use of intravascular contrast agents in patients with sickle cell disease, although the prevalence of adverse reactions has not been established. Over an extended period, bottled hyperosmolar contrast agents can leach allergens from rubber stoppers. As a rule, contrast-containing vials and bottles should be stored in an upright position.
Premedication.
The specific allergen responsible for iodinated contrast sensitization is unknown. It is difficult to prove that iodine is responsible for hypersensitive contrast reactions, a common assumption. A myosin protein rather than iodine is believed to be the allergen responsible in shellfish. Rather than ask a patient about iodine allergies, a more appropriate question appears to be whether drug allergies are present. On a practical level, the cause of an adverse reaction is often not sought, and the reaction is simply labeled as allergic, hypersensitive, or anaphylactic.
No reliable blood test detects patients who are allergic to contrast media. Risk factors associated with a contrast reaction include asthma and a history of prior reaction to contrast. However, even these are unpredictable, and a patient manifesting a severe reaction may have had prior intravascular contrast with no adverse reaction. Although patients with urticaria-like reactions have increased plasma levels of prekallikrein and α 2 -macroglobulin and lower levels of C1-esterase inhibitor, their predictive value is limited because of normal variation. Pretesting with a small dose of contrast was once popular but has been abandoned as having little or no value. Acute reactions have developed after less than 1 mL of administered contrast.
In a multi-institutional study involving ionic contrast agents, pretreatment with methylprednisolone, 32 mg, 12 hours and 2 hours before contrast injection, significantly reduced the risk of reactions. With this two-dose regimen, the number of reactions in patients receiving ionic contrast agents approximated those seen with nonionic agents and no pretreatment. Premedication is often considered for patients who have had a previous reaction to a contrast agent. Regimens that have been proposed range from 3 days to immediately before a scan. At the University of Rochester, we recommend that patients who have had a significant prior reaction to IV contrast agents be pretreated with 50 mg of prednisone orally every 12 hours, for a total of three doses, with the last dose given approximately 1 hour before the examination, and 25 to 50 mg of diphenhydramine hydrochloride (Benadryl) orally, 2 hours before the examination.
The prevalence of seizures after IV contrast injection is increased in patients with brain metastases. Capillaries in brain metastases do not exhibit normal blood-brain barrier integrity and are permeable to a contrast agent. To decrease the risk of seizures, it has been suggested that these patients be premedicated with diazepam, 5 mg IV, before contrast administration.
Treatment of Reactions.
Any physician injecting a contrast agent intravascularly can expect to encounter a broad spectrum of reactions, from mild to severe, and must be prepared to deal with them. In general, mild reactions such as flushing or mild urticaria require no treatment, and most reactions resolve spontaneously. Similarly, nausea and vomiting require general support and observation only. If symptoms occur before all the contrast agent has been administered, the rate of injection should be slowed or the injection postponed until symptoms clear.
Early IV access should be established. The catheter used for contrast injection should be kept in place, ensuring intravascular access until the possibility of a reaction has passed. With progressive hypotension, it becomes increasingly difficult to cannulate a peripheral vein.
Moderate urticaria developing in the absence of other significant symptoms can be treated with diphenhydramine, 25 to 50 mg, orally or injected. With more severe urticaria, one should also consider an H2 blocking agent such as cimetidine (Tagamet), 300 mg injected slowly (diluted) IV. For severe urticaria, epinephrine, 0.1 to 0.3 mL (1 : 1000) should be given subcutaneously unless contraindicated. If needed, the dose can be repeated in 15 minutes. Epinephrine should be used with caution in older patients who have underlying cardiovascular disease; electrocardiographic monitoring should be considered for these patients.
Severe reactions, such as severe bronchospasm, convulsions, or significant cardiopulmonary reactions, require prompt and vigorous therapy. Bronchospasm and laryngeal edema generally respond to subcutaneous epinephrine. If needed, the epinephrine dose can be repeated. Diphenhydramine and corticosteroids, such as hydrocortisone, 100 to 300 mg IV, are also often used. Oxygen should be administered by mask or nasal cannula. Beta agonist inhalers alone may be beneficial for mild bronchospasm or can be used in conjunction with aminophylline therapy. With refractory bronchospasm, aminophylline, 250 to 400 mg diluted in dextrose and water, can be administered IV over a 10- to 20-minute period. Aminophylline should be used with caution because it could exacerbate coexisting hypotension. Tracheal intubation should be considered early in the course of these symptoms; later, severe laryngeal edema may make intubation difficult if not impossible.
Because the treatment of hypotension in the settings of tachycardia and bradycardia is different, the pulse rate should be monitored. A pulse may not be palpable in a hypotensive patient; cardiac auscultation or electrocardiographic monitoring may be necessary.
Hypotension in the absence of other major signs of an anaphylactic reaction should initially be treated with oxygen, leg elevation, and rapid administration of IV fluids. Epinephrine should be considered, keeping in mind that fluid therapy alone may be sufficient therapy. Although subcutaneous epinephrine injections are adequate for a mild to moderate reaction, IV administration is needed for moderate to severe hypotension. For IV administration, epinephrine should be diluted to 1 : 10,000 and 1.0 to 3.0 mL administered slowly. The dose can be repeated in 15 minutes, and the rate of injection can be titrated to achieve the desired result. A vasopressor agent such as dopamine, 2 to 5 µg/kg/min, can be added to sustain blood pressure. For unresponsive hypotension, other agents are available for treatment of underlying shock. An H2 blocker such as cimetidine can be added, 300 mg in dextrose and water, infused slowly. Similarly, diphenhydramine, 25 to 50 mg, can be injected IV. Corticosteroids are also often used, with a typical dose of hydrocortisone being 500 mg IV. Steroids probably have no immediate effect on a reaction; their main use is to decrease delayed reactions.
At times, hypotension can be corrected with vigorous hydration alone, keeping in mind that overhydration of patients with possible underlying cardiovascular and/or renal disease also carries a risk. Thus, the initiation of therapy by adequate hydration is reasonable, but appropriate pharmacologic therapy should be instituted without undue delay.
Hypotension in the presence of bradycardia suggests a vasovagal reaction. Some patients respond to being placed in a Trendelenburg position. Hypotension in these patients should be treated with rapid IV infusion of isotonic saline. Oxygen should be administered. Bradycardia can be treated with atropine (0.5 to 1.0 mg IV), with the dose repeated every 5 minutes, to a maximal total dose of 3.0 mg.
Some patients receive long-term therapy with a beta blocker such as propranolol. A contrast reaction in these patients can be confusing because, even in the setting of anaphylactic shock, a beta blocker–induced bradycardia can persist. IV glucagon, 1.0 mg or more, may be useful for bradycardia. Dopamine is also effective. Doses of epinephrine that are usually administered may not be effective in reversing this hypotension.
Emergency cardiopulmonary resuscitation is necessary for cardiovascular collapse. Refractory seizures are treated with IV diazepam (Valium) and/or phenobarbital.
Contrast extravasation is treated by extremity elevation, warm or cold compresses and, if extensive, a plastic surgery consultation. Hyaluronidase, an enzyme that breaks down interstitial barriers, has been injected into the extravasation site by some investigators, but its impact on tissue healing is still not clear.
This discussion is only meant to be a guide. The treatment of all reactions should be individualized.
Contrast-Induced Nephropathy.
The pathogenesis of contrast-induced nephrotoxicity is incompletely understood, but it is believed that a number of intrinsic renal events lead to renal medullary ischemia, usually augmented by a reduced intravascular volume. Direct cytotoxicity, oxidative tissue damage, and apoptosis are contributing factors. This nephrotoxicity is manifested by a significant rise in the serum creatinine level. Various authors use different definitions of significant, with the ESUR guidelines using a creatinine level increase of more than 25%, or 44 µmol/L (0.5 mg/dL), within 3 days. A transient nonoliguric decrease in renal function lasting up to 3 weeks is more common than the more ominous oliguric manifestation, which may require hemodialysis.
Risk factors for nephrotoxicity appear to be multifactorial and include preexisting renal insufficiency, diabetes, dehydration, cardiovascular disease, advanced age, myeloma, hypertension, hyperuricemia, and possibly contrast osmolality and dose. Patients at the greatest risk for acute renal failure are diabetics with preexisting renal insufficiency. Precaution is also necessary with treatment by agents such as nonsteroidal anti-inflammatory drugs (NSAIDs), aminoglycosides, cyclosporin, or even sulfonamides; underlying nephrotoxicity is a common pathway.
In diabetics with underlying cardiovascular or renal insufficiency, metformin, a biguanide antihyperglycemic agent, is associated with lactic acidosis and resultant increased mortality. This association appears to be indirect and probably involves underlying renal insufficiency. However, enough patients taking metformin and receiving IV contrast have developed lactic acidosis to prompt the U. S. Food and Drug Administration (FDA) to issue a warning—metformin should be discontinued before or at the time of contrast use for 48 hours after the procedure and reinstated only if renal function remains normal. However, substantial inconsistencies exist in the guidelines. Adequate hydration should be maintained.
Because iodinated contrast agents are not protein-bound (except for cholangiographic agents), they can be dialyzed. In patients on hemodialysis, additional hemodialysis sessions are generally not necessary.
The most important preventive measure is to ensure that the patient is well hydrated. If IV hydration is necessary, some evidence suggests that the IV use of sodium bicarbonate hydration is superior to sodium chloride. Other guidelines include the use of low-osmolar contrast, discontinuing nephrotoxic drugs for at least 24 hours, and consideration of alternate imaging in high-risk patients. The osmotic diuretic mannitol does not provide any benefit; this loop diuretic furosemide exacerbates renal dysfunction. Currently, it is probably safe to assume that diuretics do not offer any protective effect, and anecdotal evidence even suggests that diuretics should be stopped prior to a contrast study.
N -acetylcysteine, an antioxidant, appears to diminish contrast-induced renal toxicity, although some have questioned its renal benefit. Many of these studies have involved coronary angiography and differ in methodology from contrast CT. The role for theophylline is less well established.
Hemodialysis after contrast use in preexisting renal failure patients is not thought to be warranted. Hemofiltration in chronic renal failure patients, on the other hand, has caused less creatinine level increase than in controls. The complexity of this procedure and the cost of hemofiltration limit its use to select patients.
Iodinated Oil
Intra-arterial iodized poppy seed oil (Ethiodol or Lipiodol) is used for several indications:
- 1.
As a CT diagnostic agent for liver tumor detection, especially hepatocellular carcinoma.
Often used as a gold standard, it detects more tumors than other imaging modalities. Nevertheless, a study of explanted livers revealed that pretransplantation iodized oil CT tumor sensitivity is still rather low. Also, one should keep in mind that iodized oil is retained by some benign tumors, even hemangiomas.
- 2.
Ethiodol is often included as a chemoembolization ingredient when injecting into a tumor feeding artery.
This acts as a chemotherapeutic agent carrier and, because of its high viscosity, is a temporary embolizing material that prolongs chemotherapeutic agent contact with a tumor. Ethiodol remains within tumor neovascularity much longer than in normal liver parenchyma and thus acts as a marker.
- 3.
Occasionally, ethiodol is injected during percutaneous radiofrequency ablation of hepatocellular carcinomas.
This aids in the CT delineation of extent of coagulation necrosis.
For a number of reasons, intra-arterial iodized oil is considerably more popular in the Far East than in the West.
Other Agents
Several reports have described the use of gadolinium (Gd)-based contrast agents for CT imaging in patients with renal insufficiency or prior severe reaction to an iodinated agent. One should keep in mind, however, that the pharmacokinetic properties of gadopentetate dimeglumine (Gd-DTPA), with only one gadolinium ion, are similar to iodinated agents containing three to six iodine atoms. Also, the toxicity of gadolinium agents, at doses achieving equivalent x-ray stopping power, is greater than that of nonionic iodinated agents. This is in distinction to the use of approved lower gadolinium MRI doses, which are insufficient for useful x-ray contrast, but have negligible nephrotoxicity. The ESUR position is that gadolinium-based contrast agents are more nephrotoxic than iodinated contrast agents in equivalent x-ray attenuation doses.
Carbon dioxide is a viable angiographic contrast agent for certain digital vascular indications in the abdomen. It has been used as a guide for vascular interventional procedures. It displaces blood, forms a gaseous column, and is cleared by the lungs.
Apart from MR, viable abdominal reticuloendothelial contrast agents have eluded clinical development. The first such agent, thorium dioxide (Thorotrast), used until the 1950s, has left a painful and sorry legacy. Iodinated oily emulsions accumulate in the liver, spleen, bone marrow and, to a lesser extent, in other organs, long enough to permit CT imaging, but high toxicity and low specificity led to their abandonment. Colloidal iodine or emulsified perfluorooctyl bromide particles are also incorporated in reticuloendothelial cells. Most studies of emulsified perfluorooctyl bromide took place in the 1980s, and its use has lost favor since then.
Liposomes are taken up by the reticuloendothelial system, and considerable effort has been expended to encapsulate water-soluble iodinated contrast agents inside liposomes. Research activity peaked during the 1990s but, in spite of occasional more recent papers, pronounced adverse reactions limit the use of liposomal CT contrast agents in humans. Most current reticuloendothelial contrast research revolves around MRI agents.
Gastrointestinal Contrast Agents
Barium Sulfate
Barium sulfate is a white crystalline powder having a molecular weight of 233. Because of its specific gravity of 4.5, patients tend to comment that a cup of barium suspension is “heavy.” The terms thick and thin should only be used when referring to viscosity. They should not be used to signify radiodensity, which has many other causative factors.
Although barium sulfate itself is inert and does not support bacterial growth, some additives in commercial preparations are organic. When a container is opened or reconstituted with tap water, the suspension should be refrigerated if it is to be kept overnight. Although many commercial formulations contain preservatives, bacterial contamination can and does occur.
Certain commercial formulations are advertised as being applicable throughout the GI tract. Invariably, these represent a compromise. The GI tract varies in pH, in composition of mucus, and in type of mucosa, and optimal coating in one part does not mean that a similar coating can be expected in another. Coating the mucosa with barium or simply opacifying the lumen requires different barium formulations. The large-particle, high-density barium suspensions designed for double-contrast use should not be simply diluted and used for single-contrast studies. Ingesting such a diluted suspension causes rapid barium particle sedimentation, with the nondependent lumen containing little barium; lesions on the nondependent wall can therefore be missed. Products designed primarily for single-contrast examinations, on the other hand, can be diluted considerably before any settling occurs, mainly because they contain relatively small barium particles.
Ingestion of a barium sulfate suspension tends to be constipating. Currently, commercial barium products have additives that minimize this effect, and the formation of a bariolith is rare.
Pharyngographic Agents
Pharyngeal radiography was already established in the 1960s, when cineradiography became popular to evaluate dysphagia. Although conventional or digital radiography produces high anatomic resolution, dynamic swallowing is best evaluated with video fluoroscopy or cineradiography. This pharyngogram, commonly called a modified barium swallow, evaluates oropharynx anatomy and function using contrast agents of varying consistencies.
After a stroke, appropriate patient feeding without inducing aspiration can be determined by using barium suspensions of different viscosities and barium-coated food. Contrast consistencies used range from barium-coated crackers to a viscosity approaching that of water. To improve patient acceptance, some investigators have developed their own contrast agents such as barium pudding or barium honey. Anatomic detail is best studied with high-density barium products such as the 250% w/v suspensions designed for gastric double-contrast examinations. Fistulas are also best studied with this type of contrast agent. A barium paste can also be used to study anatomy, but the high paste viscosity limits its application in fistula detection. The volume of barium used should be individualized. Thus, with suspected aspiration, a several-milliliter bolus is swallowed initially; if no aspiration is detected, the bolus is gradually increased in volume.
The oropharynx handles high- and low-viscosity liquids differently, so pharyngeal function should be studied with high- and low-viscosity barium suspensions. The low-viscosity suspension should have a viscosity approaching that of water, whereas the high-viscosity suspension should be similar to a thick milkshake. It should be emphasized that some high-density double-contrast barium products are relatively fluid and are not applicable as high-viscosity preparations.
A tracheoesophageal fistula is easier to detect fluoroscopically with the patient in the lateral position. With the patient in a frontal position, it may not be possible to determine whether barium in the trachea was aspirated or flowed through a fistula.
Upper Gastrointestinal Tract Studies
Studies of the esophagus consist of single-contrast, double-contrast, and mucosal relief views, together with fluoroscopic evaluation of motility. Normal esophageal tonicity leads to lumen collapse when a primary peristaltic wave has passed. Therefore, regardless of which method is used, the study must be performed reasonably quickly.
Some patient symptoms are reproduced by using a cold contrast suspension or acidic contrast. Although some have found acidified contrast useful, it is not commonly used.
Sufficient air is introduced into the esophagus for a double-contrast study in some patients with poor esophageal motility or those with gastroesophageal reflux. In most patients, however, an additional negative contrast agent is necessary to obtain double-contrast views. Solid gas-producing tablets, powder, or liquid effervescent agents are used. These contain sodium bicarbonate and an acid, such as tartaric acid or citric acid, which, in the presence of a liquid, produce carbon dioxide. About 400 to 500 mL of gas is necessary for adequate esophageal and gastric distention. One technique is to have the patient drink, in quick succession, first one and then another liquid effervescent solution, followed immediately by 60 to 120 mL of a barium suspension. The two effervescent agents distend the esophagus by releasing carbon dioxide, and barium then coats the esophageal mucosa.
The high-density, low-viscosity barium products designed for the stomach and duodenum also coat the esophagus wall. Visualization of the esophagus is impaired if barium is ingested before effervescent agents. On the other hand, the quality of the gastric mucosal coating is improved if the barium suspension is given first. The sequence of ingestion can be tailored to the patient’s symptoms—if esophageal disease is suspected, the effervescent agents are given first; if gastroduodenal disease is suggested, the barium suspension is given first.
Esophageal varices tend to be more prominent and their detection easier if the esophageal lumen is collapsed. Although high-density, low-viscosity barium products detect larger esophageal varices, commercially available barium pastes are recommended. Some of these pastes are too viscous and tend to flow in a bolus; these should be diluted with water so the paste viscosity is similar to that of honey.
In a patient with acute dysphagia, an esophagram can be therapeutic. With the patient upright, the weight of a barium column can dislodge a foreign body into the stomach. Liquid effervescent agents increase the intraluminal esophageal pressure and may also push a foreign body into the stomach. This technique should be performed with care to avoid perforation. Glucagon has also been proposed to help relieve spasm, although it is not clear whether pharmacoradiology has a significant role in acute dysphagia.
Commercial barium sulfate tablets with a diameter of 12 mm are available and are useful to evaluate subtle esophageal strictures. For results to be meaningful, the patient should be at least in a 45-degree upright position and at least 60 mL of water should be ingested with the tablet. Tablet transit time through the esophagus normally is less than 20 seconds. These tablets contain 650 mg of barium sulfate plus additives. The tablets dissolve in the esophagus or stomach ( Fig. 1-1 ). Relatively fresh tablets should be used because older tablets take longer to dissolve.

It has been proposed that barium tablets be administered routinely during chest radiography because tablet retention in the esophagus is associated with the presence of structural or functional esophageal abnormalities, but this technique is not widely used.
In patients with suspected esophageal perforation, study of the esophagus with a water-soluble agent may not detect a subtle leak. Thus, administration of higher density barium sulfate enables detection of leaks that might otherwise be missed. For this part of the examination, barium suspensions varying from 35% to 80% w/v are used. Residual barium in the mediastinum does not result in clinically detectable mediastinitis and does not interfere with subsequent radiographic evaluation.
High-density, low-viscosity barium preparations specifically designed for the upper GI tract produce best double-contrast results. A volume of 60 to 120 mL of a 250% w/v suspension is generally sufficient. A good barium formulation should result in routine identification of the areae gastricae. Small cancers, ulcers, gastritis. and duodenitis are readily detected during a high-quality examination.
When appropriate double-contrast gastric views have been obtained, a lower density barium suspension is ingested for subsequent single-contrast evaluation. For this part of the examination, barium suspensions varying from 35% to 80% w/v are used. Various external compression paddles are available and are helpful in obtaining mucosal relief views.
Small Bowel Studies
A number of techniques have been developed to study the small bowel, such as conventional antegrade study, enteroclysis, retrograde ileography, peroral pneumocolon, and CT and MR enterography. The type of examination performed varies with clinical indication. Specific contrast agents have been developed for each type of study.
An antegrade examination is the simplest and most traditional way of studying the small bowel. Serial small bowel radiographs are obtained after the patient ingests a barium suspension. The primary contrast agent requirement is that it does not flocculate or precipitate during transit. The barium does not coat the mucosa; visualization is obtained primarily by filling the bowel lumen with the barium suspension. A 40% to 60% w/v suspension is typical. Many radiologists prefer a volume of 500 to 800 mL.
Contraindications to an antegrade barium study are suspected colonic obstruction or bowel perforation. A number of clinicians hesitate to request a barium study in the setting of small bowel obstruction and prefer to wait until the obstruction clears. Such an approach is analogous to obtaining a chest radiograph only after a pneumonia clears. Small bowel obstruction is not a contraindication. Barium proximal to a small bowel obstruction continues to stay in suspension, and barium inspissation does not occur. With a small bowel obstruction, an antegrade study with barium is safe and not only can detect the site of obstruction but also suggest a cause.
In enteroclysis (small bowel enema), contrast is injected through a steerable catheter directly into the small bowel, so the flow-limiting function of the pylorus is bypassed. The barium suspension can be infused by gravity, hand-held syringes, or an infusion pump. Typical infusion rates are 75 to 100 mL/min, although the flow rate should be individualized. If the rate is too slow, excessive peristalsis results in a study similar to a conventional antegrade small bowel examination. With a flow rate that is too high, overdistention leads to bowel atonia and lack of progression.
It is debatable whether single-contrast or double-contrast enteroclysis yields better results. For the double-contrast portion, many U.S. investigators use a solution of methylcellulose in water (≥0.5%). Methylcellulose helps propel a barium suspension ahead of it. Water can be used as the second contrast agent, although water tends to wash off barium adhering to the mucosa. The total volume of the two contrast agents is tailored for each examination; in some patients, up to 2 L are required. The contrast agents are instilled until a lesion is detected or contrast reaches the right colon. If needed, glucagon is administered to induce hypotonia.
Air as a second enteroclysis contrast agent, used more commonly in Japan and Europe, results in considerably more radiographic contrast than obtained with methylcellulose. Air does not propel the barium ahead of it as quickly as methylcellulose; it tends to percolate through barium-filled loops of bowel. Nevertheless, with overlapping bowel loops, as is common in the pelvis, infusion of air is often helpful. Air bubbles can be confusing, although some think that better diagnostic results can be achieved with air, even in patients with inflammatory bowel disease.
Several tubeless enteroclysis techniques have been described. One simple method is to initially perform a conventional small bowel study, but when barium approaches the cecum, the patient swallows additional effervescent agents, turns prone, and the table is turned into a 20- to 40-degree Trendelenburg position. Efflux of gas results in a double-contrast small bowel study. A tubeless double-contrast small bowel technique consists of effervescent granules coated with an acid-resistant lacquer. Gas is released directly into the small bowel lumen.
Barium formulations specifically designed for enteroclysis provide the best results and are commercially available. For a single-contrast study, a barium suspension having a specific gravity of about 1.25 (equivalent to ≈35% w/v) is typical. For a double-contrast study, the barium suspension should be in the range of 50% to 95% w/v.
Retrograde ileography consists initially of a single-contrast barium enema, but infusion of barium is then continued retrograde into the ileum. Because flow is controlled by the examiner, the ileum can be readily studied without overlapping loops from the more proximal small bowel. Barium is instilled until the region in question is reached. Premedication with glucagon increases patient comfort and also relaxes the ileocecal valve. If a redundant sigmoid colon obscures part of the small bowel, the barium enema can be followed by a saline enema; such a solution pushes barium ahead of it and results in a see-through effect. A 20% to 25% w/v barium suspension is typical for such a study.
It is not unusual to achieve a double-contrast study of the terminal ileum during a double-contrast barium enema, especially if glucagon is used. This type of study is useful in suspected ileal Crohn’s disease or gynecologic malignancies involving the ileum.
A peroral pneumocolon, consisting of antegrade and retrograde components, is designed to evaluate the distal ileum or right colon. Initially, a conventional antegrade small bowel examination is performed. When barium outlines the terminal ileum, air is instilled through the rectum to obtain double-contrast views of the distal small bowel or proximal colon. This study can also be combined with enteroclysis. Routine use of a hypotonic agent is helpful.
Barium Enema
Single-contrast and double-contrast techniques are well established methods of evaluating the colon. Numerous studies have compared the relative accuracy of a single-contrast versus double-contrast barium enema. Some radiologists prefer a single-contrast study in older or debilitated patients.
Dry and liquid barium formulations are commercially available. If dry barium-prefilled enema bags are used, the amount of water added and degree of subsequent shaking to achieve wettability should be standardized. The level marking on the enema bag should not be used to gauge the amount of water needed; resultant dilutions tend to be erratic. Liquid barium-filled enema bags should be kept on their sides because considerable settling can occur if bags are stored before use.
A 12% to 25% w/v barium suspension is commonly used for single-contrast barium enemas. The main requirement of the barium suspension is that it neither flocculates nor settles during the examination. Because the sedimentation rate depends, in part, on the amount and type of additives present, some products that are well suspended at higher concentrations settle readily when diluted. If there is doubt about a commercial product’s sedimentation rate, a radiograph obtained with a horizontal x-ray beam should reveal any settling tendencies.
Double-contrast barium enema suspensions should consist of relatively high barium concentrations but should still be sufficiently fluid to flow readily through enema tubing. Their viscosity is greater than that of the lower concentration barium formulations designed for single-contrast studies. The resultant mucosal coating should be uniform, without undue artifacts. The suspension should not dry out while an examination is in progress. These barium formulations are generally in the range of 60% to 120% w/v, with 85% being typical.
Even when all conditions are standardized, the subsequent mucosal coating can vary among practices because of variations in local water hardness and type of water used (distilled water or cold or hot tap water). Premixed liquid formulations are available to avoid these variations. The barium suspension is simply poured into an enema bag without further dilution.
Some radiologists perform colonic lavage before a barium enema. This lavage invariably results in water retention and subsequent dilution of the barium suspension. Barium manufacturers recognize this difference and market two different preparations; the one designed to be used after colonic lavage has a barium suspension with a slightly greater specific gravity.
Tumors can be difficult to detect in a segment involved by severe diverticulosis. This segment can be further studied if the double-contrast barium enema is followed by a methylcellulose enema.
Water-Soluble Contrast Agents
Indications
Water-soluble organic iodine compounds designed for the GI tract were introduced in the 1950s. Ever since, controversy has surrounded the relative merit and role of these agents. These compounds do not coat the GI mucosa; rather, they provide bowel visualization simply by passive filling of the intestinal lumen.
For most bowel examinations, experienced radiologists prefer barium suspensions. Some surgeons, however, are still being taught about the purported dangers of barium and insist on the use of water-soluble agents. Stimulation of peristalsis in postoperative patients and lack of radiographically visible sequelae of spill from the GI tract are reasons cited by some surgeons for their preference for water-soluble agents.
Water-soluble agents are indicated if an acute perforation is suspected. The examination generally confirms or excludes a perforation, with the realization that small perforations can be missed. Similarly, walled-off perforations or a perforation in an area of spasm can be difficult to detect, and it may be necessary to complete the examination with barium.
With a chronic or loculated perforation, the higher radiographic visibility of barium often yields more information than that obtained with water-soluble contrast agents. Thus, a chronic abscess or other cavity in continuity with the bowel lumen can be safely studied with barium. If there is a possible communication with the peritoneal cavity, however, water-soluble agents are preferred.
Meconium ileus and meconium plug syndrome can be treated with an iodinated contrast enema. The patient should be well-hydrated.
Some surgeons treat postoperative adynamic ileus with oral, full-strength, ionic contrast agents, but studies on these types of agents have been limited. Also, a hypertonic fluid proximal to a mechanical obstruction results in further distention.
Contrast Agents
In general, to achieve adequate radiographic opacification of most GI structures, at least a 60% solution of an ionic contrast agent is needed. The resultant iodine concentration is 282 to 292 mg/mL for the more commonly used commercial products, with a resultant osmolality of about 1500 mOsm/kg, or approximately five times that of serum. Because of this hyperosmolarity, fluid is drawn into the bowel lumen, and diarrhea is common after their use. These agents stimulate intestinal peristalsis, so faster visualization of the distal small bowel can be achieved than with barium sulfate. The need for a faster examination should be balanced against decreased radiographic contrast obtained with these agents. In general, intraluminal dilution leads to poor visualization of the small bowel.
Some commercial ionic contrast agents designed for oral use, such as the diatrizoate meglumine preparations Gastrografin and oral Hypaque, contain flavoring agents. These are preferred to the nonflavored products, which are designed primarily for IV use.
Nonionic contrast agents with an iodine concentration of approximately 300 mg/mL have an osmolality of 600 to 710 mOsm/kg, which is less than 50% of that of ionic agents. At this concentration, however, they are still hyperosmolar compared with serum.
Ideally, one of the nonionic agents should be used whenever a water-soluble agent is indicated for evaluation of the GI tract; for certain examinations, some radiologists use nonionic agents. If a perforation into the pleural or peritoneal cavity is suspected in an adult and aspiration is not a consideration, nonionic agents probably do not offer any major advantage over their ionic counterparts. Nevertheless, some leaks are better defined with barium than water-soluble agents. In studies of the GI tract in infants and children, in whom perforation is not an issue, barium rather than a water-soluble is the preferred contrast agent.
Negative Contrast Agents
When performing double-contrast GI studies, the cheapest second contrast agent is air. Excellent double-contrast esophageal views can be obtained if the patient swallows air together with the barium preparation.
One commercial preparation incorporated carbon dioxide directly in the barium suspension; when the patient drank this “bubbly barium,” carbon dioxide was released into the esophagus and stomach. The effect was similar to that of drinking a bottle of club soda, and this product did not achieve ready acceptance.
Effervescent tablets, granules, and powders are commercially available. They produce carbon dioxide on contact with water and most are satisfactory in achieving adequate gastric and duodenal distention. There is, however, considerable variation in their dissolution time. Most commercial effervescent powders and granules come in single-dose packages. In clinical use, the patient places the effervescent agent in the mouth and uses small amounts of water to wash it down. This is immediately followed by a barium suspension. The swallowed gas is used for double-contrast views of the stomach and duodenum.
Liquid effervescent agents, consisting of separate acid and base solutions, can be prepared locally by a hospital pharmacy. The acid portion consists of citric and tartaric acids and the base portion is sodium bicarbonate. A dose of 12 to 15 mL is satisfactory for most patients.
Carbon dioxide is used by some in place of air for double-contrast barium enemas and CT colonography. Carbon dioxide is absorbed faster than air. Whether a gas or air is used probably does not influence examination quality although, with all other factors remaining constant, colonic distention is less with carbon dioxide compared with air.
Some double-contrast preparations result in excessive gas bubbles. An antifoam agent should be added empirically if these occur on a regular basis. Although many commercial barium preparations already include such an agent, in some areas the amount is not sufficient. A commonly used antifoam agent is dimethyl polysiloxane (simethicone); the addition of 1.5 mL of simethicone (equivalent to 100 mg) is often sufficient to eliminate bubbles.
Gastrointestinal Computed Tomography Agents
The term double-contrast abdominal CT is used by some to signify the use of IV and oral contrast. However, this is a misuse of the traditional connotation of double contrast and is best avoided to prevent confusion.
Full-strength barium preparations should not be diluted to the low concentrations needed for CT. The barium particles settle out after ingestion of such a dilute solution, leading to inhomogeneous bowel lumen opacification. The uppermost part of a loop of bowel may not contain enough barium for visualization, and excess barium in the dependent portion results in streak artifacts.
Stable but low-concentration barium formulations specifically designed for CT are commercially available, with most of these brand names ending in -cat . Most CT barium products contain small particles that resist settling. Additives selected also prevent barium sedimentation. At the low barium concentrations used, barium particles do not coat the mucosa but simply provide lumen opacification.
An esophagus marked with a contrast agent aids in evaluating the mediastinum during chest CT. The low-concentration CT agents used in the rest of the GI tract do not opacify the esophagus long enough, although one option is to have the patient drink small sips of a conventional CT contrast agent before each scan. More convenient is a high-viscosity, low-concentration barium paste, which provides prolonged esophageal coating. Mucosal adherence by such a paste is long enough to allow a typical CT examination to be completed. Ingested water is often a useful and satisfactory contrast agent for evaluation of the GI tract during helical CT.
The traditional method of opacifying the stomach and small bowel is to have the patient drink approximately 500 mL of a dilute CT contrast agent several hours before the examination, with a similar amount ingested immediately before scanning. Ideally, such an agent should differentiate bowel from surrounding structures without introducing artifacts. Use of a dilute iodine solution or a barium suspension is generally a personal preference. A 1% to 3% w/v barium sulfate suspension or a 2% to 5% solution of Gastrografin or similar iodinated agent is typical. One refinement (granted, little practiced) is to use a 2.0% barium concentration for jejunal opacification and a slightly lower concentration for pelvic structures. With slower CT units, an iodine solution produces fewer streak artifacts than barium, a problem of little consequence with multislice CT. Commercial barium suspensions tend to taste better than iodine solutions, a factor when examining children and nausea-prone cancer patients.
The iodine taste can be masked by adding sugar and various fruit extracts; although essentially sugar-free iodine contrast is available, barium products, in general, contain less sugar than corresponding iodine contrasts. At the dilutions used, the iodinated solutions are hypoosmolar, but some patients still develop diarrhea. The poor taste and hence poor acceptance of iodinated agents by patients can be partly overcome by the empiric addition of a flavored juice such as Kool-Aid. At the dilutions used in CT, nonionic contrast agents do not have any real advantage over ionic agents.
With suspected pelvic disease, a contrast agent can be ingested the evening before the examination. Even if full-strength Gastrografin is ingested, overnight dilution in the bowel is sufficient to eliminate most streak artifacts. Better rectosigmoid opacification is obtained after ingestion of such a full-strength contrast agent than dilute barium, probably because hyperperistalsis is induced by the iodinated agent. Nevertheless, identifying the large bowel is less of a problem than identifying fluid-filled loops of small bowel on abdominal CT.
Both CT and MR enterography can detect Crohn’s disease with somewhat similar accuracy. Adequate CT bowel opacification can be achieved by using a 2% flavored barium suspension.
CT enteroclysis consists of bowel intubation and instillation of an iodinated contrast agent, dilute barium suspension, or methylcellulose suspension. Whether a positive or water-density agent is superior is not clear; an IV contrast agent to opacify bowel mucosa aids in lesion detection. Negative oral contrast agents designed specifically for CT enteroclysis have also become available. Also, with MDCT and coronal reconstruction now more readily available, retained bowel fluid often provides a sufficient marker, especially in dilated bowel loops.
If the imaging study is performed to evaluate a rectal lesion, a high-viscosity, low-volume barium paste may suffice; approximately 100 mL of a 3.6% w/v carboxymethylcellulose and 2% w/v barium sulfate paste mixture has been proposed in this clinical setting.
A basic question when protocoling an examination on multidetector CT is whether intraluminal water-density or fat-density contrast, or even a gas, is superior to a positive contrast agent. Positive bowel contrast creates artifacts, especially with maximal intensity projection images of vascular structures. Also, bowel wall enhancement by IV contrast is useful for detecting bowel wall thickening, and a positive intraluminal contrast can obscure subtle lesions. As a result, MDCT studies can be performed with oral water-density or even negative contrast agents. Although adequate for gastric and duodenal distention because of its absorption from the bowel, ingested water does not readily distend the distal small bowel; use of a carboxymethylcellulose or polyethylene glycol solution inhibits absorption and improves distention (compared to water). A preliminary study of simethicone-coated cellulose (SonoRx, Bracco Diagnostics, Monroe Township, NJ), developed for oral use in upper abdominal ultrasonography, found no significant advantage over oral water in abdominal CT.
In the past, a number of fat density products, such as mineral oil, corn oil, and milk, and even a paraffin emulsion, were proposed for CT use, but these have had limited application. Residual bowel gas often serves as a marker, especially in the colon. If a nasogastric tube is in place, air can be injected into the stomach and small bowel. If excessive amounts of gas are present, imaging with window settings slightly wider than usual is helpful. 2D colonography and 3D virtual colonoscopy require colonic distention with a contrast agent; typically, air is used, and carbon dioxide is used less often.
Adverse Reactions
Barium Sulfate
Barium sulfate is poorly soluble in water. The constipating tendency of barium products is well known to most radiologists. Through the judicious use of additives, this side effect is minimized in most current formulations, although barium impaction in the colon is still occasionally encountered.
Aspiration of small amounts of commercial barium formulations is of little clinical significance. After barium aspiration, most is cleared from major bronchi and trachea within hours, although some is retained in the interstitium and in macrophages. This residue is generally not visualized on radiographs. Alveolarization of barium, however, can result in prolonged retention. If aspiration is suspected clinically, nonionic, sterile, iso-osmolar iodinated contrast can be used rather than barium.
Hypersensitivity reactions during GI examinations are rare, although they have been known to occur. Although barium sulfate is inert, commercial formulations contain numerous known proprietary additives. These include stabilizing, flavoring, coating, and viscosity-varying agents and range from natural flavors and gums, such as lemon, pectin, and guar, to synthetic products, such as various methylcelluloses. Some radiologists may still be familiar with chocolate-flavored barium products, which are no longer used because of common allergies to chocolate. Anaphylaxis can be caused by carboxymethyl. The role of effervescent agents in these reactions is speculative. Methylparaben and similar compounds, used as preservatives, can induce hypersensitivity reactions, but barium manufacturers have replaced them with more innocuous preservatives in most commercial barium products.
Reactions appear to be more common during double-contrast than single-contrast studies. Most reactions are mild and consist of urticaria or pruritus, although erythema multiforme, respiratory complications, anaphylaxis, GI angioedema, and even death have occurred. Patients with asthma and severe food allergies appear to be at a slightly increased risk for these reactions, but the average radiologist will probably not encounter a hypersensitivity reaction to a barium product in a lifetime of practice.
The cause of hypersensitivity reactions during most barium studies is not known. In general, the incriminating agent is not sought and no testing performed in most patients who develop a reaction.
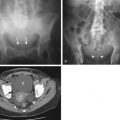
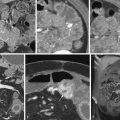
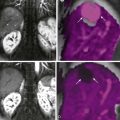
Esophageal perforation and spillage of barium into the mediastinum result in an inflammatory reaction, with barium persisting in the mediastinum for a prolonged period. Such prior extravasation can often be recognized radiographically as dense linear radiopacities, but no strong evidence exists that these sequelae have any clinical significance for the patient.
Most perforations associated with a barium enema occur in the rectum and are not immediately detected by fluoroscopy. Rectal perforations tend to result from injudicious insufflation of an enema balloon. A British survey over a 3-year period between 1992 and 1994 revealed a complication rate of 1 in 9000 and a death rate of 1 in 57,000. Although 10% of patients with a bowel perforation died (3 of 30), the mortality was 56% (9 of 16) in patients developing a cardiac arrhythmia.
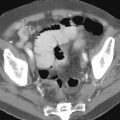
Spillage of barium into the peritoneal cavity can be secondary to a preprocedure perforation, such as in patients with ulcers. Some perforations, however, are associated with a barium study and can occur during an upper GI examination or barium enema, and even during enteroclysis. Initially, leukocytes are drawn into the peritoneal cavity, together with an inpouring of fluid. Profound hypovolemia develops if massive inpouring of fluid into the peritoneal cavity is untreated. Bacterial contamination during a perforation can result in overwhelming sepsis and shock within hours.
Immediate management of barium peritonitis includes infusion of large volumes of IV fluid. Antibiotics are administered because of associated bacterial contamination. Most patients undergo surgery, with an attempt made to evacuate barium from the peritoneal cavity. Invariably, barium crystals embedded on the peritoneal surface resist dislodgment. Attempts to remove barium particles with a wet sponge simply induces diffuse peritoneal bleeding.
Barium crystals incite an inflammatory reaction; eventually, these crystals become coated by a fibrin membrane, and extensive fibrosis and granulomatous tissue develop. Dense fibrosis can involve adjacent structures and, depending on location, subsequent ureteral obstruction or bowel deformity and stenosis develop. Perirectal fibrosis can narrow the rectosigmoid lumen and even mimic a carcinoma. Residual barium is identified with conventional radiography or CT. No evidence suggests that barium in soft tissues is a carcinogen ( Fig. 1-2 ).