Spinal vascular diseases are rare and constitute only 1% to 2% of all vascular neurologic pathologies. In this article, the following vascular pathologies of the spine are described: spinal arterial infarcts, spinal cavernomas, and arteriovenous malformations (including perimedullary fistulae and glomerular arterivenous malformations), and spinal dural arteriovenous fistulae. This article gives an overview about their imaging features on MRI, MR angiography, and digital subtraction angiography. Clinical differential diagnoses, the neurologic symptomatology, and the potential therapeutic approaches of these diseases, which might vary depending on the underlying pathologic condition, are given.
Spinal vascular diseases are a rare entity (1%–2%) within vascular neurologic pathologic conditions. Their clinical diagnosis rests mainly on MRI, although for a thorough understanding of the diseases involved and the therapeutic strategy, digital subtraction angiography (DSA) still is necessary. In this article we first discuss the normal vascular anatomy of the spine. Second, we describe causes, symptoms, and imaging findings of spinal cord ischemia. Third, we discuss spinal vascular malformations that typically lead to progressive spinal cord symptoms and myelopathy if not properly treated. Depending on the type of spinal vascular disease, initial symptoms may vary between acute or chronic onset. Pathophysiologic mechanisms include arterial ischemia, intramedullary or subarachnoidal hemorrhages, or subacute venous congestion leading to progressive myelopathy. The space-occupying nature of some of these lesions and a circulatory “steal” phenomenon are additional possible pathophysiologic mechanisms. Although acute manifestations of spinal vascular malformations are typically diagnosed early in the course of the disease, the subacute venous congestion might lead to unspecific neurologic symptoms, which in turn delay proper diagnosis. The aim of this article is to review the imaging features, clinical symptomatology, and potential therapeutic approaches of spinal vascular malformations and remind the neuroradiologists and referring physicians that the diagnosis and subsequent treatment of these treatable causes of severe and otherwise progressive neurologic deficits still remain a challenge.
Anatomy
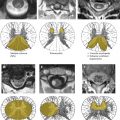
To interpret MRI and DSA findings of spinal vascular diseases, it is necessary to be aware of the normal arterial supply and venous drainage of the spine and spinal cord. Segmental arteries supply the spine, including the vertebral bodies, paraspinal muscles, dura, nerve roots, and the spinal cord with blood. Radicular arteries are the first branches of the dorsal division of the segmental arteries. The bony spine is supplied by anterior and posterior central arteries that come directly from the segmental and radicular arteries . A spinal radicular branch that supplies the dura and nerve root as a radiculomeningeal artery is present at each segment. From these radicular arteries, radiculomedullary and radiculopial arteries might branch, following the anterior or posterior nerve root to reach the anterior or posterior surface of the cord, where they form the anterior or posterior spinal artery .
In adult patients, not all lumbar or intercostal arteries have a radiculomedullary feeder, and their location for a given patient is not predictable. The anterior and posterior spinal arteries constitute a superficial longitudinal anastomosing system . The anterior spinal artery travels along the anterior sulcus and typically originates from the two vertebral arteries, whereas the typically paired posterolateral spinal arteries originate from the preatlantal part of the vertebral artery or from the posteroinferior cerebellar artery. These three arteries run from the cervical spine to the conus medullaris but are not capable of feeding the entire spinal cord. Instead, they are reinforced from the radiculomedullary arteries that derive from various (and unpredictable) segmental levels by anterior and posterolateral radiculomedullary arteries . The most well known of the anterior radiculomedullary arteries is the artery radiculomedullaris magna (ie, the Adamkiewicz artery). The anterior radiculomedullary arteries branch in a typical way to reach the spinal cord. The ascending branch continues along the direction of the radicular artery in the midline of the anterior surface. The descending branch, being the larger one at thoracolumbar levels, forms a hairpin curve as soon as it reaches the midline at the entrance of the anterior fissure .
The intrinsic network of the spinal cord arteries can be divided into central or sulcal arteries from the anterior spinal artery and into the rami perforantes of the vasacorona that supplies the periphery of the spinal cord and is derived from the anterior and paired posterolateral arteries .
The spinal cord is protected against ischemia by longitudinal and transverse anastomoses. Extradurally, interconnections between segmental arteries can compensate for a proximal occlusion of a radicular artery, whereas intradurally, the anterior and posterior spinal arteries represent a system of longitudinal anastomoses that is reinforced from different levels. The posterior and anterior arterial systems are interconnected at the level of the cone via the “basket” anastomosis and via transverse anastomoses of the intrinsic arteries that interconnect by the pial network of the vasocorona . Spinal infarctions are highly variable, and predictable vascular territories as present in the brain do not exist.
The venous drainage of the cord is via radially symmetric intrinsic spinal cord veins and small superficial pial veins that open into the superficial longitudinal median anastomosing spinal cord veins. These veins more or less follow the arteries (ie, the anterior and posterior median spinal vein) but have many anastomoses (including transmedullary anastomoses) that create a network with commonly more than one anterior and posterior vein . They may also use the roots as a vehicle to reach the epidural plexus and the extraspinal veins and plexus with a reflux-impeding mechanism within the dura mater . It is important to note that the transition of a median vein into a radicular vein shows the same hairpin shape as the artery. At the superior cervical part, they can run through the occipital foramen to connect to the vertebral plexus or the inferior dural sinus. Drainage of blood from the spine occurs through the valveless internal and external venous vertebral plexus, which is connected to the azygos and hemiazygos venous systems .
Spinal cord ischemia
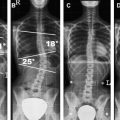
Compared with brain ischemia, spinal cord infarction is exceedingly rare and caused by more diverse etiologies . This rarity is mainly caused by the multiple anastomoses of the spinal cord, which supply arteries. Spinal cord supplying arteries are—for unknown reasons—not significantly affected by atherosclerotic vessel wall changes. Causes for acute arterial ischemia include surgery for aortoiliac occlusive disease , dissection of the aorta, vasculitis, fibrocartilaginous embolism , Caisson disease, lumbar artery compression , cardiac arrest and systemic hypotension, thrombosis/embolism, coagulopathies, and toxic effects of contrast medium. Acute ischemia has an acute onset; transverse cord symptoms and radicular pain are common initial complaints. Symptoms may include nerve root deficits or sphincter weakness. Typical clinical symptoms (ie, an anterior spinal artery syndrome) are the exception rather than the rule because of the high variability of spinal cord vascular supply. Common sites of spinal cord infarction are the thoracolumbar enlargement and the conus medullaris .
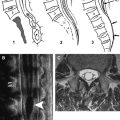
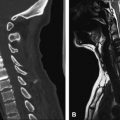
MRI is the diagnostic modality of choice; however, in the early phase it can show negative results on routine sequences, because only 50% of patients show an early demarcation of T2 hyperintensity of the spine within the first 24 hours. The role of MRI in the acute phase is to rule out other lesions that might go along with an acute nontraumatic transverse spinal lesion. On T2-weighted images, a pencil-shaped T2 hyperintensity is visible predominantly in the anterior part of the cord, typically sparing the outer rim of the cord ( Figs. 1 and 2 ) . In few studies, spinal diffusion–weighted images were performed in spinal cord infarction . These studies found an area of reduced diffusion after 8 hours ( Fig. 3 ).
Moderate swelling is generally present in the acute stage followed by contrast enhancement of the cord and the cauda equina in the subacute stage (typically after 5 days) ( Fig. 4 ). Contrast enhancement may persist for up to 3 weeks after onset . Abnormal signal or evolution of signal abnormalities within the vertebral body caused by associated vertebral body infarction often is encountered ( Fig. 5 ) . The field of view should be large enough to rule out aortic dissection.
Spinal cord ischemia
Compared with brain ischemia, spinal cord infarction is exceedingly rare and caused by more diverse etiologies . This rarity is mainly caused by the multiple anastomoses of the spinal cord, which supply arteries. Spinal cord supplying arteries are—for unknown reasons—not significantly affected by atherosclerotic vessel wall changes. Causes for acute arterial ischemia include surgery for aortoiliac occlusive disease , dissection of the aorta, vasculitis, fibrocartilaginous embolism , Caisson disease, lumbar artery compression , cardiac arrest and systemic hypotension, thrombosis/embolism, coagulopathies, and toxic effects of contrast medium. Acute ischemia has an acute onset; transverse cord symptoms and radicular pain are common initial complaints. Symptoms may include nerve root deficits or sphincter weakness. Typical clinical symptoms (ie, an anterior spinal artery syndrome) are the exception rather than the rule because of the high variability of spinal cord vascular supply. Common sites of spinal cord infarction are the thoracolumbar enlargement and the conus medullaris .
MRI is the diagnostic modality of choice; however, in the early phase it can show negative results on routine sequences, because only 50% of patients show an early demarcation of T2 hyperintensity of the spine within the first 24 hours. The role of MRI in the acute phase is to rule out other lesions that might go along with an acute nontraumatic transverse spinal lesion. On T2-weighted images, a pencil-shaped T2 hyperintensity is visible predominantly in the anterior part of the cord, typically sparing the outer rim of the cord ( Figs. 1 and 2 ) . In few studies, spinal diffusion–weighted images were performed in spinal cord infarction . These studies found an area of reduced diffusion after 8 hours ( Fig. 3 ).
Moderate swelling is generally present in the acute stage followed by contrast enhancement of the cord and the cauda equina in the subacute stage (typically after 5 days) ( Fig. 4 ). Contrast enhancement may persist for up to 3 weeks after onset . Abnormal signal or evolution of signal abnormalities within the vertebral body caused by associated vertebral body infarction often is encountered ( Fig. 5 ) . The field of view should be large enough to rule out aortic dissection.
Spinal vascular malformations
Multiple different classification schemes have been proposed for spinal vascular malformations. Recently, the Bicetre group classified spinal cord arteriovenous malformations (AVMs) into three main groups . The first group includes genetic hereditary lesions that are caused by a genetic disorder affecting the vascular germinal cells. Spinal cord malformations associated to hereditary hemorrhagic telangiectasia fall into this category. The second group includes genetic nonhereditary lesions that share metameric links, such as Cobb syndrome (or spinal AV metameric syndrome), that affect the whole myelomere. Patients typically present with multiple shunts of the spinal cord, the nerve root, bone, paraspinal, subcutaneous, and skin tissues. Klippel-Trenaunay and Parkes-Weber syndromes also belong to this group. The third group includes single lesions that may reflect the incomplete expression of one of the previous mentioned situations and includes spinal cord, nerve root, and lesions of the filum terminale.
For practical therapeutic reasons and because most spinal vascular malformations fall into the last group, we use a classification that is based on the vascular anatomy of the spinal cord and the inborn or acquired nature of the lesion . According to this classification, spinal vascular malformations can be differentiated, similar to vascular malformations of the brain, into true inborn lesions and the acquired lesions, the latter being dural AV fistulae, whereas AVMs and cavernomas constitute inborn lesions of the spine ( Table 1 ) . In contrast to the brain, in which capillary teleangiectases also belong to the inborn lesions, they have not been found in the spine and are not discussed further.
| Type | Etiology | Feeding artery | Draining vein | Pathophysiology | Age of onset (y) | Therapy | |
|---|---|---|---|---|---|---|---|
| AVM | Perimedullary fistula (types I–III) | Inborn | Radiculomedullary | Intramedullary and superficial spinal cord veins → epidural venous plexus (orthograde) | Intraparenchymal or subarachnoid hemorrhage, chronic venous congestion, space occupation | 20–40 | Type I: surgery; types II, III: coil or balloon embolization |
| Glomerular | <20 | Particle or glue embolization | |||||
| Iuvenile | <15 | Embolization and/or surgery | |||||
| Cavernoma | Inborn | NA | NA | Hemorrhage and progressive myelopathy | 20–60 | Surgery | |
| Dural AV fistula | Acquired | Radiculomeningeal | Radicular vein → perimedullary veins (retrograde) | Chronic venous congestion | 40–60 | Surgery or glue embolization | |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree